- LOGIN
- MemberShip
- 2026-05-05 23:17:31
- Company
- Qalsody receives orphan drug designation in Korea
- by Eo, Yun-Ho Aug 08, 2024 09:24am
- ‘Qalsody (tofersen),’ a new drug for Lou Gehrig's disease, was designated as an orphan drug in Korea. The Ministry of Food and Drug Safety (MFDS) recently announced the designation through an orphan drug designation notice. More specifically, the drug is indicated for amyotrophic lateral sclerosis (Lou Gehrig's disease) associated with a mutation in the SOD1 (Superoxide Dismutase 1) gene. Qalsody, which was developed by Biogen, is an antisense oligonucleotide (ASO) drug that blocks the messenger ribonucleic acid (mRNA) associated with the SOD1 gene mutation to prevent its expression. The drug was approved by the U.S. FDA in June last year, followed by the European EMA in May. The efficacy data for Qalsody is not exactly glowing. However, this is likely a reflection of the scarcity of treatment options for the disease. In the Phase III VALOR trial, Qalsody failed to meet its primary endpoint, ‘Amyotrophic Lateral Sclerosis Functional Rating Scale (ALSFRS).’ However, it did reduce the secondary endpoints of ‘increased SOD1 protein levels in the cerebrospinal fluid’ and ‘neurofilament light chain (Nfl) concentrations’ by 26-38% and 48-67%, respectively. The most common adverse events reported in the trial were pain (back, arms, legs), fatigue, muscle and joint pain, fever, and increased protein and white blood cell counts in the cerebrospinal fluid. ALS is a rare neurological disease that affects nerve cells in the brain and spinal cord responsible for muscle movement and can lead to progressive paralysis and death. Although a considerable number of clinical trials are being conducted relative to the incidence rate, most medications only provide symptom relief.
- Opinion
- [Reporter's View] govt's foreign drug price comparisons
- by Kim, Jin-Gu Aug 08, 2024 09:24am
- The pharmaceutical industry's concerns grow over the government's plan to reevaluate foreign drug price comparisons. The government has established a policy to reduce domestic drug prices by comparing them to those in 'A8 countries (United States·Japan·Germany·France·Italy·Switzerland·Canada).' As the policy is set to be effective, the industry is busy estimating the scale of damage. Companies are estimated to experience a 5-20% reduction in annual sales. Therefore, the pharmaceutical industry voices concern that a sales reduction and an operational profit reduction will result in bigger damage. There are concerns that the scale of damage this time will be more substantial than the recent reintroduction of a stepped pricing system and reimbursement appropriateness reassessment. After considering the pharmaceutical industry's concern, the companies are not opposing to the reassessment soley due to the estimated damage. The industry has criticized that the criteria for comparing South Korean prices to those of foreign countries as unfair. For example, the industry considers referencing drug prices of Germany and Canada unfair. The government announced that when referencing drug prices in Germany and Canada, their public reimbursement prices, excluding outpatient payment, will be used. However, Korean drug prices include outpatient payments. In other words, the drug prices of countries for comparison will be lower, resulting in a greater degree of drug price reduction. For the past ten meetings since the end of last year, the pharmaceutical industry has strongly criticized the discrepancy in reassessing foreign drug price comparisons, including the issue mentioned earlier. However, sources said the government hesitated to take the pharmaceutical industry's opinion. Ten meetings between the government and the industry have concluded, and an agreement has yet to be reached. After the tenth meeting officially concluded, the Ministry of Health and Welfare (MOHW)'s Director of Pharmaceutical Benefits, who is in charge of the agenda, was replaced. In the second half of July, the MOHW held a regular personnel appointment and appointed a new Director of Pharmaceutical Benefits. Consequently, the person in charge of the final implementation of reassessing foreign drug price comparison has changed. The pharmaceutical industry has high hopes for these changes. They hope that the new Director will consider the industry's submitted opinions. At the last meeting, the industry had suggested several alternative measures. Opinions included improving how the government references drug prices in Germany and Canada, reducing drug price reductions amount by 50%, and putting a ceiling cap on the percentage of drug price reduction. It remains to be seen whether the new Director will consider these opinions. The prevalent opinion in the industry is that the government must completely reconsider the plan for reassessing foreign drug price comparisons. However, some are likely to follow the plan when unfairness is addressed. Now, the government is left to decide. The industry waits for appointment of the new Director.
