- LOGIN
- MemberShip
- 2026-05-11 11:24:35
- Company
- Chong Kun Dang has the domestic license for the 110 billion
- by Chon, Seung-Hyun May 18, 2023 05:44am
- Chong Kun Dang bought the domestic license for MSD’s blockbuster diabetes treatment ‘Januvia series’. It is equipped with a stable cash cow that raises more than 100 billion won a year. Chong Kun Dang signed a license agreement with MSD headquarters in Switzerland to introduce all domestic rights for three diabetes treatments, Januvia, Janumet, and Janumet XR. Chong Kun Dang acquires not only domestic sales and distribution rights for the three Januvia series, but also all rights such as licenses, trademarks, and manufacturing. The contract period is from July 15th to August 31st, 2038. The total contract amount is 45.5 billion won. Chong Kun Dang paid MSD headquarters a down payment of 23 billion won, and the milestone scale according to sales is 17 million dollars (approximately 22.5 billion won). But Since 2016, Chong Kun Dang has jointly sold the Januvia series with MSD Korea. Through this contract, it exclusively secured domestic rights for the Januvia series for the next 15 years. Chong Kun Dang receives the Januvia series from MSD headquarters and sells them exclusively in Korea. Januvia is a DPP-4 inhibitory antidiabetic drug containing Sitagliptin. Janumet is a combination drug combining Januvia and metformin. According to UBIST, a pharmaceutical research institute, the Januvia series jointly invested a total of 109.4 billion won in outpatient prescriptions last year. Januvia and Janumet raised 40.5 billion won and 68.9 billion won, respectively. Outpatient prescription amount of Januvia series by year (unit: KRW 100 million, source: UBIST) The amount of prescriptions for the Januvia series last year decreased from 130.7 billion won in 2020 and 124.6 billion won in 2021, which is the aftermath of drug price cuts. In March of last year, through a 'trade-off' agreement with the government, MSD lowered the insurance cap for the Januvia series by an average of 6.0%. The drug price of the Januvia series was voluntarily lowered as a condition of expanding reimbursement for Keytruda, an immuno-oncology drug. Considering the drug price reduction rate of the Januvia series, it means that it still has a great influence in the market. As Chong Kun Dang has secured all licenses for the Januvia series, profitability from future sales is expected to increase. An official from Chong Kun Dang said, “We were leading the market with a diverse portfolio of diabetes treatments, including Duvie,” and “We stably expanded the treatment options for patients by securing the Januvia series.”
- Company
- Benefit extended DM Drug
- by Moon, sung-ho May 17, 2023 05:38am
- Clinical sites are busy finding the optimal prescription combination while the expansion of diabetes treatment reimbursement standards for each class and the release of generics following the patent expiration of original items coincided. It is an effort to find the optimal combination for each treatment category that can be covered by health insurance to minimize patient burden. According to the pharmaceutical industry on the 26th, the Ministry of Health and Welfare significantly eased the criteria for the accreditation of diabetes medications this month. The key is that various drug combinations are possible without specifying the SGLT-2 inhibitor component. With this revision, SGLT-2 inhibitors such as Ipragliflozin, Empagliflozin, and Ertugliflozin can be reimbursed when used together. Combinations of Metformin + SGLT-2 inhibitor + DPP-4 inhibitor and Metformin + SGLT-2 inhibitor + TDZ combination are also acceptable if the HbA1C is 7% or higher even if the two-drug regimen is administered for more than 2 to 4 months. Insurance cannot be provided if only SGLT-2 inhibitor + DPP-4 inhibitor or TZD is used without Metformin. In the clinical field, following the release of Forxiga's generic products in April, guidance is being given to prescribing DPP-4 inhibitors or TZD with reimbursement instead of prescribing inexpensive SGLT-2 inhibitors through the full cost of the patient's expenses. This is because the 2nd union is excluded from the benefit target. A professor of endocrinology at A University Hospital, an executive officer of the Korean Diabetes Association, said, "Three-drug therapy was applied as reimbursement, but SGLT-2 inhibitor + DPP-4 inhibitor or TZD two-drug therapy is not reimbursed, so it can be a prescription form." It is still in the early stages of expanding the salary standard, so there are various opinions coming and going.” In addition, there is an opinion