- LOGIN
- MemberShip
- 2026-06-10 11:33:43
- Policy
- Will PPC injections make a comeback? MFDS review underway
- by Lee, Tak-Sun Jun 10, 2026 08:41am
- AI-generated imagePPC (phosphatidylcholine) injections, which dominated the aesthetic and obesity market in the late 2000s but subsequently disappeared, are preparing for a spectacular comeback with a formal marketing authorization in hand. In the past, PPC injections were approved as an adjunct treatment for hepatic coma caused by liver cirrhosis, but they became controversial due to their widespread "off-label" use for fat reduction.According to industry sources on the 9th, the Ministry of Food and Drug Safety (MFDS) recently initiated a Good Clinical Practice (GCP) inspection regarding the application for a change in product approval for "Amiraju Inj. 5mL," a PPC-based drug. The GCP inspection is a critical final hurdle where the MFDS verifies the reliability of clinical data just before granting drug approval. This implies that the MFDS has entered the final stages of the product approval review.Amipharm has been known to have applied for approval. The company has developed a proprietary formulation technology that completely eliminates surfactants and utilizes only pure soybean-derived phosphatidylcholine (PPC) to induce the "apoptosis" (programmed cell death) in fat cells. Unlike earlier formulations that forcibly caused cell necrosis, the new composition selectively induces the death of fat cells only, thereby significantly reducing pain and swelling while maintaining a semi-permanent fat reduction effect.Amipharm successfully completed a domestic Phase III clinical trial involving 252 patients with submental (double chin) fat, demonstrating a high improvement rate of approximately 73.7%. Following this, the company applied for formal product approval as a ‘localized fat reduction agent’ and is known to be s proceeding simultaneously with manufacturing license approval procedures.PPC injections like Lipobin Inj were originally approved as prescription drugs for the treatment of hepatic coma associated with liver cirrhosis. However, rumors that American pop star Britney Spears had lost weight using PPC injections fueled a craze in aesthetic clinics and plastic surgery practices, where the products were widely prescribed off-label to reduce fat under the chin and in the upper arms.The product's popularity, however, did not last. Manufacturers ultimately failed to demonstrate post-marketing efficacy for the originally approved indication of hepatic coma, while growing criticism over the indiscriminate use of PPC injections as obesity treatments further intensified. As a result, the products were eventually withdrawn from the Korean market.An industry official commented, “It is highly meaningful that PPC, a substance that once disappeared from the aesthetic market, is returning as a formally approved therapeutic product after demonstrating its safety. If the MFDS inspection proceeds smoothly, official marketing authorization is certainly within reach.”
- Policy
- OTC Formulation changes to become easier in KOR
- by Lee, Tak-Sun Jun 09, 2026 10:01am
- Going forward, companies seeking approval for over-the-counter (OTC) drugs that use the same route of administration as an already approved or notified product but differ only in formulation will be required to submit significantly fewer review documents. By establishing specific criteria for what constitutes a ‘minor formulation difference,’ a concept that had previously remained ambiguous, the revision is expected to substantially reduce the burden associated with product development and approval changes for pharmaceutical companies.The Ministry of Food and Drug Safety (MFDS) announced on the 5th that it had issued a public notice of a proposed amendment to the ‘Regulations on the Approval, Notification, and Review of Pharmaceutical Products.’ The revision was introduced to address shortcomings in the current pharmaceutical approval, notification, and review system while rationalizing existing regulations.The core objective of the amendment is to streamline the administrative procedure by clearly specifying which supporting documents are required when changing the formulation of OTC drugs.Previously, if a company sought approval for a new product that had the same route of administration as an already approved or notified drug but differed in formulation, it was required to submit extensive review materials, such as head-to-head trial data or bioequivalence study data.However, under the revised proposal (Appendix 1), if the difference between the proposed OTC formulation and the existing formulation is considered ‘minor’ and falls within the same specified category, existing data for the drug may be used in place of the required review data.According to examples provided by the MFDS, eligible formulation changes include: Changes among tablets (plain tablets, sugar-coated tablets, and non-enteric film-coated tablets); capsule formulations; granules and powders; Changes among ointments, creams, gels, topical solutions, and topical powders; Changes among oral liquid formulations (excluding emulsions and suspensions) and syrups; Changes between cataplasms and adhesive patches.The pharmaceutical industry expects the measure to significantly reduce both development time and costs when diversifying OTC formulations to better align with consumer preferences and improve dosing convenience.In addition to simplifying formulation-change procedures, the amendment includes a number of broader regulatory rationalization measures.For medical high-pressure gases such as oxygen, a new exemption provision has been established, allowing safety validation data (long-term stability and accelerated stability study results) to be replaced by prior use cases for already approved or registered products. For orphan drug products whose active pharmaceutical ingredient quality data have already been submitted, multiple specifications for the active ingredient may be listed if it can be demonstrated that they do not affect the quality of the finished pharmaceutical product.In addition, a new provision stipulates that when only the name of a general test method in an official compendium has changed without any modification to the test procedure itself, the approval or notification will be deemed automatically updated without requiring a separate amendment procedure. In addition, efforts to refine legal terminology into easy-to-understand Korean, such as replacing Japanese-style Chinese characters like ' sujaedoen (recorded)’ with 'sillin(included)’ and ' jeokbu (judgment of suitability)’ with 'judgment of conformity/non-conformity’ were also includedThe MFDS will accept public comments on the proposed amendment until August 4, 2026. The revised regulation will take effect immediately upon promulgation and will apply to pharmaceutical products for which manufacturing, marketing, or import approval and notification applications (including amendments) are first submitted after the effective date.