- Policy
- Price-Volume Agreement negotiations to be finalized soon
- by Lee, Tak-Sun Aug 08, 2024 09:24am
- The ‘Type C’ price-volume agreement (PVA) negotiations are expected to be completed next week. When the negotiations are complete, the insurance price ceiling of the subject items is expected to be adjusted from September after passing the Health Insurance Policy Review Committee review this month. According to the industry sources on the 6th, negotiations for the PVA ‘Type C’ items started in July and are expected to be finalized next week. The negotiations involved 60 product groups that contain identical ingredients. The Type C negotiation is conducted for drugs that do not fall under 'Type A' or 'Type B', which are negotiated upon listing, and whose claims for the same product group from the 4th year of listing have increased by more than 60% from the claims of the previous year, or by more than 10% but exceeds KRW 50 billion. Most Type C drugs are generic drugs that have received a set price upon listing. The National Health Insurance Service conducts Type C negotiations every year with the goal of adjusting the insurance price ceiling of subject drugs by September of the same year. During this year's negotiations, in particular, the pharmaceutical companies enjoyed increased options due to the amendment made to the details of the operating standards. For example, drugs whose usage has temporarily increased due to inevitable reasons such as the COVID-19 pandemic can choose to refund part of their claims instead of lowering the price. In the first round of negotiations that were given the option, several companies reportedly opted for the refund option. In addition, innovative pharmaceutical companies or companies with an R&D ratio of 10% or more, which have been subject to PVA negotiations 3 times within 5 years, will be eligible for a 30% less price reduction than the amount calculated via the reference formula rate in its third time. Although small, there are reportedly a small number of companies that have agreed to negotiate their price using this option. The pharmaceutical industry had previously expressed concerns to the government on how drugs that have completed bioequivalence tests for the price ceiling reevaluations may be subject to larger price cuts during PVA negotiations. The logic is that the price difference between a drug that has maintained its price through bioequivalence testing and the weighted average price of the same-ingredient drug becomes larger than the weighted average price of drugs with the same ingredient, resulting in a larger price reduction during PVA negotiations. In response, the pharmaceutical industry requested price adjustments for drugs that underwent bioequivalence tests for a prior reevaluation, considering how the companies have followed government policy. In response, the NHIS is said to have partially accepted the industry's opinion and reflected it qualitatively during negotiations. As a result, it is observed that all the negotiations will be completed within the deadline this year. "We understand that the negotiations will be completed by next week," said an industry official, "and the results will be announced after being reported to the HIPDC this month."
