- LOGIN
- MemberShip
- 2026-06-10 19:52:16
- Company
- Why are generic firms rechallenging Precedex's patent?
- by Kim, Jin-Gu May 04, 2026 10:33am
- An invalidation trial has been filed against the premix formulation patent of Pfizer’s sedative ‘Precedex (dexmedetomidine).’ What is interesting is that generic drug manufacturers had already succeeded in circumventing this patent back in 2020. In this context, some interpret this move as the generic companies’ attempt to launch a premix formulation and glass vial product containing the same active ingredient.According to industry sources on the 1st, Ilsung IS recently filed an invalidation trial against Hospira regarding the Precedex premix formulation patent.Precedex, approved in June 2010, is used for ‘sedation of patients who are intubated early under intensive care and receiving mechanical ventilation.’ Pfizer acquired the product through its acquisition of Hospira in 2015. Subsequently, Pfizer Korea received additional approval for the premix formulation in 2017.There are two patents related to Precedex. The substance patent expired in January 2013, leaving the formulation patent, which is set to expire in June 2032, as the current target of generic challenges.Generics were launched sequentially following the expiration of the substance patent. Companies including Ilsung IS, Hanlim Pharm, Penmix, Kyongbo Pharmaceutical, Pharmbio Korea, Hana Pharm, and Jeil Pharmaceutical obtained approvals. However, due to the active premix formulation patent, products were released in ampoule form, requiring mixing the dexmedetomidine-containing drug into a basic intravenous solution.In 2020, JW Life Science and Dai Han Pharm attempted to circumvent the premix formulation patent. JW argued that its proprietary container did not fall within the patent scope of Pfizer’s Precedex and won both first and second trials. It subsequently launched premix generics with its partners Hanlim and Hana Pharm. However, these products used special plastic infusion containers rather than the original glass vial.What is notable this time is that Ilsung IS chose to file an invalidation trial rather than a circumvention trial, as JW had filed. Given that circumvention trials generally have higher chances of success at first instance, Ilsung appears to have chosen a more difficult path.This is interpreted as an attempt to secure not only the premix formulation but also the glass vial packaging format.The premix formulation patent describes a ‘ready-to-use liquid pharmaceutical composition containing dexmedetomidine or its pharmaceutically acceptable salt thereof, placed in a sealed glass container for parenteral administration.’ Since the patent explicitly specifies a “sealed glass container,” its circumvention allows the sale of premix formulations but not in glass vials, as doing so would risk patent infringement.Glass vials and plastic infusion bags each have clear advantages and disadvantages. Plastic IV bags have the advantage of a relatively lower risk of breakage. Their light weight, which facilitates transportation, is also cited as an advantage. However, compared to glass vials, there is a greater risk of drug molecule leaching or adsorption. Additionally, reduced stability during long-term storage is pointed out as a drawback.On the other hand, glass vials prevent the drug from reacting with the packaging material, allowing it to maintain its purity for a long time. They also remain stable during long-term distribution and storage. This is why glass vials are the standard for pharmaceuticals that require long-term stability, despite the higher risk of breakage.The domestic market size for dexmedetomidine-based sedatives is estimated at around KRW 19 billion annually. According to the Ministry of Food and Drug Safety, domestic production and import volumes increased from KRW 16 billion in 2022 to KRW 19 billion in 2023, but declined back to KRW 16 billion in 2024 due to reduced usage amid the healthcare crisis. Pfizer’s two products account for about 40% of the total supply as of 2024.