that there may be a situation where there is no choice but to recommend the patient to take without 'metformin' instead of prescribing the three-drug therapy as the two-drug regimen is not covered. In the case of patients who cannot take metformin due to side effects, SGLT-2 inhibitor + DPP-4 inhibitor or TZD two-drug therapy is a prescription pattern that can occur in clinical settings because reimbursement is not possible. As the two-drug regimen is not possible, instead of prescribing SGLT-2 inhibitors as non-covered drugs, it is possible to recommend taking metformin out of the three-drug regimen that can be covered. In the pharmaceutical industry, it is a phenomenon that can occur in clinical settings, but it is evaluated that it will be only a small part. An executive of a domestic company A, who used to be a doctor, said, “It is a prescription pattern that can happen at the moment, but it is a concern raised because of changes in the clinical field due to the expansion of the reimbursement standard have not yet taken place.” The Ministry of Health and Welfare plans to list SGLT-2 inhibitors and DPP-4 inhibitor complexes of major pharmaceutical companies in May, following the expansion of the reimbursement standard and the Forshiga generic this month. Specifically, ▲AstraZeneca Qtern, ▲Boehringer Ingelheim's Esgliteo, ▲MSD Stegluzan, and ▲LG Chem's Zemidapa are included. In addition, Daewoong Pharmaceutical's Envlo, a new diabetes drug in the domestic SGLT-2 inhibitor class, will enter the prescription market in earnest with reimbursement.
- Company
- SK Bioscience & MSD signed a consignment production contract
- by Jung, Sae-Im May 17, 2023 05:38am
- At the contract signing ceremony held in Jongno-gu, Seoul, government officials such as Second Vice Minister of Health and Welfare Park Min-soo, MSD Vice President Sanat Chattopadhyay, Hilleman Research Center CEO Raman Rao, SK Discovery Vice Chairman Choi Chang-won, SK Bioscience President Ahn Jae-yong, Hoon Kim, CEO of Global R&BD, etc. attended. MSD is developing a next-generation Zaire Ebola vaccine candidate with improved process efficiency and stability of the currently approved and used Zaire Ebola vaccine Ervebo with the Hillemann Institute, an international non-profit research institute. In the future, if the candidate substance is successfully developed and approved by regulatory authorities, it is expected to contribute to increasing the global supply and improving accessibility of the Zaire Ebola virus vaccine. The candidate material will be produced at Andong L House after SK Bioscience has transferred related development and technology, and will be supplied to international organizations after obtaining approval from relevant health authorities to be used in the management of Ebola virus disease. Ebola virus disease is a serious hemorrhagic fever disease caused by infection with the Ebola virus. The main cause of outbreaks in the past 20 years has been the Zaire Ebola virus. Since the Ebola virus was first discovered in 1976, several outbreaks have caused serious human and economic damage. Starting with this contract, SK Bioscience plans to expand its CMO and CDMO business in earnest. Based on R&D technology proven with various self-developed vaccines and state-of-the-art vaccine production facilities, it is a strategy to expand the C(D)MO business for various infectious diseases to respond quickly to new pandemics and take the lead in promoting public health. In addition to the existing vaccine platform, C(D)MO business for new platforms such as mRNA and CGT will also be promoted. A pilot plant that will enhance competitiveness in the C(D)MO market will be built in the ‘Global R&PD Center’ established in Songdo, Incheon through the largest facility investment by SK Bioscience after its launch. The pilot plant, which is a small-scale test facility built before introducing new methods or products, will be equipped with facilities that can carry out research tasks such as CGT, mRNA, and viral vectors. Vice Chairman Choi Chang-won of SK Discovery said, "This collaboration is the result of SK bioscience's production capacity and global network recognized through COVID-19, and it will be an important milestone in our efforts to contribute to the promotion of human health." We hope that the cooperation between SK, MSD, and Hilleman Laboratories will be further expanded with the government, such as the Ministry of Health and Welfare and the Korea Centers for Disease Control and Prevention, based on the common belief that it will solve the problem of imbalance in the country's vaccine supply and expand access to vaccines."