- Company
- Vaxneuvance increases presence in NIP mkt
- by Hwang, Byung-woo Aug 08, 2024 09:24am
- MSD Korea is accelerating its efforts to capture the national immunization program (NIP) market by touting the high immunogenicity of Vaxneuvance. The vaccine has already been rapidly introduced to general hospitals and clinics upon its launch, and the company is highlighting the vaccine’s clinical benefits to gain a competitive advantage. Dr. Hyun-mi Kang, Professor of Pediatrics, St. Mary Vaxneuvance, which was approved late last year, is the first new pneumococcal vaccine introduced to Korea in 13 years. It is a 15-valent vaccine that added 2 serotypes -22F and 33F – to the existing 13-valent vaccine. In particular, the vaccine made the pediatric NIP list at an unprecedented speed, just one month after its approval. Since its launch in April, Vaxneuvance has been available for NIP vaccination in children aged 2 months to 5 years. Previously, Pfizer's Prevenar 13 was the only available NIP vaccine for pediatric pneumococcal disease. Now, MSD is expanding Vaxneuvance’s presence in the market by emphasizing that children who have received one or more doses of Prevenar 13 can also cross-vaccinate the remaining doses with Vaxneuvance. At the Vaxneuvance media seminar that was held on June 6, experts said that the introduction of Vaxneuvance has changed clinical practice. "We don't know the overall vaccination rate because we don't have specific data, in my hospital, Vaxneuvance is being administered first due to its broader coverage since April," said Hyun-mi Kang, professor of Pediatrics at St. Mary's Hospital in Seoul. Jaeyong Cho, Executive Director of MSD Korea's Vaccine Business Unit, added, "Although the data is unofficial, there has been an increase in Vaxneuvance’s use as third and fourth doses along with the initial dose since its launch. Overall, the rate of those receiving an initial dose of Vaxneuvance is higher, but the proportion of cross-vaccinations is also continuing to grow." Vaxneuvance’s strength is in the high immunogenicity of all serotypes Immunogenicity was also highlighted as the competitive advantage of Vaxneuvance during the media seminar. This means that there is a need to choose a highly immunogenic option to prevent invasive pneumococcal disease (IPD), which is highly fatal in children. The WHO's definition of immunogenicity is "the ability of a vaccine to induce a measurable immune response. The specific serotype-specific immunogenicity criteria are 'IgG concentration of 0.35 μg/mL or higher'. "In a Phase III study in healthy Korean infants and young children, Vaxneuvance demonstrated a primary immunogenicity endpoint of IgG of 0.35 μg/mL or higher across 15 serotypes after the 3rd dose in more than 95% of subjects," said Professor Kang. While Vaxneuvance is rapidly penetrating the market based on its clinical performance, there is hesitation in the field as Prevenar 20, which contains more serotypes, will be launched in the second half of the year. In this regard, Professor Kang emphasized that it is also important to consider the prevention effect of each serogroup in addition to the broader spectrum of protection. "We have been saying that preventing more is important for invasive pneumococcal disease due to its many serotypes, but the more number of serotypes does not necessarily mean that (the vaccine) is good. I think we need to consider the safety of the vaccine along with the immunogenicity for each serotype."
- Company
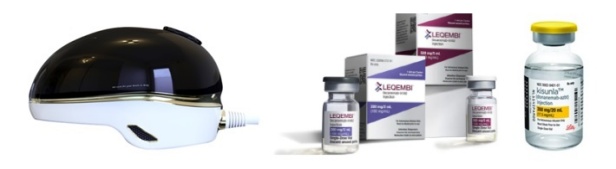
- Electronic drugs show potential to conquering Alzheimer's
- by Son, Hyung-Min Aug 08, 2024 09:24am
- The Korean pharmaceutical bio industry is making a bid into the electronic drug market for Alzheimer's disease. Recently, Remed unveiled the results of its transcranial magnetic stimulation (TMS) therapy that demonstrated an effect in treating Alzheimer's disease. AriBio, which is developing a new drug for Alzheimer's disease, is developing an electronic drug that uses vibroacoustic stimulation On July 7, Remed disclosed clinical results showing that an electronic drug being developed by Remed was effective in patients with Alzheimer's disease. Remed is a Korean biotech company that has been identifying the potential of electronic drugs in various areas including depression, attention deficit hyperactivity disorder (ADHD), and dementia. The electronic drug being developed by Remed for Alzheimer's disease uses transcranial magnetic stimulation. Transcranial magnetic stimulation is a brain stimulation procedure that uses magnetic fields to stabilize or activate brain nerve cells in a specific area at the base of the skull. It has been used to treat various brain disorders such as depression, intractable obsessive-compulsive disorder (OCD), and chronic pain. Remed’s subsidiary NextCure’s helmet-type wearable electronic therapyRemed has confirmed a change in the Alzheimer's Disease Assessment Scale-Cognitive Subscale (ADAS-Cog), an important indicator in the diagnosis of dementia, with its electronic drug in clinical development. The clinical trial was