- Policy
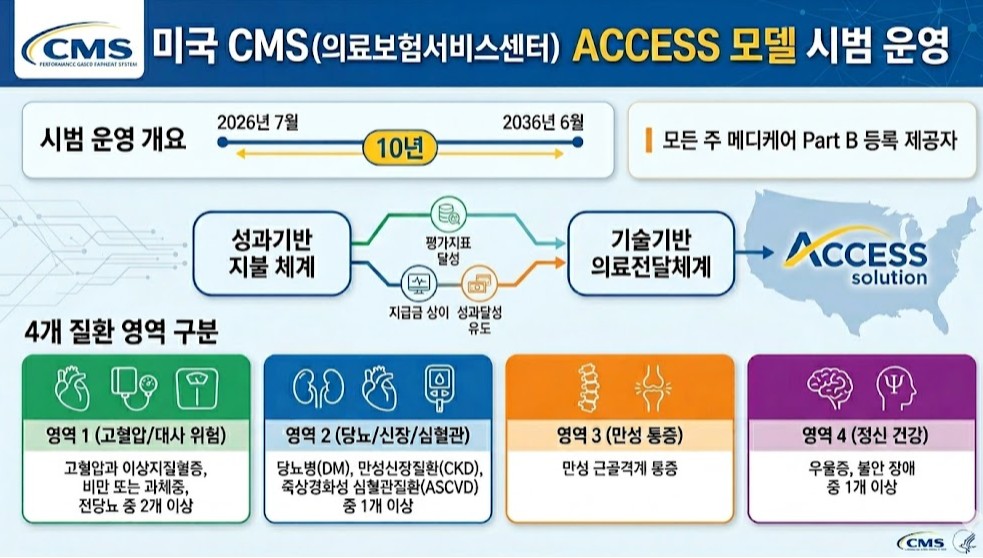
- US tests pay-for-performance to address fee-for-service limits
- by Jung, Heung-Jun May 04, 2026 10:33am
- To address the issues of an aging population and rising healthcare costs, the U.S. will launch a pilot program for a pay-for-performance system nationwide starting this July.This model links payments to the achievement of chronic disease management metrics and represents an attempt to overcome the limitations of the traditional fee-for-service (FFS) system.On the 30th, Soo-min Kwon, a senior researcher at the Health Insurance Review and Assessment Service (HIRA) Benefits Policy Research Division, published the policy implications of “The US’s New Performance-Based Payment Model for Chronic Disease Management” through an HIRA Issue report.The US Centers for Medicare & Medicaid Services (CMS) will pilot the ACCESS (Advancing Chronic Care with Effective, Scalable Solutions) model from July this year through June 2036. This model combines performance-based payment systems with technology-enabled healthcare delivery.All Medicare Part B providers across all states are eligible. Patients are categorized into four groups;▲ those with at least two of hypertension, dyslipidemia, obesity/overweight, or prediabetes, ▲ those with at least one of diabetes (DM), chronic kidney disease (CKD), or atherosclerotic cardiovascular disease (ASCVD); ▲ chronic musculoskeletal pain; ▲ those with at least one of depression or anxiety disordersParticipating institutions provide integrated services including clinical consultations, lifestyle interventions, education, and medication management. They also conduct clinical improvement activities, collecting outcome data through remote monitoring tools and implementing interventions using digital health platforms.Final control and minimum improvement targets are set for each evaluation metric, with targets adjusted annually over the treatment period.Institutions receive payments for managing patients over a one-year period. During the initial six months of treatment, they receive 50% of the annual payment in monthly installments, while the remaining 50% is paid based on whether performance targets were met and whether the patient received services for the same condition from other healthcare providers.The payment structure differs between the initial treatment year and subsequent management phases to incentivize early achievement of clinical outcomes.Kwon explained, “This represents a shift from service-based to performance-based payment systems, redesigning reimbursement criteria around clinical results rather than service quantity.”She added, “In line with global trends in payment system reform, this could serve as a reference model for designing integrated chronic disease payment systems in Korea and ensuring the sustainability of the national health insurance finances.”
- Company
- ‘Zipalertinib’ targets the Exon 20 lung cancer mkt…presenting new trt option
- by Son, Hyung Min May 04, 2026 10:33am
- While treatment options for EGFR exon 20 insertion mutation non-small cell lung cancer (NSCLC) remain limited, the potential for a change in the therapeutic environment is being proposed as the oral targeted therapy ‘zipalertinib’ enters the U.S. regulatory approval process.With consistent efficacy confirmed in global clinical trials and analysis of Asian patient populations amid limited existing treatments, zipalertinib is garnering attention as a competing product to ‘Rybrevant (amivantamab),’ the only approved medicine in this space.According to industry sources on the 4th, the U.S. Food and Drug Administration (FDA) recently accepted a New Drug Application (NDA) for zipalertinib. The drug’s target population includes patients with locally advanced or metastatic NSCLC with EGFR exon 20 insertion mutations whose disease has progressed following platinum-based chemotherapy. The goal review date under the Prescription Drug User Fee Act (PDUFA) is February 27, 2027.Zipalertinib is a next-generation irreversible EGFR tyrosine kinase inhibitor (TKI) being co-developed by Taiho Pharmaceutical of Japan and Cullinan Therapeutics of the U.S. This drug is designed to selectively inhibit mutant EGFR while minimizing impact on wild-type (normal) EGFR.The NDA is based on results from Part 2b of the Phase 1/2 REZILIENT1 study.The REZILIENT1 study included 176 patients with locally advanced or metastatic NSCLC harboring EGFR exon 20 insertion mutations as the primary efficacy evaluation group. The study evaluated the clinical efficacy of zipalertinib in patients whose disease progressed after platinum-based chemotherapy.Notably, 51 of the total patients had prior experience with Rybrevant, confirming zipalertinib’s potential as a subsequent treatment option after existing targeted therapies. Patients received 100 mg of zipalertinib orally twice daily, with objective response rate (ORR) and duration of response (DOR) analyzed as primary endpoints.Clinical results showed an ORR of 35.2% in the Zipalertinib group, with a median DOR of 8.8 months. It is noteworthy that responses were also confirmed in patients with prior Rybrevant treatment. In that specific patient group, the ORR was 30%, suggesting its viability as a follow-up treatment option.Zipalertinib results were presented at ESMO Asia 2025, held in Singapore last year.The efficacy in Asian patients did not differ significantly from that in the global patients.According to a subgroup analysis presented at the ESMO Asia 2025, the ORR for the Asian patient group was 33%, similar to the 37% observed in the non-Asian group. The duration of response (DOR) was 8.3 months and 10.5 months, respectively, while progression-free survival (PFS) showed almost identical patterns at 9.5 months and 9.0 months.In terms of overall survival (OS), the median has not yet been reached in the Asian patient group, whereas it was 24 months in the