- Policy
- GLP-1 obesity drugs may receive 'concern for misuse and abuse' designation
- by Lee, Tak-Sun Jun 09, 2026 10:01am
- AI-generated imageAs the Ministry of Food and Drug Safety (MFDS) pursues the designation of GLP-1 obesity treatments as 'drugs of concern for misuse and abuse,' it has released an analysis showing that the public interest benefits of protecting citizens' rights to know and to health are significant.Consequently, GLP-1 obesity treatments containing liraglutide, semaglutide, and tirzepatide are highly likely to be designated as drugs subject to concern for misuse and abuse following a collection of industry opinions. Once designated as a drug subject to concern for misuse and abuse, warning phrases will become mandatory on packaging, and sales without a prescription will be prohibited even in areas exempt from the separation of prescribing and dispensing.The MFDS issued an administrative notice on the 5th regarding a partial amendment to the "Regulations on the Designation of Drugs Subject to Concern for Misuse and Abuse," which features these primary measures. The MFDS has also concluded its economic and social feasibility review through a regulatory impact assessment report.6.3-fold increase in supply...severe safety insensitivity including inappropriate prescribing for pediatrics and pregnant womenThe background of the MFDS is to tighten regulatory oversight, which lies in the recent explosive growth of the obesity treatment market and the resulting indiscriminate misuse and abuse.According to the regulatory impact assessment report, the volume of GLP-1 receptor agonist obesity treatments (liraglutide, semaglutide, and tirzepatide) imported and supplied domestically increased by 6.3-fold (approximately KRW 901.0 billion) in just one year, skyrocketing from around KRW 169.9 billion in 2024 to approximately KRW 1.0709 trillion in 2025.The problem is that instances where these medicines are abused for simple cosmetic weight loss by individuals with normal body weights or are illegally distributed are surfacing consecutively. In fact, it was verified that 69 cases of inappropriate prescribing occurred for pediatric patients under the age of 12 and 194 cases for pregnant women. At the same time, crackdowns by relevant authorities revealed a sharp increase in the detection of online false or exaggerated advertisements and illegal distribution.Notably, pharmacy supply volumes in 'areas exempt from the separation of prescribing and dispensing', such as remote islands and mountainous regions lacking physicians, have also experienced a steep spike. For the flagship product 'Wegovy', supply volume to pharmacies in these exempt regions increased by 975.7% year-over-year.History of Designations for 'Drugs Subject to Concern for Misuse and Abuse'There are two core measures newly established through the introduction of this regulation. First, it restricts the sale of these obesity treatments so that they can be dispensed only upon a physician's prescription at pharmacies nationwide, including those located in areas exempt from the separation of prescribing and dispensing. The intent is to block indiscriminate acquisition channels that bypass prescriptions fundamentally.Second, pharmaceutical manufacturers and importers must clearly print the phrase "Drug Subject to Concern for Misuse and Abuse" on product containers, packaging, and package inserts. Through this, the agency plans to heighten awareness regarding drug misuse and abuse among both medical professionals and consumers.Direct costs to the pharmaceutical industry estimated at KRW 24 Million... "Public interest benefits overwhelmingly outweigh compliance costs"The MFDS found that the economic burden imposed on the pharmaceutical industry by this regulation remains marginal. Currently, there are a total of 24 authorized obesity treatment products containing the relevant active ingredients in South Korea. Upon calculating the costs of modifying labeling materials, such as replacing printing ink and copper plates, for the 12 items currently launched and actively selling in the market, it was estimated that a one-time direct cost of approximately KRW 2 million per product, totaling KRW 24 million (equivalent to an annualized net cost of KRW 2.9 million), would be incurred. Six products that have not yet been launched were excluded from the cost-incurring factors.The MFDS emphasized, "Compared to private losses such as material modification expenses for certain regulated entities or revenue declines for pharmacies, the public interest benefits of preventing side effects from inappropriate drug administration and protecting the citizens' right to know and right to health are significant."The MFDS plans to gather both positive and negative opinions on this administrative notice through June 26, 2026, and to implement the policy immediately. Consumers and the industry will be granted a one-year transitional grace period to use pre-manufactured packaging materials produced before the implementation date.