conducted from May 2020 to April 2022 to evaluate the efficacy and safety of the electronic drug in 30 patients with Alzheimer's disease. Trial results showed that the ADAS-Cog value of patients treated with the electronic drug improved significantly compared to those who did not receive treatment. More specifically, the change in ADAS-Cog values was above the ADAS-Cog MCID threshold of 4.0, which is regarded as a clinically significant change in dementia medications, and showed no serious adverse events. "These results suggest that transcranial magnetic stimulation may be effective in Alzheimer's patients in addition to depression, intractable OCD, chronic pain, and stroke," said the researchers involved in the trial. Remed plans to complete the exploratory trial and submit a confirmatory IND to the Ministry of Food and Drug Safety. AT&C is developing a treatment system that combines an electronic drug that uses transcranial magnetic stimulation and digital therapeutics (DTx) for the treatment of mild Alzheimer's disease. The company is currently conducting a confirmatory clinical trial with 158 patients at Korea University Anam Hospital, Dong-A University Hospital, Chonnam National University Hospital, Jeonbuk National University Hospital, Chung-Ang University Gwangmyeong Hospital, Chungnam National University Hospital, and Hanyang University Hospital. According to the company, the combination of transcranial magnetic stimulation and digital therapeutics showed better and longer treatment effects than conventional medication and cognitive training, which showed low treatment effects and short treatment duration. In addition to Remed and AT&C, another company, AriBio, is developing electronic drugs for Alzheimer's disease. In May, Aribio received approval from the Ministry of Food and Drug Safety to conduct a clinical trial for an electronic drug that uses vibroacoustic stimulation on Alzheimer's disease patients. The company’s brain vibroacoustic stimulation electronic drug offers a new approach to treating Alzheimer's disease. The company explained how their trial is the first clinical trial of an electroacoustic brain stimulation device that uses vibroacoustic stimulation, although there have been devices that use electric, electromagnetic field, and ultrasonic methods in the past. The clinical trial will be conducted by Professor Sang-Yoon Kim’s team from the Seoul National University Bundang Hospital, to confirm the efficacy and safety of brain vibroacoustic stimulation. The efficacy of the electronic drug will be evaluated in a double-blind, 6-month trial in 30 patients with early-stage Alzheimer's disease, including those with mild cognitive impairment. If the exploratory clinical trial confirms the efficacy of the electronic drug, AriBio aims to launch the electronic drug for dementia by 2026 after conducting a confirmatory clinical trial. In addition to the electronic drug, AbiBio is also conducting a Phase III clinical trial on AR1001, a novel drug candidate for Alzheimer's disease. AR1001 targets multiple causes of Alzheimer's disease, including the PDE5 protein. Recently, studies have shown that phosphodiesterase 5 (PDE5) inhibitors, such as Viagra and Cialis, are effective in preventing Alzheimer's disease, raising interest in AR1001's commercialization potential. The introduction of electronic drugs in the field...will Alzheimer's disease finally be conquered? Eisai and Biogen’s Alzheimer’s treatmentAlzheimer's disease has been a challenging area for drug development. After the hypothesis that Alzheimer's disease is caused by amyloid beta emerged, targeted drugs were developed, and then were found ineffective. One such drug was Eisai’s Aduhelm (aducanumab). The amyloid beta hypothesis was highly suspect, so there was not much confidence in the drug, but the drug worked. However, Aduhelm was withdrawn from the market due to its high price and concerns about side effects. Then came Leqembi (lecanemab), developed by Eisai and Biogen. Leqembi showed efficacy in early-stage Alzheimer's disease in clinical trials and has cleared regulatory hurdles in South Korea, the U.S., Japan, and China. Lilly’s Alzheimer’s treatmentMore recently, Lilly's Kisunla (donanemab) has also been approved by regulatory authorities. Kisunla was shown to delay cognitive deterioration in patients with early Alzheimer's disease in the Phase III TRAILBLAZER-ALZ2 study. In the study, donanemab delayed cognitive deterioration regardless of disease progression or pathologic stage. However, there is currently no cure for Alzheimer's disease. Although Leqembi and Kisunla have emerged, experts believe that they are only effective in patients with early-stage Alzheimer's disease. This is why the pharmaceutical industry continues to tackle Alzheimer's disease with a variety of possibilities, including not only new drugs but also electronic drugs and digital therapeutics. To date, clinical trials using electronic drugs have been limited to exploratory trials, but if they show effectiveness in confirmatory trials, they may also show synergy with the new drugs on the market.