non-Asian group.Regarding safety, the primary adverse events included paronychia, rash, dry skin, diarrhea, and stomatitis, most of which were Grades 1–2 and manageable.Limited treatment environment…”Oral drug option is highly significant”Janssen’s RybrevantThe development of treatments for NSCLC with an EGFR exon 20 insertion mutation has faced significant challenges. Unlike treatments targeting Exon 19 deletions or Exon 21 L858R mutations, Exon 20 insertion mutations have diverse subtypes, making drug design structurally difficult.Takeda’s Exkivity, which garnered high expectations as an oral targeted therapy, received conditional approval based on a 28% ORR in early trials. However, it was withdrawn from the global market after failing to prove PFS improvement in the confirmatory Phase 3 trial (EXCLAIM-2).Poziotinib, previously developed, was also suspended after failing to meet efficacy expectations and experiencing toxicity issues in Phase 2 trials.As a result, Janssen’s Rybrevant is now the only treatment approved in this therapeutic area. However, as an intravenous-based therapy, it has been noted for limitations regarding administration convenience and treatment persistence.Given such a treatment gap, zipalertinib, which can be administered orally, is being evaluated as a new alternative because it offers manageable safety while increasing mutation selectivity compared to previous agents. The industry is focusing on the possibility that the Exon 20 mutation treatment landscape will transition from a monotherapy-centered environment to a more competitive one, depending on future approvals.Professor Ross Soo of the National Cancer Centre Singapore explained, “Zipalertinib demonstrated efficacy in Asian patients equivalent to that of the global patient population,” and added, “Because it is an oral drug, it is significant in terms of patient accessibility and treatment persistence.”
- Policy
- Kwangdong secures first generic exclusivity for Tagrisso
- by Lee, Tak-Sun May 04, 2026 10:33am
- Domestic pharmaceutical companies are intensifying their push into the KRW 110 billion non-small cell lung cancer (NSCLC) treatment market centered on Tagrisso (osimertinib). Following Chong Kun Dang’s first generic approval in January, Kwangdong Pharmaceutical has also joined the ranks of companies obtaining first generic exclusivity, marking the start of a full-scale race to capture the market.Kwangdong Pharmaceutical’s ‘Ktinib Tab’ Approved… Second after Chong Kun DangAccording to the Ministry of Food and Drug Safety, Kwangdong Pharmaceutical obtained first generic exclusivity on April 30 for its osimertinib mesylate product ‘Ktinib Tab (40mg, 80mg).’ This is the second such approval following Chong Kun Dang’s ‘Otinib Tab,’ which received approval on January 27.The drug’s indications include, as a monotherapy: ▲Adjuvant treatment following complete resection in patients with NSCLC harboring EGFR exon 19 deletions or exon 21 (L858R) substitution mutations ▲Treatment of patients with unresectable locally advanced (Stage III) NSCLC harboring EGFR exon 19 deletions or exon 21 (L858R) substitution mutations whose disease has not progressed during or after platinum-based chemoradiotherapy ▲First-line treatment of patients with locally advanced or metastatic NSCLC harboring an EGFR exon 19 deletion or exon 21 (L858R) substitution mutation ▲Treatment of patients with EGFR T790M-positive locally advanced or metastatic NSCLC who have previously been treated with an EGFR-TKI.Additionally, as a combination therapy, it is indicated for the first-line treatment of patients with locally advanced or metastatic non-squamous NSCLC harboring EGFR exon 19 deletions or exon 21 (L858R) substitutions, in combination with pemetrexed and platinum-based chemotherapy. These indications are identical to the original Tagrisso.Like Chong Kun Dang, Kwangdong succeeded in circumventing Tagrisso’s formulation patent (set to expire January 2035), thereby securing first generic exclusivity. As a result, both companies will hold a roughly 9-month market exclusivity period starting from the expiry of the substance patent.First, generic exclusivity is granted when a company successfully challenges a patent and meets the criteria of being among the first to file for approval. Both companies achieved patent circumvention in September last year by winning a passive scope confirmation trial regarding the formulation patent. AstraZeneca has since appealed, and a lawsuit to cancel the ruling is currently ongoing at the Intellectual Property High Court.The approval applications were also submitted simultaneously to the MFDS, meaning both companies met the requirements for exclusivity.The exclusivity period for Ktinib (during which sales of identical drugs are restricted) is set from December 28, 2033, to September 27, 2034.Since the original drug Tagrisso’s substance patent remains valid until 2033, immediate market entry is not possible. However, both companies plan to enter the market immediately upon patent expiry, block latecomers, and capture the KRW 110 billion market.Tagrisso vs. Leclaza… generics join the competitionCurrently, the domestic NSCLC treatment market is dominated by AstraZeneca’s original drug Tagrisso and Yuhan’s domestic new drug Leclaza, which are competing fiercely for market share. With Chong Kun Dang and Kwangdong preparing to launch generics, the market landscape is expected to become even more complex.A pharmaceutical industry insider analyzed, “Given the high commercial potential of the product, generic companies are continuing to challenge the patents. With Chong Kun Dang and Kwangdong securing generic exclusivity, the generic market is highly likely to evolve into an initial two-horse race starting in 2033
- Company
- Biosimilars expand access to osteoporosis treatment
- by Choi Da Eun Apr 30, 2026 08:24am