- Policy
- ‘MFDS will review new drugs faster than the US and Europe’
- by Lee, Tak-Sun Jun 08, 2026 08:52am
- Joon-Soo Shin, Director General of Pharmaceutical Safety Division, delivers a keynote speech at the 2026 Spring Conference of the Korean Society of FDA Regulatory Science on June 5.“Even if a multinational pharmaceutical company submits applications simultaneously in the United States, Europe, and Korea, Korea could be the first country to grant approval.” The Ministry of Food and Drug Safety (MFDS) has set an ambitious goal of elevating its review capabilities and regulatory systems to a global top-tier level, to ultimately make Korea the first country in the world to approve innovative new drugs.On June 5, Joon-Soo Shin, Director General of the Pharmaceutical Safety Division at the MFDS, unveiled the agency’s Five-Year Comprehensive Plan for Pharmaceutical Safety Management during a keynote address at the 2026 Spring Conference of the Korean Society of Food, Drug and Cosmetics Regulatory Sciences, held at the Hoam Faculty House Convention Center of Seoul National University under the theme “Interdisciplinary Perspectives and the Immediate Markett Entry System in the AI Era.”During the speech, Shin repeatedly emphasized that the “240-Day Approval and Review System” would represent a major milestone not only for patients and pharmaceutical companies, but also for the MFDS as the regulatory authority.Shin said, “Achieving a 240-day review timeline is both highly challenging and highly significant for the MFDS. If a multinational pharmaceutical company were to submit applications simultaneously in the U.S., Europe, and Korea, Korea would be positioned to grant approval sooner than the U.S. (300 days) or Europe (365 days), which poses a significant burden on our part.”Shin continued, “To achieve this timeline, we must thoroughly prepare our review capabilities and build a system capable of completing the process within 240 days, including all periods required for supplementary submissions. We will ensure smooth implementation through close collaboration with industry, continuous review procedures, and greatly expanded pre-submission consultations.”To support this initiative, the MFDS implemented detailed operational guidelines on June 1 and has already begun applying them in practice. Rather than simply eliminating procedural steps, the agency introduced a process innovation that converts sequential reviews into parallel and simultaneous reviews. In addition, the dedicated review team was expanded from 15 to 20 personnel. A communication framework has also been established to improve predictability by providing checklists in advance and continuous feedback.The substantial reduction in review timelines is expected to maximize palpable benefits for both patients and pharmaceutical companies. Shin explained, “By reducing the average review period from 420 days to 240 days, patients with rare and intractable diseases gain 180 days, roughly 6 months,of valuable treatment time.” He added that, from an industry perspective, “In addition to the promotional advantages associated with accelerated approval, faster review timelines will be particularly beneficial for companies that develop biosimilars, as speed is of the essence in securing market competitiveness.”Addressing concerns raised by some observers regarding potential declines in review quality, Shin firmly rejected the notion. “This initiative does not involve omitting data reviews; rather, it is a procedural improvement. Therefore, this will not compromise review quality. We will allocate the majority of our personnel resources to safety evaluations.”In fact, the MFDS previously implemented an unprecedented expansion of its review workforce, hiring 195 full-time reviewers with the revenue generated by raising new drug application fees to KRW 410 million 2 years ago. Shin stated, “We were initially concerned about recruiting talent because the positions are based in Osong, but applications exceeded expectations, with competition among highly competent reviewers reaching 12-to-1. We will provide the support necessary for these reviewers to fully demonstrate their capabilities and plan to assign our most highly competent reviewers to handle approximately 80% of new drug evaluations.”
- Policy
- Opdivo's reimbursement for gastric cancer set to expand soon
- by Jung, Heung-Jun Jun 08, 2026 08:52am
- Ono Pharma Korea's immuno-oncology drug Opdivo (nivolumab) has entered drug price negotiations for expanded reimbursement scope in gastric cancer.Opdivo will undergo the negotiation process alongside its competitor, MSD Korea's Keytruda (pembrolizumab), for the same indication.According to industry sources on the 5th, Keytruda and Opdivo, which passed the Health Insurance Review and Assessment Service (HIRA) Pharmaceutical Reimbursement Evaluation Committee last March, have entered negotiations for mismatch repair-deficient (dMMR) or microsatellite instability-high (MSI-H) gastric cancer.Opdivo was the first cancer immunotherapy to secure reimbursement as a first-line treatment for HER2-negative gastric cancer back in 2023. It was included in the reimbursement listing for gastric cancer, a field where therapeutic options had been limited. It dominated the market by specifically targeting HER2-negative patients, who take a significant proportion of gastric cancer cases.Keytruda, a competitor to Opdivo, expanded its reimbursement coverage last January across 11 indications spanning 9 cancer types, including gastric cancer. At that time, Keytruda expanded its reimbursement coverage to include both HER2-positive and HER2-negative gastric cancers in the first-line setting.The two cancer immunotherapies are continuously expanding their reimbursement scope in gastric cancer. Unlike the previous process for HER2 gastric cancer reimbursement, these drugs are both undergoing the listing process for the 'dMMR·MSI-H' gastric cancer population.Although dMMR·MSI-H patients account for a relatively small proportion of overall gastric cancer cases, this will add a therapeutic option for patients who previously failed to meet existing reimbursement criteria, such as specific PD-L1 expression thresholds.Notably, both dMMR and MSI-H represent genetic alterations that make this subpopulation among the most clinically challenging to treat within gastric cancer.If both Opdivo and Keytruda obtain expanded reimbursement, treatment access is expected to be strengthened for gastric cancer patients who have previously been left behind. For this reason, medical societies have reportedly submitted official opinions directly to the authorities, calling for the expansion of reimbursed indications for gastric cancer.Meanwhile, Opdivo is undergoing expansion of reimbursed indications beyond gastric cancer. During last April's Cancer Disease Review Committee (CDRC) meeting, reimbursement criteria were established for Opdivo combination therapy with Bristol Myers Squibb (BMS) Korea's Yervoy (ipilimumab) as a first-line treatment for unresectable or metastatic hepatocellular carcinoma (HCC).The company also applied for an expanded reimbursement for this drug as a first-line combination therapy for non-small cell lung cancer (NSCLC), but it failed to pass the CDRC threshold.