- Company
- Duloxetine shows strength in ₩30B antidepressant mkt
- by Nho, Byung Chul May 17, 2023 05:38am
- Last year, top-line sales of the antidepressant duloxetine recorded KRW 17.7 billion, taking a big lead ahead of venlafaxine, which achieved KRW 12.7 billion in sales, and is solidifying its position in the relevant prescription market. According to drug distribution data, sales of duloxetine drugs grew from KRW 15.5 billion to KRW 17.7 billion from 2019 to 2022, whereas sales of venlafaxine drugs showed a declining trend from KRW 13.1 billion to KRW 12.7 billion. The contrasting performance of the top two major antidepressant substances in the market is interpreted to be attributed to the difference in their indications. The duloxetine original, Lilly’s Korea’s Cymbalta Cap can be prescribed in a broader scope as its indications include the treatment of generalized anxiety disorder, diabetic peripheral neuropathic pain, fibromyalgia, and musculoskeletal pain that does not respond appropriately to non-steroidal anti-inflammatory drugs (NSAIDs). On the other hand, the venlafaxine leader, the indication of Viatris Korea’s Efexor Xr Cap. Is limited to depression, such as social phobia and panic disorder, which fall under the category of generalized anxiety disorder. The unwavering lead in the duloxetine antidepressant market is Cymbalta, which has raised sales of KRW 8.6 billion in 2018, KRW 9.1 billion in 2019, KRW 9.3 billion in 2020, KRW 9.7 billion in 2021, then KRW 9.7 billion in 2022. The second duloxetine in line is Hanlim Pharm’s Duxela Cap., which raised sales of KRW 1.7 billion last year. Myung-In Pharm’s Droctin Cap ranked third has shown aa over twofold growth from KRW 0.74 billion in 2018 to KRW 1.35 billion last year. Top-line sales of the duloxetine drugs that followed are HR Inno. N’s Culocta, ‘Hwan-In Pharm’s Duloxeptol, Korea Celltrion Pharm’s Julotine, then Korea Pharm’s Duroprex. The respective drugs raised sales of KRW 0.85 billion, 0.77 billion, 0.53 billion, and 0.38 billion, respectively. The combined sales of the 18 other minor products in 2022 were in the KRW 1.4 billion range. The lead product among venlafaxine drugs is Efexor XR, which recorded KRW 4.4 billion last year. The rapid growth of generic versions of venlafaxine is noteworthy in the market. Korea Pharma’s Pharma Venlafaxine arose to become the second-most sold the drug in the venlafaxine market, making a 1800% growth from KRW 0.15 billion in 2018 to KRW 2.89 billion in 2022. The third was Hanlim Pharm’s Venexor XR, which recorded sales of KRW 2.1 billion last year. Sales of Myung-In Pharm’s Cofexor, which had been in the KRW 3 billion range until 2019, were reduced to sales of KRW 1.7 billion last year. Daewoong Bio’s Verakan and Sam Chung Dang Pharm’s Venlafect, Youngjin Pharm’s Venfaxine raised sales of KRW 0.26 billion, 0.53 billion, and KRW 6.8 billion last year. Meanwhile, observations show that Alvogen Korea’s Ivenxine withdrew from the prescription market after raising KRW 12 million in sales in 2019.