- The paradigm for osteoporosis treatment is shifting. There is a growing recognition that the condition should be approached as a ‘long-term management disease’ aimed at preventing fractures and maintaining quality of life, rather than merely slowing the decline in bone density. In particular, with the recent emergence of denosumab biosimilars improving treatment access, changes are being detected in the market as well.Professor Yoo Mee Kim of the Department of Endocrinology and Metabolism at Catholic Kwandong University International St. Mary’s Hospital emphasized, “Osteoporosis has almost no symptoms, so it is often first detected as a fracture before patients even realize they have the condition. Prevention and early treatment before fractures occur are most important.”Professor Kim has been serving as president of the Korean Society for Bone and Mineral Research since January 2026. She graduated from Yonsei University College of Medicine and earned her master’s and doctoral degrees from the same university. She currently heads the Endocrinology and Diabetes Center at International St. Mary’s Hospital. She is an active member of the Korean Endocrine Society, Korean Diabetes Association, Korean Society for the Study of Obesity, and other organizations.Professor Yoo Mee Kim. Director of the Endocrinology and Metabolism Center at International St. Mary’s HospitalOsteoporosis progresses without symptoms…fracture is the first signalAn osteoporotic fracture refers to a fracture that can occur even with a minor impact. It can even occur from a mild fall or coughing. If such a fracture occurs, it is necessary to suspect osteoporosis and undergo testing.Professor Kim explained, “Since osteoporosis causes no specific symptoms even as bones weaken, patients are often unaware of their condition. Consequently, many cases are only discovered after fractures occur in the wrist, spine, or hip.”The problem is what happens after a fracture. It does not simply end with a broken bone. She said, “When a fracture occurs, activity levels drop sharply and can lead to muscle loss and worsening chronic diseases. In elderly patients, reduced immunity may also increase the risk of complications such as pneumonia.”Hip fractures, in particular, are directly linked to survival. Professor Kim said, “When a hip fracture occurs in the elderly, it can lead to long-term hospitalization and complications, greatly increasing mortality. Even if a relatively minor fracture, such as a wrist fracture, occurs, it already means that the bones have weakened. Because fractures often begin at the wrist and progress to the spine and hip, early response is extremely important.”Osteoporosis increases sharply among women and after the 50s…menopause is a key variableOsteoporosis is particularly common in women. Professor Kim explained, “Female hormones play a role in protecting bones, but after menopause, hormone levels drop sharply, and bone loss progresses rapidly. From the 50s onward, prevalence and fracture risk increase significantly.”Recently, due to the aging population, the incidence of osteoporosis in men has also been on the rise. However, because hormonal decline occurs gradually in men, the onset of the disease tends to be slower than in women.Osteoporosis is diagnosed through Dual-energy X-ray absorptiometry (DXA). If a fracture has already occurred, or if there are factors such as advanced age, low body weight, family history, chronic disease, or polypharmacy, the patient is classified as high risk even if bone density is relatively good.Professor Kim emphasized, “Osteoporosis is diagnosed when the T-score, which indicates the degree of bone density loss compared with young adults, is -2.5 or lower. However, it is important to evaluate fracture risk alongside bone density measurements rather than relying solely on bone density values.”A shift in treatment paradigms… now capable of improving bone densityIn the past, osteoporosis treatment focused primarily on slowing progression. Today, major treatment options have diversified to include bisphosphonates, denosumab, and anabolic agents (PTH analogs, romosozumab).Bisphosphonates have long been the first-line treatment. Currently, denosumab-based formulations account for over 40% of the total osteoporosis treatment market share.Denosumab is characterized by a stronger effect through a mechanism that blocks bone resorption by inhibiting osteoclast formation. Since the emergence of denosumab, many changes have occurred in the osteoporosis treatment market.Professor Kim said, “While existing treatments were limited to slowing bone density loss, the recent emergence of more potent drugs has made it possible to actually increase bone density.”Daewoong Pharmaceutical’s osteoporosis treatment StobocloEmergence of biosimilars…raises expectations for improved treatment accessRecently, Prolia (denosumab) biosimilar ‘Stoboclo’ has been rapidly gaining traction in the market. It was first launched in March 2025. While the original drug exceeded KRW 200,000 when it first received reimbursement, Stoboclo was launched at about KRW 100,000, roughly half that level.When insurance reimbursement is applied, the actual cost borne by patients is only about KRW 180 per day and about KRW 5,400 per month on average. As a result, it is widely regarded as having significantly improved patient access. Just 10 months after its launch, it has established itself in the market with cumulative sales of KRW 11.8 billion.Professor Kim said, “If a drug has similar efficacy and safety, cost burden ultimately becomes an important factor from the patient’s perspective. Because osteoporosis treatment must be continued for a long period, a lower drug cost can have a positive impact on treatment persistence.”She continued, “Osteoporosis is not merely a disease of aging but a condition requiring active management. Preventing fractures through early diagnosis and continued treatment is of the utmost importance.”