- Policy
- "Fastest review in the world…K-bio global competitiveness"
- by Lee, Tak-Sun Jun 05, 2026 09:36am
- Minister Yu-Kyoung Oh of the Ministry of Food and Drug Safety (MFDS) highlighted milestones of her ongoing regulatory reform initiative, stating a robust commitment to driving the global expansion of the South Korean pharmaceutical-biotech sector. In an exclusive interview marking the anniversary of Daily Pharm, Minister Oh said 'regulation is not operational hurdles' but 'protective boundaries and a support system,' highlighting goals of establishing a virtuous cycle of patient safety and industrial growth.Addressing a long-standing industry bottleneck, the MFDS finalized its 'Medical Product Marketing Authorization and Review Innovation Plan,' which dramatically compresses the review timeline for novel drugs to a world-class standard of 240 days. The plan was officially enacted on June 1, Daily Pharm's founding anniversary. The initiative deploys 195 highly qualified new personnel across core technical divisions to significantly elevate review expertise.In accordance with the innovation plan, the MFDS began accepting applications for 'pre-submission face-to-face consultations' on June 1. Selected applications will be managed under the dedicated review teams. To maximize administrative efficiency, the MFDS is shifting away from legacy sequential review workflows, previously necessitated by workforce constraints, toward a concurrent, parallel review matrix where multiple specialists evaluate technical dossiers simultaneously across distinct disciplines.Furthermore, the MFDS is accelerating its digital transformation by integrating artificial intelligence (AI) into the complex, voluminous drug approval process. Following the establishment of the 'Food and Drug AI Transformation Task Force' in February, the MFDS launched a three-year, multi-phase initiative dedicated to constructing an advanced AI-driven regulatory review framework.The initial phase will be launched this October, utilizing AI tools to evaluate CMC (Chemistry, Manufacturing, and Controls) and quality data for both active pharmaceutical ingredients (APIs) and finished drug products, with plans to progressively expand AI utilization across the entire novel drug evaluation spectrum by 2028.Minister Oh emphasized that the international credibility of a domestic regulatory body directly correlates with a nation's industrial competitiveness. The MFDS recently achieved full functional listing on the WHO List of Authorities (WLA) register for both medicines and vaccines. Excluding traditional benchmark jurisdictions such as the US, EU, Canada, and Switzerland, South Korea's MFDS is the sole regulatory authority globally to provide comprehensive validation across all functional parameters for both therapeutic sectors.Based on this enhanced global reputation, the MFDS was recognized as a reference regulatory authority by the UAE in January and Lebanon in April, expanding South Korea's footprint to seven reference nations, which also include the Philippines, Paraguay, Egypt, Ecuador, and Nigeria.The following is a summary of the interview with Minister Oh.Q. The pharma-biotech industry holds high expectations for the MFDS's regulatory overhauls. What are the ongoing initiatives and public-private collaborative strategies for this year?The MFDS has consistently driven regulatory overhauls to fortify public trust while empowering commercial developers. It is encouraging to see positive feedback from the field. This year, we remain dedicated to actively capturing industry feedback and executing agile reforms that yield immediate, palpable results.Primarily, we have instituted a 240-day review cap for novel therapeutics, compressing what was historically a lengthy and frustrating process to match the fastest international standards, effective June 1.By expediting the review cycle for innovative drugs, biosimilars, and cutting-edge medical devices, we aim to expand patient access to treatment while significantly enhancing the global market standing of our domestic life sciences sector.Furthermore, the CDMO Act, designed to underpin the biomanufacturing ecosystem, will be implemented this December. The framework introduces a new export manufacturing registration registry to stimulate cross-border commerce, complemented by structured regulatory consultations on GMP compliance for contract manufacturing facilities to accelerate K-Bio's international expansion.The 'Innovative Product Pre-consultation Hotline' has also been operational since March, offering a streamlined, one-stop advisory portal that provides customized feedback and preliminary evaluations from early discovery through the formal filing stage.Additionally, we have improved orphan drug designation criteria to facilitate rapid market entry for rare disease therapeutics and expanded expedited import mechanisms for self-administered medications to ensure timely patient access to critical therapies.These systemic overhauls are fundamentally rooted in gathering public opinions. We continue to operate our policy-linking open forums to translate corporate and public feedback directly into regulatory directives, with a parallel focus on emerging entrepreneurs and small business operators.Since May, we have initiated a series of regional forums across six major domestic clusters to capture localized industrial needs. This feedback will be synthesized into a formalized regulatory roadmap slated for public disclosure this July.We plan to intensify our stakeholder-centric administrative model by ensuring absolute transparency in our policy implementation metrics and running continuous post-hoc audits to confirm that these reforms are