- Opinion
- [Reporter’s View] Bitter ERPs following spin-offs and sales
- by Eo, Yun-Ho May 17, 2023 05:38am
- The operation of Early Retirement Program (ERP) schemes following sales of business units has been frequenting as an issue in the pharmaceutical industry. Although the spin-off and sales by multinational pharmaceutical companies aim at ‘separation of innovation and legacy, ‘this premise of ‘selecting and focusing’ on its strengths inevitably entails the negative situation of ‘layoffs.’ Also, the layoffs that occur from sales of businesses are different from general ERPs. Despite the notion of ERPs being a ‘voluntary retirement,’ ERPs that occurs during such situations are much less ‘voluntary.’ This is why such ERPs bring about labor-management conflict. Of course, multinational pharmaceutical companies provide immense compensation for their ERPs. In particular, the ERPs offered after spin-offs or sales offer industry-leading compensation. For employees that were considering whether to change jobs, ERPs can even be a fortune. However, there is no guarantee that there will be new jobs for everyone. Also, for some, a company is more than a place to earn a living but is also a place of value and pride. If layoffs are unavoidable, the company should focus on providing maximum compensation and ensuring succession of employment. The coercions being made under the word ‘voluntary ERP’ must be addressed, and the size of the layoffs should not be determined by a rule of thumb. There is no such thing as good layoffs. Only a few leave satisfied. Some will feel a sense of loss and separation just by the fact that they were being laid off as 'legacy'. As the companies that were the pride and hope of their employees, the companies should work to live up to their employee's expectations, whether those are being laid off as ‘legacy’ or not, and actively work to persuade its head office and take responsibility for the future course of the employees, if there is even the smallest room for improvement.
- Company
- Hemophilia drug Hemlibra coverage expansion
- by Chon, Seung-Hyun May 17, 2023 05:37am
- Hemlibra JW Pharmaceutical has laid the foundation for developing Hemlibra, a treatment for hemophilia, into a large-scale product. With the expansion of benefits, treatment benefits are provided to more patients, and expectations for sales growth are rising due to increased prescriptions. According to the Ministry of Health and Welfare on the 16th, Hemlibra will be covered for hemophilia A patients over the age of 1 who do not possess factor VIII antibodies from this month. Hemlibra is a routine prophylaxis for hemophilia A, which is caused by a deficiency of coagulation factor VIII. It is a product that applies bispecific antibody technology that simultaneously binds to blood coagulation factor 9 and factor 10. Unlike existing coagulation factor 8 preparations, it is the first non-coagulation factor preparation and can be administered subcutaneously once every 4 weeks at most. Hemlibra was developed by Chugai Pharmaceutical, a subsidiary of the global pharmaceutical company Roche. JW Pharmaceutical secured domestic development and sales rights for Hemlibra in 2017 and received permission from the Ministry of Food and Drug Safety in 2019. Chugai Pharmaceutical applied health insurance benefits to severe hemophilia A antibody patients in May 2020, and from this month, benefits were extended to non-antibody patients. According to JW Pharmaceutical, there are 1,746 patients with hemophilia A in Korea. Among them, severe hemophilia A patients were 1259, or 72%. Among patients with severe hemophilia A, 27 patients had antibodies and 1171 patients had non-antibodies. JW Pharmaceutical explained, “More than 60% of hemophilia A patients in Korea are covered by Chugai Pharmaceutical’s benefits.” JW Pharmaceutical expects that Hemlibra will provide great treatment benefits to patients based on the efficacy and safety proven by large-scale clinical trials. In the HAVEN1 clinical trial for antibody patients, it was confirmed that Hemlibra prophylaxis compared to the existing bypass therapy prophylaxis therapy reduced the annual bleeding rate (ABR) by 3.3 times by about 79%. In the HAVEN3 clinical trial for non-antibody patients, it proved an effect of reducing