- Company
- Tepezza receives orphan drug designation in Korea
- by Eo, Yun-Ho Aug 06, 2024 09:16am
- Tepezza (teprotumumab), a targeted treatment for thyroid eye disease (TED), has been designated as an orphan drug in Korea. The Ministry of Food and Drug Safety (MFDS) recently announced the designation through an orphan drug designation notification. Specifically, Tepezza is indicated for the treatment of adult patients with moderate-to-severe thyroid ophthalmopathy. Tepezza is an insulin-like growth factor-1 receptor inhibitor class monoclonal antibody that is administered intravenously every 3 weeks (8 injections in total). The drug was first approved by the U.S. FDA after receiving a fast-track designation in January 2020 but has not yet been commercialized in Korea. The company recently submitted applications for the drug’s approval in Europe and Japan. The drug was originally developed by Irish pharmaceutical company Horizon Therapeutics, and Amgen acquired the rights to the drug when it acquired Horizon in 2022. In the Tepezza trials, patients with TED were administered 8 infusions of Tepezza or placebo every 3 weeks. The Phase II and OPTIC studies included a total of 121 patients with TED with a Clinical Activity Score (CAS) of 4 or greater and TED onset of 9 months or less. Among the 99 patients aged 40 years or older, the mean age was 54.5 years. 72.7% were women and double vision was confirmed in 74.7% of the patients. Among the 22 patients under the age of 40, the mean age of 32.8 years, 59.1% were female, and 68.2% had double vision. When analyzing the response to Tepezza in patients with acute TED, the proportion of patients who responded to treatment with at least a 2mm improvement in ocular protrusion at week 24 was 86.4% in the under-40 group and 79.8% in the over-40 group, showing no statistically significant difference between the two groups. Tepezza has been recognized for changing the treatment landscape for TED. Prior to Tepezza, the only treatment options available for TED were steroids and orbital decompression. Although steroids reduce inflammation, they are associated with side effects and have a risk of recurrence upon discontinuation, and orbital decompression is associated with surgical complications.
- Company
- Lily Korea appoints John Bickel as new General Manager
- by Hwang, Byung-woo Aug 06, 2024 09:16am
- John Bickel, new General Manager of Eli Lilly KoreaEli Lilly Korea announced on August 5th the appointment of John Bickel as the new General Manager of Eli Lilly Korea, effective August 1st. Bickel is an expert with 26 years of extensive leadership experience at Eli Lilly and Company. In the headquarters, he was responsible for the U.S. and global market in oncology and neuroscience divisions, overseeing various pharmaceuticals. Bickel served as the Chief Marketing Officer (CMO) in the Japanese subsidiary of Eli Lilly and Company from 2019. He also served as the Chief Operating Manager (COO) from January 2023, managing Japan's entire marketing and sales organization. Bickel is known to have contributed to strengthening Japanese subsidiary's business capacity, introducing a digital and omnichannel marketing approach, and utilizing data analysis tools to make key decisions. Furthermore, Bickel played a vital role in talent recruitment and training to carry out innovative marketing strategies. "I am pleased to be appointed as the new leader of Eli Lilly Korea and acknowledge the responsibility. In Korea, innovative pharmaceuticals are gaining attention in the fields of diabetes, obesity, cancer, and immune disease, which Eli Lilly has focused on," Bickel said. "As the new GM of Eli Lilly Korea, I will put efforts into quickly supplying Lilly's new pharmaceuticals to South Korea so that Korean patients no longer suffer from life-threatening diseases that lower quality of life and have better daily lives. Furthermore, I will strive to be a trusted partner in contributing to domestic healthcare and the development of pharmaceutical industry," Bickel said. Meanwhile, Bickel graduated with a Bachelor of Pharmacy from Butler University in 1988 and joined Eli Lilly the same year.