operating successfully on the ground.Q. Following recent breakthroughs such as the WHO Listed Authorities (WLA) listing, the international stature of the MFDS has risen. What are your strategic plans for international engagement to further promote export growth?In the global market, commercial products are evaluated alongside the capabilities and scientific rigor of the regulatory bodies that oversee them. Because regulatory trust maps directly to asset valuation and national competitiveness, the MFDS has successfully become a premier global regulator.Achieving a comprehensive listing across all functional blocks on the WHO WLA register for both medicines and vaccines establishes South Korea as the only nation outside the traditional quad of the US, EU, Canada, and Switzerland to secure such a validation. This milestone serves as an objective, international endorsement of the expertise of our regulatory framework. Based on this global achievement, we secured reference authority status in the UAE and Lebanon this year, bringing our total to seven reference nations.This institutional credibility translates into reduced transactional friction, lower operational costs, and smoother market entry for domestic developers expanding overseas, demonstrating that regulatory diplomacy serves as a practical trade facilitator.Furthermore, South Korea's selection as host of the International Coalition of Medicines Regulatory Authorities (ICMRA) Summit this November reflects our growing leadership. We intend to spearhead international regulatory harmonization in emerging areas such as AI and advanced therapeutics, reinforcing our regulatory leadership to champion the global commercialization of K-Pharma actively.Q. To sustain the 240-day novel drug review mandate, 195 new personnel have been deployed. Are the core operational pillars, including the pre-submission face-to-face consultations launching this June, executing on schedule?The revised framework went live on June 1, structured around three core operational shifts designed to grant patients rapid access to innovative therapies while preserving safety margins.There are three main items to this innovation plan. First, we will introduce a preemptive regulatory support service starting from the 'submission dossier preparation phase' to support systematic data compilation by the industry. In the 'application submission phase', a new 'pre-submission face-to-face consultation' will be introduced to preemptively identify and resolve hurdles to marketing authorization, and a dedicated review team will be established to build a tailored, close communication system throughout the entire marketing authorization and review process. In the review phase, we plan to dramatically increase review speed by transitioning from sequential reviews, which were previously unavoidable due to limited review personnel, to concurrent and parallel reviews, where multiple reviewers evaluate dossiers simultaneously within their respective specialized disciplines.Starting June 1, the industry can apply for the 'pre-submission face-to-face consultation', and the submitted items can receive marketing authorization within 240 days through relevant procedures after the consultation.The MFDS will continue to promote innovation in medical product marketing authorization and review without a hitch, ensuring greater safety while accelerating review speed to the fastest in the world, thereby spearheading the global competitiveness of the K-Bio industry and expanding treatment opportunities for the public.Q. What is the current status of integrating AI technology to shorten the marketing authorization review timeline, and what are the countermeasures regarding data security concerns?The MFDS established an Information Strategy Plan (ISP) last year to preemptively utilize AI in massive and complex pharmaceutical marketing authorization and review workflows, and created the 'Food and Drug AI Transformation Task Force' this February to centralize its capabilities.A project to construct an AI-driven review system will be executed for three years starting this year, launching services this October with CMC and quality (drug substance, drug product) evaluation data for novel drugs, and progressively expanding the scope to cover comprehensive novel drug reviews by 2028.We anticipate that reviewers will be supported during the marketing authorization and review process by AI functionalities, such as large-scale document translation, summarization, historical precedent searches, and draft generation of review reports, enabling a more meticulous yet expedited review process.Furthermore, the AI review system will install AI servers and models within the secure internal government network to prevent the leakage of proprietary submission data and ensure impeccable security management.The MFDS will lay the foundation for securing international-standard review quality by optimizing marketing authorization and review workflows using AI, such as automating simple, repetitive tasks in real time.Q. Achieving two objectives of 'ensuring safety' and 'industry revitalization' concurrently does not seem easy. As the longest-serving Minister, we would like to hear about your expertise and the challenges you face.It is common for people to view regulation as a financial cost or an operational hurdle. Indeed, there were such aspects in the past, and the logic that emphasizing safety directly translates into a barrier to industrial development used to prevail. However, with technological innovations like AI increasingly part of daily life, the regulatory paradigm is shifting.Sustainable industry growth is only possible when it is backed by safety. We frequently observe