approximately 68% of the ABR (Annual Bleeding Rate) 1.5 times compared to prophylactic factor 8 therapy. The company explains that Hemlibra has also proven safety through clinical results. HAVEN 1~4 Clinical Results Among patients who received Hemlibra, most side effects were mild injection site manifestations. Quarterly Hemlibra sales (unit: million won, source: IQVIA) The company said, "Hemlibra has a long half-life of 28 days, so it increases patient convenience by reducing the number of administrations once every 4 weeks, compared to 2-3 times a week for conventional prophylaxis." In contrast to having to do intravenous injection, subcutaneous injection is possible, so it can help patients who have difficulty in intravenous injection or pediatric patients.” According to IQVIA, a pharmaceutical research institute, Hemlibra recorded sales of 7.6 billion won last year. In the first year of application in 2020, it raised 2.1 billion won, followed by 7.2 billion won in 2021, and showed an upward trend last year. Looking at quarterly sales, it recorded 2.2 billion won in the fourth quarter of last year, surpassing 2 billion won in sales for the first time. The cumulative sales of Hemlibra over the past three years were 17 billion won. The company expects that the amount of use will increase sharply as the target of benefit coverage expands significantly in the future. Last year, Hemlibra's global sales increased by 27% to CHF 3.823 billion. Currently, more than 20,000 patients in 144 countries around the world are using Hemlibra. “Hemlibra is an innovative new drug that can dramatically improve the quality of life of hemophilia patients with ease of administration and excellent efficacy,” said an official from JW Pharmaceutical. He said, "We expect that this new standard will expand the treatment accessibility of patients suffering from hemophilia and the choice of medicines for medical staff."
- Policy
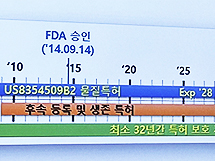
- Patent protection for 42 yrs for Humira/32 yrs for Keytruda
- by Lee, Hye-Kyung May 16, 2023 09:07pm
- It was analyzed that Humira maintained its patent protection period for at least 42 years and Keytruda for at least 32 years after filing for material patents with the Evergreening patent strategy. Evergreening refers to extending the term of a patent or extending the term of a patent for more than 20 years in the case of a patent to obtain more exclusive rights during the patent protection period. The types of evergreening strategies are representative of salt compounds, solvates, crystalline forms, optical isomers, dosage forms and pharmacokinetic data, manufacturing methods, and uses. Kim Tae-Kwon, responsible for the Korea Institute of Patent Technology Advancement, held on the last day of the 'Bio Korea 2023' event held on the 12th, 'Strategies for responding to original drugs and late-comer drugs according to the expiration of blockbuster drug patents'. Introducing the patent strategy. Kim Tae-kwon, head of the Korea Institute of Patent Technology Promotion The reason Humira and Keytruda are cited as examples is that the two medicines, excluding vaccines, respectively, ranked first and second in global sales in 2021, with sales of Humira at $20.694 billion and Keytruda at $17.186 billion that year. Humira applied for a substance patent in the United States in 1996, was registered as a patent in 2000, and received FDA approval in 2002. Since then, it has received FDA approval for additional indications such as psoriatic arthritis, ankylosing spondylitis, and Crohn's disease, and the substance patent period expired in 2016. So far, Humira has confirmed 746 patents based on 79 original patent applications. By type, 23 family groups (29.11%) for medicinal use and manufacturing method were distributed the most, followed by other 9 (11.39%), 8 diagnoses (10.13%), 7 formulations (8.66%), and 4 compositions. (5.56%), and 2 material improvements (2.53%). Responsibility Kim said, “Before the substance patent of Humira expired, we continuously extended the patent period with patents for medicinal use, formulation and composition patents, manufacturing method patents, diagnostic patents, automatic administration device patents, and pharmaceutical improvement patents.” It maintained its monopoly." In other