- Company
- Outpatient Rx mkt growth slows amid reimbursement curbs and price cuts
- by Chon, Seung-Hyun Apr 30, 2026 08:24am
- The outpatient prescription market has shown steady performance this year. Analysts attribute this to a steady stream of flu patients and the expansion of insurance coverage for medications utilizing new technologies, such as oral anticancer drugs, which have spurred growth in the outpatient prescription market. However, the growth rate has slowed compared to previous years, partly due to reimbursement restrictions and drug price cuts.According to the pharmaceutical research institution UBIST on the 29th, the outpatient prescription market reached KRW 5.3484 trillion in Q1, a 3.5% increase year-on-year. This marks the third-largest quarterly market size on record, following Q3 and Q4 last year.Quarterly outpatient prescription volume (Unit: KRW 100 million, Source: UBIST)The outpatient prescription market has steadily expanded over time, growing 40.1% over five years from KRW 3.8173 trillion in Q1 2021.Following the COVID-19 endemic transition, persistent influenza and respiratory illness cases have sustained market growth. The Korea Disease Control and Prevention Agency reported that influenza alerts were issued continuously from January through last month, exceeding the epidemic threshold. For six months starting from Week 40 of last year (September 29–October 5), the incidence rate exceeded the seasonal epidemic threshold for the 2025–2026 season, 9.1 cases per 10,000 people.Proportion of Influenza-like Illness patient rates for the 2025–2026 season (Unit: Persons; Source: Korea Disease Control and Prevention Agency).Even without extraordinary factors like pandemics, the outpatient market is structurally set to grow due to aging populations and increasing chronic disease prevalence.Recently, expanded insurance coverage for innovative therapies has further driven market growth.For instance, AstraZeneca’s anticancer drug Tagrisso recorded KRW 54.2 billion in outpatient prescriptions in Q1, a 26.2% increase year-on-year. Tagrisso is used for EGFR-mutated metastatic non-small cell lung cancer (NSCLC). Since 2024, its reimbursement has expanded alongside Yuhan’s Leclaza for first-line treatment of certain mutation-positive NSCLC.Although anticancer drugs are typically used more in inpatient settings, Tagrisso’s oral formulation has significantly boosted outpatient prescriptions. Its outpatient sales more than doubled from KRW 22.7 billion in Q1 2023 to current levels. Leclaza also saw Q1 prescriptions reach KRW 19.1 billion, more than tripling in three years.However, Q1 growth appears to have slowed somewhat compared to previous years. This marks the lowest growth rate in five years, following a 2.9% year-over-year increase in prescription sales in Q1 of 2021. The outpatient prescription market grew by 10.9% year-over-year in Q1 2022 and expanded by 9.9% in Q1 2023. In Q1 2024 and last year, the market grew by 5.5% and 5.3% year-on-year, respectively. The strong upward trend continued as infectious disease patients continued to emerge amid the COVID-19 pandemic and endemic phase.Government drug pricing policies are cited as a major factor. Repeated reimbursement restrictions and price cuts have dampened market expansion.For example, the brain function enhancer choline alfoscerate market shrank from KRW 146.2 billion in Q1 last year to KRW 103 billion this year, a 29.5% drop in one year.Since September 21st, the patient co-insurance rate has increased from 30% to 80% for non-diagnosed patients, sharply reducing prescriptions. In Q4 last year, the prescription market for choline preparations stood at KRW 103.7 billion, a 29.9% decrease from the previous quarter’s KRW 147.9 billion, and remained at a similar level in Q1 this year. This means that over the past six months, the prescription market for choline preparations has shrunk by more than KRW 90 billion due to the reimbursement cut.In Q1, the outpatient prescription market for mugwort extract-based drugs stood at KRW 25.3 billion, a 18.7% decrease year-on-year. Mugwort extract-based drugs are natural medicines made from mugwort and are used to improve gastric mucosal lesions, bleeding, redness, and swelling associated with acute and chronic gastritis. Following the government’s reimbursement reevaluation, the insurance price ceiling for 74 types of mugwort extract-based medicines was reduced by an average of 14.3% starting in February, causing the prescription market to contract.Unexpected variables also impact the market. The COVID-19 drug Paxlovid recorded KRW 47.7 billion in prescription sales in Q3 last year, but this figure shrank to KRW 2.1 billion in Q1 this year. Paxlovid is primarily prescribed to high-risk patients at risk of severe progression. Initially, the government purchased and supplied it free of charge, but in June 2024, the government halted the supply of new batches, shifting to regular prescriptions through general medical institutions.With the decision on Paxlovid’s health insurance reimbursement in October 2024, it officially entered the prescription market. The reimbursement price was set at KRW 941,940, with a 5% co-insurance rate. Due to the fluctuating number of COVID-19 patients and Paxlovid’s high price, there was significant variation in prescription volume. Paxlovid’s prescription sales of KRW 47.7 billion in Q3 last year accounted for 0.9% of total outpatient prescription sales.