historical cases in which a failure to secure safety downstream has led to catastrophic risks, such as large-scale product recalls or severe erosion of brand equity. Safety is like a perimeter fence. When the fence is strong, the industry can grow healthily within it. We are striving to ensure that MFDS regulatory validation guarantees product credibility and translates into product competitiveness, serving as a springboard to clear global regulatory barriers.Another point I would like to emphasize is that when novel technologies like AI emerge, the industry can easily fall into disarray if there are no laws or frameworks in place. Preemptive codification of rational baselines by the regulator is necessary to enable market players to operate with confidence.The MFDS is supporting the formation and challenges of new industries by proactively providing benchmarks, such as enacting the world's first Digital Medical Products Act and publishing review guidelines for generative AI-driven medical devices.Furthermore, if domestic standards converge with global standards, they can serve as a powerful promotional mechanism to drive international market expansion. To this end, the MFDS is broadening the horizon of regulatory diplomacy by leading the Asia-Pacific Food Regulatory Authority Summit (APFRAS), hosting the 'International Coalition of Medicines Regulatory Authorities (ICMRA) Summit (November), and driving the launch of the Global Cosmetics Regulatory Authority Summit (GCORAS) this September.The MFDS will ensure that creative innovation can occur freely within the protective boundary of safety. We will cultivate a rational regulatory ecosystem where regulation opens pathways for innovation to advance safely.Q. Lastly, Daily Pharm was founded on June 1, 1999, and marks its 27th anniversary this year. The objectives pursued by a specialized media outlet like Daily Pharm, public health enhancement and industrial development, seem to parallel your own considerations. Could you offer your advice on the future direction and role of specialized pharmaceutical journalism?First, I extend my sincere congratulations to Daily Pharm on its 27th anniversary. Over the past 27 years, Daily Pharm has dedicated itself to enhancing public health and advancing the pharmaceutical industry. It has served as a benchmark for specialized media, delivering information from a balanced perspective so the public can easily understand the rapidly changing pharma-biotech industry and the institutional frameworks driven by advanced technology.The opinions and sharp analysis from the pharmaceutical industry are a major asset to the MFDS's policy formulation, and you continue to perform your role as a constructive critic. In particular, functioning as a policy microscope that does not overlook even the minor issues that might go unnoticed, you are helping the MFDS draw closer to the public.We hope Daily Pharm will continue elevating the public's right to know by professionally identifying the latest trends in the pharmaceutical industry, at a level the public can understand. The MFDS will also collaborate with Daily Pharm through communication to collectively build a world where food and drug safety becomes an everyday reality.
- Policy
- Accelerated review may attract first-ever global new drug filings in Korea
- by Lee, Tak-Sun Jun 05, 2026 09:36am
- Could a paradigm shift be underway in which global pharmaceutical companies submit new drug applications to Korea's Ministry of Food and Drug Safety (MFDS) before filing in the United States or Europe? Expectations are rising as the MFDS launches a major regulatory service reform aimed at shortening review timelines for new drugs and biosimilars to 240 days, potentially positioning Korea as a preferred first-filing market.The MFDS, led by Commissioner Yu-Kyoung Oh, announced that it has implemented a comprehensive ‘Medical Product Approval and Review Innovation Plan,’ effective June 1, to accelerate patient access to innovative therapies while supporting the global expansion of Korea's biotechnology industry.A key component of the initiative is a comprehensive regulatory support framework covering the entire development process—from early-stage product preparation through final approval. To support this effort, the MFDS recruited approximately 200 additional reviewers and established dedicated review teams by specialty area. The agency also introduced a ‘simultaneous and parallel review system’ designed to significantly improve efficiency.Previously, due to limited personnel, the initial request for additional information typically occurred 87 days after submission (60 working days). Under the new system, the first feedback is expected to be provided within 25 days of submission, enabling companies to identify and address deficiencies much earlier in the review process.MFDS officials expect the new review framework to work in tandem with the recent elimination of the CCP requirement, which had long been viewed as a barrier for imported medicines entering the Korean market.During a press briefing held on June 2, Young-Joo Kim, Head of the Regulatory Science Policy Promotion Division, said, “With the CCP requirement removed, shortening review timelines for new drugs could become a true paradigm shift. We may soon see global pharmaceutical companies submitting applications simultaneously in the U.S., Europe, and Korea—or even choosing Korea as the first country in the world for regulatory filing. Given that possibility, our review organization feels a significant sense of responsibility and pressure.”On the 2nd, the Ministry of Food and Drug Safety held a briefing with journalists regarding innovation plans for medical product approval