words, Humira used an ever-greening patent strategy, such as forming a relatively superior patent barrier in the field of pharmaceutical use and manufacturing methods and forming a patent barrier in the pharmaceutical field as well. As a result, Humira was able to have a protection period of at least 42 years after applying for a substance patent and 35 years after FDA approval. After applying for a material patent in 2008, Keytruda registered a patent in 2013 and received FDA approval in 2014. Based on 67 original applications for patents, a total of 345 patents were confirmed. Looking at the distribution by type, 48 (71.64%) of the medicinal uses showed an overwhelming distribution. Next, 7 drugs (10.45%), 4 double antibodies (5.97%), 3 substances (4.48%), 3 diagnoses (4.48%), and 2 others (2.99%) are shown. Kim said, "There was also a case where the dosage and usage were changed as a barrier patent for the evergreening strategy of Keytruda products." "There was also data that it was to block the development of biosimilars as the patent term expires in 2028." and interpreted. In the case of Keytruda, it used the evergreening patent strategy, and it was analyzed that it had a protection period of at least 32 years after the substance patent and about 26 years after FDA approval. Kim explained, "As a result of the analysis of patent applications by type of biopharmaceutical, material patents must be filed, and based on material patents, applications are filed in the order of pharmaceutical use, composition, formulation, manufacturing method, diagnosis, and material improvement patents." As a result of comparing the evergreening strategy of biopharmaceuticals and synthetic drugs, there are patents for use, manufacturing methods, and formulations after substance patents, but synthetic drugs have many improved patents such as crystal forms, optical isomers, polymorphs, and intermediate patents for chemical formulas. However, as for biopharmaceuticals, there were patents for improving antibody or protein drugs. In addition, substance patents are important, but in the case of manufacturing method patents, it was noted that the distribution of biopharmaceuticals was high and the distribution of synthetic drugs was low. Kim said, "One thing to look carefully at is the expiry of the duration of a biopharmaceutical, but we need to look at changes in the patent for formulation, usage, and dosage in the use patent."
- Company
- Crysvita, the first fast-track drug by Yoon gov, is expected
- by Eo, Yun-Ho May 16, 2023 09:06pm
- It is now possible to prescribe 'Crysvita', the No. 1 rapidly registered drug of the Yoon government. This drug recently passed the Drug Committee (DC) of medical institutions such as SNUH. Kyowa Kirin Korea's (XLH rickets treatment Crysvita is a pediatric treatment that has been clinically proven effective in the field of diseases for which there is no equivalent drug or treatment. ) was listed on the list of insurance benefits. Since it is a drug that requires prior approval for reimbursement, it will take more time until the first prescription comes out. Crysvita's reimbursement criteria are for patients with X chromosome-linked hypophosphatemic (XLH) rickets aged 1 to 12 years who have not been controlled despite continuous administration of existing treatments (active vitamin D preparations, etc.) for more than 6 months. Symptoms such as growth retardation, dental abnormalities, lower extremity bone deformity, premature cranial fusion, and increased intracranial pressure are confirmed, and 'RSS level is 2 points or more, hypophosphatemia (less than 3.0 mg/dL) and loss of renal phosphorus (TmP/GFR reference value) less) must be proven. Until the registration of Crysvita, pediatric XLH rickets was treated with large doses of phosphate and vitamin D, but it was administered 4 to 6 times a day, and about 30% required surgery. Kang Hee-kyung, a professor of pediatrics at Seoul National University Hospital, said, "The existing treatment method of directly administering insufficient phosphate has a limit in that a vicious cycle occurs in which 'FGG23' is further activated and the degree of phosphate reabsorption further decreases. Crysvita is not a method of adding phosphate, It is a fundamental treatment that inhibits FGG23.”