- Opinion
- [Reporter’s View] Beyond generics to new drugs
- by Choi Da Eun Apr 30, 2026 08:24am
- The Korean pharmaceutical industry’s push into new drug development has reached a turning point. Companies are moving away from a business model centered on generics and contract manufacturing (CMO), expanding into next-generation anticancer drugs and obesity treatments, signaling a clear shift toward innovation-driven growth to improve the company’s structure with a focus on new drugs.Whereas it was once common to acquire candidate compounds from global pharmaceutical companies and focus on late-stage clinical trials or commercialization, there is now a growing movement to take the lead directly, from early discovery through Phase 1 and 2, and even late-stage clinical trials. This strategy aims to go beyond simply securing a pipeline to internalize the entire new drug development cycle.This shift is led by major pharmaceutical and biotech companies such as Hanmi Pharmaceutical, Daewoong Pharmaceutical, JW Pharmaceutical, HK inno.N, Celltrion, and Samsung Bioepis. These firms are diversifying portfolios across oncology, obesity, and immunology by expanding pipelines of new drug candidates.A notable trend is the growing emphasis on in-house development. Companies are increasingly attempting to internalize the entire R&D cycle, moving beyond simple pipeline acquisition toward full-spectrum innovation, from early-stage compound discovery through Phase 1 and 2 clinical trials, and on to late-stage clinical trials. This marks a strategic shift away from a focus on in-licensing toward internalizing the entire R&D cycle.For example, Celltrion and Samsung Bioepis are accelerating the development of antibody-drug conjugate (ADC) anticancer drugs, thereby expanding their footprint into the field of biopharmaceuticals. This is an attempt to leap to the next stage by leveraging the technological capabilities and capital accumulated through biosimilars.In particular, obesity treatments and anticancer drugs are emerging as next-generation growth engines. Although these are fields with fierce competition in the global market, they are also areas where the landscape can rapidly shift depending on technological innovation. Multi-mechanism-based obesity treatments and next-generation ADC anticancer drugs are cited as representative fields where domestic companies can compete based on their technological prowess.Hanmi Pharmaceutical is taking one of the most aggressive approaches, expanding both bispecific antibody oncology pipelines and obesity drug programs simultaneously. The company is aiming to secure global competitiveness by leveraging multi-mechanism obesity drugs and anticancer platforms as its two main pillars.JW Pharmaceutical has demonstrated the ability to independently advance late-stage development by bringing a gout treatment candidate to Phase 3. This is significant because it demonstrates that these companies have internalized development capabilities not just to acquire a pipeline, but to reach the stage immediately prior to commercialization.HK inno.N is strengthening its in-house R&D capabilities by combining the introduction of obesity drug candidates with an open innovation strategy. This is a hybrid strategy that leverages external technology while aiming to secure an independent pipeline in the long term.Chong Kun Dang is developing innovative drugs through its subsidiary Achelra, while Yuhan Corporation is advancing biologics via ImmuneOncia. These specialized subsidiaries aim to improve research efficiency and success rates.The strengthening of new drug development capabilities reflects both technological advancement and improved financial stability among Korean companies. However, the industry is still in a transitional phase. Some companies are balancing between in-licensing and in-house development, but cases where clinical trials successfully lead to commercialization remain limited. A gap between expectations and actual outcomes persists.Nevertheless, the reason domestic pharmaceutical companies are shifting course is clear. With generic drug price cuts and intensifying competition among contract manufacturing organizations (CMOs), the limits to growth under the existing business model have become evident. Ultimately, the prevailing view is that it is difficult to secure a meaningful foothold in the global market without proprietary new drugs.The fact that domestic pharmaceutical companies have entered a stage where they are charting their own course in new drug development is clearly a significant change. With accumulated manufacturing expertise, global expansion experience, and strengthening R&D infrastructure, the industry’s structure is gradually evolving.Whether the challenge of shifting the R&D structure from a focus on generics and biosimilars to new drugs will remain a temporary trend or become a turning point for securing a presence in the global market depends on clinical outcomes and commercialization capabilities.This effort is not merely business expansion but a structural transformation of the industry. Market evaluation should therefore consider both outcomes and the process of capability building. If this trend continues, the leap from a generic powerhouse to a powerhouse in new drug development will no longer be a distant prospect.