- Company
- Trials start for 'cancer vaccine+immunooncology drug’ combo
- by Son, Hyung-Min Aug 06, 2024 09:16am
- Global pharmaceutical companies are making progress in developing messenger ribonucleic acid (mRNA) cancer vaccines and immuno-oncology combinations. Recently, Regeneron and BioNTech confirmed efficacy in a Phase II clinical trial for melanoma. Moderna, a company specializing in the development of mRNA vaccines, has also shown promise in melanoma and head and neck cancer using its vaccine in combination with the immuno-oncology drug Keytruda. In Korea, LG Chem, ImmuneOncia, and others are developing new mRNA and immuno-oncology drug combinations. A cancer vaccine activates the immune system by administering cancer cell antigens to patients, rather than being a preventive concept. Like a vaccine, it has a mechanism that activates the immune response and causes cancer cells to kill themselves. Major pharmaceutical and biotech companies are combining immuno-oncology drugs and antibody-drug conjugates (ADCs) focusing on maximizing the treatment effect rather than cancer prevention to increase their commercialization potential. #i According to industry sources on the 15th, a cancer vaccine and immuno-oncology drug combination using the cancer vaccine developed by the German BioNTech had demonstrated efficacy in a Phase II study in melanoma patients. The trial evaluated the clinical activity of BioNTech's mRNA vaccine BNT111 in combination with Regeneron's PD-1 immuno-oncology drug Libtayo (cemiplimab). BNT111 is a cancer vaccine that enhances T-cell responses to 4 antigens (NY-ESO-1, MAGE-A3, tyrosinase, and TPTE) expressed in melanoma patients. In clinical trials, the BNT111 and Libtayo combination demonstrated a statistically significant improvement in overall response rate (ORR). The specifics will be presented at an upcoming international conference. BioNTech is also currently conducting a Phase II clinical trial comparing the efficacy and safety of another cancer vaccine candidate, Autogene cevumeran, with Roche's immuno-oncology drug Tecentriq (atezolizumab). Autogene cevumeran is a therapeutic personalized cancer vaccine candidate that screens and encodes up to 20 patient-specific cancer mutations that can act as neoantigens. The trial evaluated the efficacy of the Autogene cevumeran + Tecentriq combination with the FOLFIRINOX regimen ((luorouracil+leucovorin+irinotecan+oxaliplatin). In addition to melanoma, Moderna is also exploring the potential of combining its cancer vaccine candidate mRNA-4157 with MSD's immuno-oncology drug Keytruda for patients with head and neck cancer. The company recently announced clinical results from a study on patients with head and neck cancer The trial evaluated the efficacy and safety of Moderna’s candidate mRNA-4157 in combination with MSD's immuno-oncology drug Keytruda (pembrolizumab) in 22 patients with human papillomavirus (HPV)-negative head and neck squamous cell carcinoma. In the trial, the objective response rate (ORR) for the mRNA-4157 plus Keytruda combination was 27%, compared with the 18% achieved in the Keytruda monotherapy arm. Overall survival (OS) for the combination was 24.6 months, which was longer than the 9.8 months achieved in the Keytruda monotherapy arm. Moderna is currently conducting a Phase III clinical trial for the mRNA-4157 plus Keytruda combination in melanoma. In an ongoing Phase II trial in melanoma patients, mRNA-4157 plus Keytruda was shown to reduce the risk of recurrence or death by 49% compared to Keytruda monotherapy. Domestic companies such as Yuhan Corp and ImmuneOncia start developing mRNA+ immuno-oncology drugs# The domestic pharmaceutical bio-industry is also confirming the potential of the mRNA and immuno-oncology combination therapy. Yuhan Corp is developing new mRNA and LNP (lipid nanoparticles) source technologies through industry-academia collaboration. Yuhan Corp is developing mRNA and LNP (lipid nanoparticle) source technologies with the research teams of Professor Hyukjin Lee from Ewha Womans University and Professor Ju Youp Lee from the University of Cincinnati. The company plans to develop a new type of mRNA structure technology that can increase the amount of protein expression while overcoming the easily