- Company
- JAKi Cibinqo owns strengths in dosage adjustments
- by Jung, Sae-Im May 16, 2023 05:40am
- Pfizer's JAK inhibitor ‘Cibinqo (abrocitinib)' has embarked on a full-fledged journey to expand its prescriptions. After landing in major general hospitals at the end of last year, the drug is likely to be registered for reimbursement within the first half of this year. Cibinqo is a Janus kinase 1 (JAK1) inhibitor approved by the Ministry of Food and Drug Safety in November 2021. It is the 4th JAK inhibitor introduced to Korea and the second JAK inhibitor drug introduced by Pfizer after Xeljanz. Unlike Xeljanz, which is only used for ulcerative colitis, Cibinqo is used to treat severe atopic dermatitis. Treatment options have increased significantly for severe atopic dermatitis starting with the introduction of the injection-type biologic medication and oral JAI inhibitors. In particular, a total of 3 oral JAK inhibitors are available for patient use in Korea. Olumiant (baricitinib) and Rinvoq (upadacitinib) have first entered the market and are being used with reimbursement, followed by Cibinqo. Although Cibinqo is a latecomer, it owns a differentia table property from existing drugs. Dailypharm met with Jung-Im Na, Professor of Dermatology at Seoul National University Bundang Hospital to hear about its differentiable properties in the field. Jung-Im Na, Professor of Dermatology at Seoul National University Bundang Hospital Its first advantage is in its free dose control. Cibinqo comes in three doses: 50, 100, and 200mg. Although the recommended starting dose is 200mg, the dosage can be adjusted to 100 mg or 50 mg depending on the progress of treatment. If symptoms worsen with dose reduction, the doctor can increase treatment response again by increasing the dose and using local treatment together (JADE REGIMEN study). Professor Na said, “JAK inhibitors are fast and effective, but its exit strategy is considered a problem after the patient’s condition improves. In other words, ending treatment after seeing an effect is difficult with the use of JAK inhibitors. However still, due to its free dosage adjustments, it is attractive that the dosage can be reduced step by step from 200mg to 50mg.” He added, “No drug can be used for the rest of one’s life. Therefore, how to complete the treatment well is an important factor, and dose plays an important role. In particular, JAK inhibitors generally have a short half-life, and therefore disappear quickly from the body, leading to recurrence. Therefore, you cannot terminate treatment at once,” emphasizing the importance of dosage control. Major efficacy endpoint results in the JADE DARE trial Another advantage is in how it holds grounds based on a head-to-head trial for switching to the biological drug Dupixent. Among Cibinqo’s 7 Phase III clinical trials, the JADE DARE study is a head-to-head clinical trial comparing Cibinqo with Dupixent. As a result of separately administering Cibinqo 200mg and dupilumab 300mg in combination with a topical treatment for 26 weeks, the proportion of patients who achieved an improvement of 4 points or larger in the Peak Pruritus Numerical Rating Scale (PP-NRS4) at 2 weeks was 48.2% in the Cibinqo group, higher than the 25.5% of the Dupixent group. The proportion of patients who achieved a 90% improvement in the Eczema Area and Severity Index (EASI-90) at Week 4 was also significantly higher in the Cibinqo group (28.5%) than in the Dupixent group (14.6%). Moreover, the company also conducted the JADE EXTEND study, which studied the effect of switching patients who used Dupixent to Cibinqo. Patients were divided into those who were responsive to Dupixent and those that were non-responsive to Dupixent. 93.5% and 90.2% of patients who showed a response to Dupixent achieved EASI-75 after switching to Cibinqo 200mg and 100mg, respectively. PP-NRS4 was 89.7% and 81.6%, respectively. Among patients who did not respond to Dupxient, the proportion of those that reached EASI-75 was 80% and 67.7%, respectively, and PP-NRS4 was 77.3% and 37.8%, respectively. Efficacy of switchin gto Cibinqo(Abrocitinib) among patients thatresponsive to Dupxient(left) and those that were unresponsive (Data: Phase 3 efficacy and safety of abrocitinib in adults with moderate-to-severe atopic dermatitis after switching from dupilumab). The study provided evidence that patients who did not see an effect with Dupixent can expect an effect when they switch to Cibinqo. Previously, a clinical trial was also conducted with the JAK inhibitor Rinvoq on switching therapy from Dupixent. However, Cibinqo is the only drug that analyzed the effect of switching administration according to the patient’s presence or absence of an effect with Dupixent. This is why the trials have raised expectations of JAK inhibitors receiving expanded reimbursement as a replacement therapy when switching from Dupixent. Professor Na said, “The reason why the JADE EXTEND clinical trial is significant is that there are many patients who do not see an effect while using Dupixent, even though it is a good drug. However, these patients have to discontinue treatment and use immunotherapies for 3 months and see a deterioration in their condition before switching to a different treatment to receive reimbursement due to limited reimbursement standards. As Cibinqo has evidence prepared with clinical trials, the trials may be grounds to extend the drug’s reimbursement to switching medications. Also, For those who have not seen any effect after using Dupixent, the evidence is there to switch to Civinco.”