- Company
- Boryung loses first trial in Lenvima + Keytruda combo patent dispute
- by Kim, Jin-Gu Apr 30, 2026 08:24am
- Boryung has lost the first trial in a patent dispute over the combination therapy of the liver cancer treatment Lenvima (lenvatinib) and Keytruda (pembrolizumab). Although Boryung had already launched a generic after overcoming listed patents, the latest ruling has left the company facing marketing risks for key indications. To mitigate this risk, the company must now seek a reversal of the ruling by appealing to the Intellectual Property Court.According to industry sources on the 29th, the Intellectual Property Trial and Appeal Board recently issued a ruling of partial dismissal and partial rejection in a passive scope confirmation trial filed by Boryung against Eisai Korea and Merck regarding an unlisted patent (No. 10-2662228) related to the Lenvima + Keytruda combination therapy. The IPTAB also dismissed the invalidation trial request for the same patent.This patent covers the ‘combination of a PD-1 antagonist and a VEGFR/FGFR/RET tyrosine kinase inhibitor for treating cancer.’ Merck and Eisai registered this patent in April 2024. The patent covers the combination therapy of Lenvima and Keytruda for the first-line treatment of advanced endometrial cancer and advanced renal cell carcinoma.Among Lenvima’s indications - ▲ locally recurrent or metastatic progressive differentiated thyroid cancer, ▲first-line treatment for unresectable hepatocellular carcinoma, ▲combination therapy with pembrolizumab for the treatment of patients with advanced endometrial cancer, ▲combination therapy with pembrolizumab as first-line treatment for advanced renal cell carcinoma - this case is related to the 3rd and 4th indications.Industry analysis suggests that Boryung has effectively suffered a decisive defeat in the first-instance trial over the Lenvima + Keytruda combination patent. However, the ruling does not block the sale of Boryung’s Lenvima generic, Lenvanib. Previously, Boryung had bypassed or invalidated all four listed patents for Lenvima except the substance patent, and received marketing approval for Lenvanib in February last year. It then secured reimbursement listing in July and launched the product. Currently, Boryung’s Lenvanib is the only Lenvima generic on the market.However, due to this ruling, Lenvanib can no longer be used to treat advanced endometrial cancer and advanced renal cell carcinoma. This is because if Lenvanib is used for these indications, Boryung could face patent infringement lawsuits from Eisai and Merck.Eisai has already demonstrated a proactive stance in protecting its patent rights, as evidenced by its filing of an active scope confirmation trial regarding the scope of rights during a dispute over another use patent (10-1470653) against Boryung. Although that case concluded with Eisai voluntarily withdrawing its claim following Boryung’s victory in the invalidation trial, had the invalidation trial yielded the opposite result, it could have served as grounds for a patent infringement lawsuit.At present, Lenvima’s core market remains thyroid cancer and hepatocellular carcinoma (indications 1 and 2). However, Boryung’s concerns have grown as Lenvima’s focus in the global market is rapidly shifting toward combination therapy with Keytruda for endometrial cancer and renal cell carcinoma.To resolve this patent risk, Boryung must now decide whether to appeal. Industry observers expect that, given the growing importance of combination therapy in Lenvima’s global revenue mix, Boryung is unlikely to accept the first-instance defeat without a challenge.