degradable stability limitations of mRNA. ImmuneOncia is conducting joint research on mRNA-based next-generation immuno-oncology drugs with mRNA therapeutics developer NuclixBio. The two companies will develop next-generation therapeutics using ImmuneOncia’s immuno-oncology drug IMC-001 and NucleixBio's mRNA-based antibody platform 'ringRNA'. The principle is that when mRNA expressing the same amino acid sequence as ImmuneOncia’s antibody is produced and administered, antibodies are produced in the body, providing an effect. Earlier this year, GI Innovation signed a business agreement with ProGen and SL Vaxigen to evaluate the potential of the immuno-oncology drug 'GI-101A' in combination with a cancer vaccine. The three companies will initiate a Phase II clinical trial of GI-101A and DNA vaccine therapy PG-101 in prostate cancer patients. The trial will be led by Prof. Jae-Lyun Lee of the Department of Medical Oncology at Asan Medical Center in Seoul and has received investigator-initiated clinical approval from the Korean Cancer Study Group (KCSG). GI-101A has a mechanism of action that acts on CD80 and interleukin (IL)-2. IL-2 is involved in immune cell proliferation and activation, and CD80 blocks CTLA4, a receptor that inhibits immune cells from attacking cancer cells.
- Company
- Entresto sales soar… rises 22% in 1 year
- by Kim, Jin-Gu Aug 06, 2024 09:15am
- Pic of Entresto Sales of Novartis' heart failure drug 'Entresto' continues to grow over 20% YoY in its 7th year on the market. At this rate, the drug is expected to generate prescription sales of around KRW 70 billion by the end of this year. The variable is the patent dispute. The company is currently awaiting its second judgment on its patent dispute with generic companies. The patent court’s ruling is expected to serve as a turning point for the early release of generics. Entresto in its 7th year of release, continues to grow 22% YoY According to the drug market research firm UBIST on the 5th, Entresto posted prescription sales of KRW 17.1 billion in Q2. This is a 22% YoY increase from the KRW 14 billion it had posted in Q2 last year. Entresto is a first-in-class angiotensin receptor-neprilysin inhibitor (ARNI) that combines the angiotensin receptor blocker (ARB) valsartan and neprilysin inhibitor sacubitril. It was granted reimbursement and released in 2017. Quarterly prescriptions of Entresto (Unit KRW 100million, Source: UBIST)The drug is still showing strong growth in its 7th year of release. Since its launch, its drug price has been reduced by 21%, through 7 price cuts until earlier this year through the price-volume agreement and voluntary reductions, but it did not have a significant impact on the upward sales trend. This is likely due to the fact that the drug's reimbursement coverage has been expanded at the same time as the price cuts. When Entresto was first added to the reimbursement list in 2017, it was reimbursed as a treatment for patients with chronic heart failure with reduced ejection fraction (HFrEF). Coverage was limited to patients previously receiving an ACE inhibitor or ARB inhibitor in combination with standard therapy at a stable dose for at least four weeks. In March 2022, the reimbursement standard was extended to first-line therapy and became available for patients who were previously untreated with an ACE inhibitor or an ARB inhibitor. In July of the following year, the drug became available for prescription in both inpatient and outpatient settings. The drug's longtime dominance of the heart failure treatment market has also contributed to its steady rise in sales. Entresto was the only prescribed treatment targeting heart failure for 6 years until Bayer launched Verquvo (vericiguat) in September last year. Will its sales exceed KRW 70 billion by the end of the year?...the 2nd ruling of the patent dispute remains a variable The industry expects Entresto to generate prescription sales of around KRW 70 billion by the end of the year. Entresto’s sales have grown rapidly since exceeding the KRW 10 billion mark in annual prescription sales in 2019. The drug posted KRW 57.5 billion last year. In the first half of this year, it generated KRW 32.9 billion. If sales continue to grow at this pace, sales could reach KRW 70 