- Opinion
- [Reporter's view] AI drug development, should face reality
- by Hwang, Jin-joon May 16, 2023 05:39am
- "Artificial intelligence (AI) drug development will be useful, but there is a problem with only talking too much hope. Domestic AI drug development is still at an early stage, so more R&D is needed." These are the words the representative of an AI drug development company attending a recently held event related to digital healthcare threw to the debaters in the audience. It seems to point out that although new drug development using AI is being carried out in various parts of the world, companies that have achieved tangible results in Korea have yet to be seen. According to the Ministry of Science and ICT, new drug development is one of the projects that take an average of 10 years or more for a new drug to come to the market. It takes about 5 years to discover 5,000 to 10,000 promising candidates. Among the discovered candidate substances, 10 to 25 substances are entering preclinical trials. Of these, nine substances are approved for phase 1 clinical trial plans. The success rate is so low that there are only 5 in Phase 2, 2 in Phase 3, and only 1 in the market. In order to overcome the low success rate of new drug development and entry barriers that require long-term and high costs, a plan to utilize AI in the early R&D stage has been proposed. For traditional new drug development, a method of selecting a target disease and searching for a candidate substance by a research team reviewing about 400 to 500 related papers is used. AI is expected to have the advantage of being able to search for more than 1 million papers and hundreds of thousands of chemicals so that dozens of researchers can do the work that they have to do for 1 to 5 years more quickly. The AI drug development market is estimated to have high growth potential. The AI drug development market is expected to grow from 636.2 billion won in 2019 to 4.7693 trillion won by 2027, growing at an average annual rate of 28.63%. Global pharmaceutical companies such as AbbVie, Amgen, AstraZeneca, Boehringer Ingelheim, GSK, Janssen, Eli Lilly, MSD, Novartis, Pfizer, Sanofi, and Roche have also actively collaborated with AI drug developers to discover candidate substances. In 2021, a candidate substance for the treatment of pulmonary fibrosis discovered and designed through AI by Insilico Medicine in Hong Kong entered clinical trials. In October of last year, Verge Genomics, a US AI drug developer, started clinical trials of candidate materials for amyotrophic lateral sclerosis (ALS, Lou Gehrig), which were secured using AI's new drug discovery platform Converge. Verge Genomics is a biotech company that has received investments from Eli Lilly, MSD, WuXi AppTec, and BlackRock. In Korea, the Ministry of Science and ICT invested 25.8 billion won for three years from 2019 to 2021 to develop new AI drugs. The main goal was to build a customized AI platform for each stage of new drug development. It aimed to lead the development of three areas: candidate substance discovery, drug re-creation, and smart pharmacovigilance. Through a three-year support project in the field of AI new drug development, it developed an AI model that can be applied to neurodegenerative diseases and new anticancer drugs. The developed AI model is loaded into the public platform KAIDD and is openly operated so that researchers from industry, academia, and research institutes can utilize it. As a follow-up project that started in September of last year, the company plans to improve the data sharing and utilization environment by advancing the AI new drug development model by 2026. We plan to develop new drug candidates that can be applied for a clinical trial plan. There are several areas that need improvement in clinical prediction and targeting of clinical patient groups. It is also an important issue to establish social consensus and specific action plans that can utilize personal medical data classified as sensitive information. It is problematic that the AI drug development industry is not securing tangible results such as sales while talking only about the ideal use of technology, overseas cases, and the growth potential of the global AI market. It is necessary to face reality and solidify internal stability to be competitive.