and review. (From the left) Young-joo Kim (Head, Regulatory Science Policy Promotion Team), Seung-ryeol Ryu (Director, Advanced Medical Device Division), Kyung-seung Shin (Director, Medical Device Approval Division), Hyun-jung Park (Director, Biopharmaceutical Approval Division), Nam-soo Kim (Director-General, Pharmaceutical Approval Management Division), So-hee Kim (Director, Cardiovascular and Neurology Products Division), Ho-Jung Kim (Director, Recombinant Protein Products Division), Yong-seok Koh (Director, Advanced Drug Quality Division II)Efforts to improve communication and reduce trial-and-error throughout the review process will also be significantly strengthened. A pre-submission checklist outlining key requirements prior to filing will be provided, while the existing consultation process, which was previously limited to a single meeting, will be formalized into Pre-NDA Meetings, guaranteeing at least two opportunities for discussion before submission.At the briefing, Nam-Soo Kim, Director-General of the Pharmaceutical Approval Management Division, explained, “The checklist will be particularly valuable for venture companies and small-to-medium-sized pharmaceutical firms that often have limited regulatory resources. Even before the official implementation date, companies with products that are sufficiently prepared and request a Pre-NDA Meeting will receive support with the goal of completing the review within 240 daysHowever, challenges do remain. The expertise of the massively increased personnel needs to be elevated in a short period. Regarding this, Kyung-seung Shin, Director of the Medical Device Division, stated, "It is a fact that it is difficult to immediately deploy new personnel to review new drugs. As we have selected highly experienced reviewers with extensive backgrounds in pharmaceutical companies, we will quickly deploy them to the field through thorough training and internal mentoring to fill the gap."The pharmaceutical industry and patient groups also seem to actively welcome this improvement in the system. Yeon-hong Noh, Chairman of the Korea Pharmaceutical and Bio-Pharma Manufacturers Association, said, "This plan is an innovation that fundamentally changes the structure of the approval and review system beyond a simple race for speed. The industry will also cooperate so that the system can settle in the field by increasing the completeness of application materials." Ki-jong Ahn, representative of the Korea Alliance of Patient Organizations, also said, "I hope that an environment will be created where patients in urgent need of treatment can return to their daily lives as soon as possible."The MFDS began accepting applications for Pre-NDA Meetings for new drugs, biosimilars, and novel medical devices on June 1. The full 240-day review timeline will officially apply to applications submitted on or after October 1.
- Policy
- Novo Nordisk discontinues Victoza Pen supply in Korea
- by Lee, Tak-Sun Jun 02, 2026 08:55am
- Novo Nordisk Korea will discontinue the supply of its type 2 diabetes treatment, Victoza Pen 6 mg/mL (liraglutide), in the Korean market. The move follows the reimbursement listing of its successor product, Ozempic (semaglutide), in February and appears to be part of a broader portfolio optimization strategy aimed at replacing older-generation therapies with next-generation products.According to industry sources, Novo Nordisk Korea recently decided to discontinue Victoza Pen under its product portfolio integration and enhancement strategy and officially reported the decision to the Ministry of Food and Drug Safety on June 1.Victoza Pen, which was approved in Korea in 2010, is a first-generation GLP-1 receptor agonist for diabetes. Unlike Victoza, which required daily administration, Ozempic, which recently secured reimbursement coverage, demonstrated superior glycemic control and weight-loss benefits despite requiring only once weekly administration. Under pressure to make way for newer and more effective products, Novo Nordisk appears to be accelerating its portfolio transition in earnest with Ozempic’s inclusion in the national health insurance reimbursement.The company emphasized that extensive measures have been established to ensure that patient care is not disrupted by the discontinuation. A company official stated, “Supply will be discontinued as part of our portfolio integration efforts to provide stable treatment options to a greater number of patients. We will provide transition guidelines until supplies are completely cut off to ensure continuity of care for existing patients."The representative added, “We will carefully manage existing inventory until stocks are exhausted and maintain close communication with healthcare professionals to prevent any impact on patient treatment. Based on current inventory depletion forecasts, the likelihood of a sudden supply shortage or widespread stockout situation is low.”Novo Nordisk plans to recommend Ozempic Prefilled Pen as an alternative treatment option for patients currently receiving Victoza. The company noted, “Treatment transitions will be carried out appropriately based on healthcare professionals’ clinical judgment and each patient’s individual health status.”The medical community largely views the discontinuation as an expected development. One endocrinology professor at a university hospital commented, “The market is currently transitioning from once-daily liraglutide (Victoza) to once-weekly semaglutide (Ozempic), as it offers improved convenience and clinical utility. Since Ozempic entered the reimbursement system earlier this year, I believe the transition will be relatively smooth, as the patients’ financial burden will also be reduced.”