- Company
- Generic companies win ‘Jardiance Duo’ unlisted patent 1st instance trial
- by Kim, Jin-Gu Apr 30, 2026 08:24am
- Product photo of Jardiance TabFourteen pharmaceutical companies, including Chong Kun Dang, have won a favorable ruling in a patent dispute regarding unlisted patents for Jardiance Duo (empagliflozin + metformin).Analysis suggests that following their victory in the unlisted patent dispute for the Jardiance monotherapy late last year, this subsequent win for the metformin combination therapy significantly mitigates patent risks for generic products.According to pharmaceutical industry sources on the 29th, the Intellectual Property Trial and Appeal Board (IPTAB) issued a decision on the 28th in favor of the invalidation trial filed by Chong Kun Dang and others against Boehringer Ingelheim regarding the use patent for Jardiance Duo. In addition to Chong Kun Dang, generic companies participating in this trial include Genupharma, Pharmgen Science, Dongkoo Bio & Pharma, Daehan Nupharm, Dongwha Pharm, Young poong Pharmaceutical, Withus Pharmaceutical, JW Pharmaceutical, Medica Korea, Boryung, Aprogen Biologics, Korea Pharma, Hanmi Pharm, and Aju Pharm.This patent is concerning the use patent for the combination therapy of empagliflozin and metformin, which is set to expire in November 2027. Notably, it was not listed in the Ministry of Food and Drug Safety’s (MFDS) patent Green List.Boehringer Ingelheim has registered two listed patents and seven unlisted patents related to Jardiance with the Korean Intellectual Property Office. Generic manufacturers successfully bypassed one of the listed patents in 2019. Based on that ruling, companies challenging the patent obtained generic product licenses and launched their generics in October last year, around the time of the expiration of the substance patent.However, the seven unlisted patents have been analyzed as potential obstacles to the sale of Jardiance generics. While they do not affect product authorization, selling products without overcoming unlisted patents carries the risk of patent infringement litigation.Boehringer Ingelheim’s unlisted patents related to Jardiance include ▲three use patents for empagliflozin single-agent ▲one use patent and one formulation patent for empagliflozin + metformin ▲one use patent for empagliflozin + linagliptin ▲one use patent for the triple combination therapy of empagliflozin + linagliptin + metformin.Korea's fourteen pharmaceutical companies, including Chong Kun Dang, Genupharma, Pharmgen Science, Dongkoo Bio & Pharma, Daehan Nupharm, Dongwha Pharm, Young poong Pharmaceutical, Withus Pharmaceutical, JW Pharmaceutical, Medica Korea, Boryung, Aprogen Biologics, Korea Pharma, Hanmi Pharm, and Aju Pharm have won a favorable ruling in a patent invalidation trial concerning unlisted patents for Jardiance Duo (empagliflozin + metformin).Among these, generic companis, including Genuone Sciences, Chong Kun Dang, Korea Prime Pharm, Boryung, and Huons, successfully invalidated one of the use patents for the Jardiance monotherapy formulation in November last year. Boehringer Ingelheim has appealed to the Patent Court following this first-instance defeat.With the sequential invalidation of the use patents for both the Jardiance monotherapy and the metformin combination product, industry analysts believe generic companies have resolved a substantial portion of the patent risks associated with product sales.However, generic manufacturers have failed to overcome the unlisted use patent for Esglito (empagliflozin + linagliptin). Companies including Genuone Sciences, Boryung, Dongkook Pharm, Medica Korea, Aprogen Biologics, Korea Prime Pharm, Daehwa Pharm, GC Biopharma, and Aju Holdings filed invalidation and passive negative scope confirmation trials against the Esglito patent. These companies received dismissal rulings in February and March of this year, respectively.Additionally, decisions regarding two cases of use patents for Jardiance and one case of formulation patent for Jardiance Duo is pending.
- Company
- Darzalex SC Inj, expansion of three indications approved
- by Eo, Yun-Ho Apr 30, 2026 08:23am
- Product photo of Darzalex SC The application for the subcutaneous (SC) formulation of ‘Darzalex’ is expanding.According to industry sources, Janssen Korea obtained additional approvals from the Ministry of Food and Drug Safety (MFDS) on the 24th for three indications of Darzalex SC (daratumumab) Inj.This drug’s specific indications are ▲combination therapy with bortezomib + lenalidomide + dexamethasone (DVRd) for patients with newly diagnosed multiple myeloma who are eligible for autologous stem cell transplant ▲combination therapy with bortezomib + lenalidomide + dexamethasone (DVRd) for patients with newly diagnosed multiple myeloma who are ineligible for autologous stem cell transplant ▲combination therapy with pomalidomide + dexamethasone for patients with multiple myeloma who have received at least one prior therapy, including a proteasome inhibitor and lenalidomide.With these indications, Darzalex SC can now be utilized in a variety of ways in combination with ‘Revlimid (lenalidomide).’The newly added indications demonstrated efficacy in various Phase 3 clinical studies, including PERSEUS, CEPHEUS, and APOLLO.In these studies, Darzalex SC met primary endpoints, including progression-free survival (PFS).Meanwhile, Darzalex is the first human monoclonal antibody authorized for multiple myeloma. This drug’s active pharmaceutical ingredient, daratumumab, is designed to recognize and directly bind to CD38, a surface glycoprotein overexpressed on multiple myeloma cells.In January, the light chain (AL) amyloidosis indication for Darzalex SC Inj passed the Health Insurance Review and Assessment Service's (HIRA) Pharmaceutical Reimbursement Evaluation Committee. Currently, this drug is undergoing drug price negotiations with the National Health Insurance Service (NHIS).