billion in annual prescription sales. The variable is in the patent dispute. Domestic pharmaceutical companies have filed patent appeals against several of Entresto's patents at the same time. Since January 2021, generic companies have filed a challenge on Entresto's crystalline form patent, salt and hydrate patent, 2 use patents, and 2 formulation patents. The generic companies won the first trial. After losing the first trial, Novartis appealed 3 cases: the crystalline form patent, the use patent, and the salt and hydrate patent. Of these, the patent court rejected the use patent case in November last year. Novartis appealed again to the Supreme Court, but the court ordered the discontinuance of the trial in April this year. Entresto Patent Dispute Status (Source:KIPO) As a result, 2 patent disputes now remain. The arguments for the salt-hydrate patent have been finalized. The crystalline patent is also reportedly in the final stages of defense. The industry expects the patent court to make a ruling as early as this year. The decision of the second trial is expected to serve as a turning point for early generic launches. Even if Novartis loses the second trial, the company will likely appeal to the Supreme Court, but if generic companies win both the first and the second trial, it will raise the probability of the early launch of Entresto generics.
- Company
- Nabota records KRW 92B in North American sales in Q2
- by Son, Hyung-Min Aug 05, 2024 07:51am
- Evolus has posted record quarterly sales thanks to a surge in sales of its botulinum toxin Nabota. The company generated USD 126 million in sales in the first half of the year, lighting a green light to achieving its performance target for the year. Evolus plans to continue to grow sales by expanding Nabota's international approvals and launching new filler products to market. According to industry sources on the 2nd, Evolus' revenue in Q2 this year was USD 66.9 million (KRW 92 billion), up 35.6% from the USD 49.35 million it had made in the same period a year ago. Evolus Most of Evolus' revenue came from sales of Nabota (U.S. brand name: Jeuveau). Daewoong Pharmaceutical signed a supply agreement with Evolus in 2013 worth USD 296.8 million (approximately KRW 375 billion). Evolus then introduced Nabota to the North American market in 2020. Nabota was first approved in the North American market. Nabota was initially approved in Canada in 2018, then in the U.S. in February 2019. Since then, Nabota has been further approved in Europe, Australia, Russia, South Africa, and Japan. Evolus' revenue has been increasing steadily since Nabota’s launch in North America in 2020. In Q1 2021, when Nabota was launched, Evolus posted revenue of USD 13.5 million, but the amount increased 151.9% to KRW 34 million in Q1 2022. Evolus surpassed USD 50 million in quarterly revenue for the first time in Q3 last year. Evolus' revenue continued to grow this year as well. In Q1 of this year, the company generated USD59.3 million in revenue, and Q2 revenue was KRW 66.9 million, breaking its record every quarter. Evolus is also looking to expand into the European market upon the official launch of Nabota in Spain in June. In Europe, Nabota is currently available under the brand name Nuceiva in Spain, the United Kingdom, Germany, Austria, and Italy. Botulinum toxin Nabota(U.S. brand name:Jeuveau)Evolus added 770 new Nabota accounts in Q2. The company has increased the number of customers who have purchased Nabota since its launch to 14,000. The reorder rate of Nabota users is 70%. The growth of Nabota puts Evolus on track to meet the revenue goals it set last year. Evolus had set a revenue target of USD 265 million (KRW 350 billion) this year. In the first half of the year alone, the company achieved about half of its annual target, generating USD 126 million. In addition to Nabota, Evolus is looking to expand sales through new filler products. In May last year, the company secured exclusive U.S. sales and distribution rights to a filler developed by France's Symatese. Evolus recently submitted a premarket approval application (PMA) to the U.S. Food and Drug Administration (FDA) for 2 filler products, Evolysse and Evolysse Lift. The company expects the two to be approved in Q2 next year