- Policy
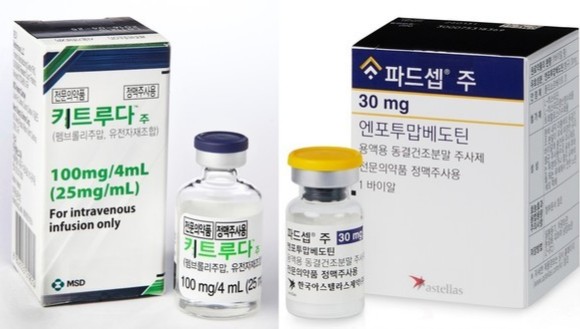
- Price negotiations begin for Padcev and Keytruda
- by Jung, Heung-Jun Jun 02, 2026 08:55am
- Astellas Korea’s Padcev (enfortumab vedotin) and MSD Korea’s Keytruda (pembrolizumab) have entered reimbursement price negotiations with the National Health Insurance Service (NHIS) for combination therapy in urothelial carcinoma.At the same time, Keytruda is also under separate negotiations for reimbursement expansion in gastric cancer, placing MSD in a pricing defense dilemma.According to industry sources on June 2, negotiations have begun for Padcev and Keytruda with the NHIS for urothelial carcinoma. In April, Padcev was recognized by the Drug Reimbursement Evaluation Committee (DREC) as being adequate as a first-line treatment for adult patients with locally advanced or metastatic urothelial carcinoma.However, the requirement to negotiate jointly with MSD represented a significant hurdle. Unlike Padcev, Keytruda reportedly joined the negotiations only after considerable deliberation, even after receiving a favorable DREC recommendation.The challenge stems from the fact that when a company participates in reimbursement negotiations for its drug in combination with a drug developed by another manufacturer, it may be forced to repeatedly defend a price that has already been reduced, or may soon be reduced, through prior reimbursement expansions.In January, Keytruda accepted a price reduction following reimbursement expansions across 11 indications, including endometrial cancer, breast cancer, and biliary tract cancer.Subsequently, the company has also pursued reimbursement expansion for Keytruda to dMMR/MSI-H metastatic gastric adenocarcinoma and gastroesophageal adenocarcinoma, following requests from the medical community. The indications passed DREC review in March and are currently under NHIS negotiation.As a result, MSD faces the difficult task of negotiating both the Padcev-Keytruda combination therapy for urothelial carcinoma and reimbursement expansion for gastric cancer during the same period.Industry observers view this as a clear example of the limitations inherent in negotiating reimbursement prices for multi-indication drugs, and stressed the need for an indication-based pricing system.The government is currently reviewing the validity and effectiveness of indication-based pricing. The NHIS plans to complete a research project by the end of this year and may consider implementation if deemed necessary. Factors under review include pricing equity across different cancer types and the administrative burden associated with drug management.Because indication-based pricing remains at the evaluation stage, the key question is whether a breakthrough can be achieved in these negotiations.A Korea MSD official commented, “Although it was not intentional, we ended up conducting the two negotiations simultaneously. We believe this case highlights the need for institutional improvements, such as indication-based pricing. However, since the negotiations have only just begun, further discussion will be necessary.”
- Policy
- "Phase 3 trial waiver·review shortcut"…for K-biosimilar
- by Lee, Tak-Sun Jun 01, 2026 09:04am
- AI-generated imageThe competitiveness of Korean biosimilars is projected to strengthen as the Ministry of Food and Drug Safety (MFDS) pursures regulatory reform that waive the requirement to submit therapeutic confirmatory clinical trial (Phase 3) data during the marketing authorization application process for biosimilars, provided that comparability in quality and non-clinical data is demonstrated.In addition to the 'Biosimilar Review Innovation Plan (reducing the review timeline to up to 240 days)' recently introduced by the MFDS to bolster global competitiveness, this measure is expected to create a powerful synergistic effect, significantly lowering the development burden on domestic biotech companies and accelerating their entry into global markets.A 'Two-Way Synergy' between Phase 3 clinical trial waivers and the 240-Day review In the biosimilar development pipeline, Phase 3 clinical trials have been the greatest hurdle, consuming the most time and requiring astronomical costs. Under the reform plan, developers can be exempted from Phase 3 trials if they successfully demonstrate comparability to the reference product in terms of quality, non-clinical data, and pharmacokinetic (PK) profiles.Notably, this is closely linked to the MFDS's recently announced initiative to reduce the biosimilar review timeline to up to 240 days. When promising biosimilar pipelines, already significantly truncated during the clinical trial stage, are placed on the MFDS's innovative and expedited review track, the overall commercialization cycle is projected to be advanced by several years compared to legacy timelines.Industry experts evaluate that companies will now have a springboard to secure 'first-mover' advantages or early price competitiveness in the global market, not only by reducing clinical development costs but also by minimizing administrative review bottlenecks.The benefits of shortened development timelines and accelerated review speeds will also translate directly to patients. As safe and effective biosimilars capable of substituting for high-cost original biologics are rapidly introduced to the market, the financial burden on patients is expected to ease significantly. It is expected to have a positive impact on safeguarding the fiscal soundness of the national health insurance fund.Through this reform, the MFDS has cleared a path to waive not only Phase 3 clinical trials but also the submission of non-clinical 'repeated-dose toxicity study data,' provided that analytical comparability in terms of quality and pharmacological profiles with the reference product is established. Consequently, companies will be able to gain momentum starting from the early candidate validation phase, substantially accelerating overall pipeline development efficiency.Notably, the public notice ensures that the revised regulations can be applied even to products whose marketing authorization or variation applications have already been submitted to the MFDS and are currently under review at the time of enforcement. The pharma-biotech industry is actively welcoming this 'surprise retroactive application' and the MFDS's consecutive forward-looking regulatory innovations, given that products awaiting approval could have immediate benefits.The MFDS plans to gather opinions from institutions, organizations, and individuals through an administrative notice period ending on June 19, after which the reform plan will be finalized and enacted. Attention is focused on whether the MFDS’s comprehensive deregulation spanning the entire life cycle of development (Phase 3 waiver) and review (240-day reduction) will activate a sluggish investment market and serve as a catalyst to further solidify South Korea’s status as a global biosimilar powerhouse.