- LOGIN
- MemberShip
- 2026-05-11 11:24:39
- Company
- Attempts to develop new drugs for hypertension
- by Hwang, Jin-joon May 22, 2023 05:41am
- Opinions were raised that it would be difficult for candidates under development as new drugs for hypertension, such as Baxdrostat, Aprocitentan, and Firibastat, to replace existing drugs. It is expected that it will fill the unmet demand rather than take the place of the prescribed treatment. Choi Ung-gil, professor at Konkuk University Chungju Hospital, is giving a presentation. (photo by Dailypharm) Professor Woong-Gil Choi of Chungbuk National University Hospital held a hypertension drug treatment update session at the '2023 Korean Society of Hypertension Spring Conference' held at COEX in Daegu on the 20th and said, "Major hypertension drug candidates are existing drugs rather than replacing drugs already prescribed in clinical settings. It will be a drug that can help the unfilled part.” The reason why the development of a new antihypertensive drug is needed is that it is a method to treat treatment-resistant hypertension. Professor Woong-Gil Choi explained, "Although the treatment control rate of hypertension has improved a lot, there is no further development after exceeding 70%." According to Professor Choi, major antihypertensive drug candidates include Baxdrostat, Aprocitentan, and Firibastat. Baxdrostat is a candidate substance secured by AstraZeneca, a global pharmaceutical company when it acquired CinCor Pharma, a US bio company. It is a new drug candidate for hypertension in the class of aldosterone synthase inhibitors (ASI). The efficacy of Baxdrostat in lowering blood pressure was confirmed in phase 2 clinical trial (BrigHTN) conducted on patients with treatment-resistant hypertension. In phase 2 clinical trial (HALO) conducted for uncontrolled hypertension patients taking up to two blood pressure medications, statistical significance was not achieved in the primary endpoint, but systolic blood pressure was reduced in subgroup analysis. Phase 3 clinical trials are expected to begin at the end of this year. Professor Choi said, “Baxdrostat appears to be relatively beneficial for hypertensive hypertension,” and “it is expected to give benefits to patients with primary aldosterone and metabolic syndrome.” Aprocitentan is a new drug candidate for hypertension being jointly developed by global pharmaceutical company Janssen and Swiss bio company Idorsia. It is an endothelin receptor antagonist. Applications for product approval were submitted to the FDA and EMA in December of last year and January of this year, respectively. Clinical data of Baxdrostat (Photo by Dailypharm) Aprocitentan has been confirmed to have a significant blood pressure-lowering effect in patients whose hypertension is not well controlled despite taking three or more existing treatments in phase 3 clinical trials (PRECISION). Professor Choi explained, "Although Aprocitentan has a stronger blood pressure lowering effect when used with other drugs, care should be taken about the fact that edema occurred after using the drug compared to placebo." Firibastat is a candidate material being developed by Quantum Genomics, a French biotech company. Firibastat is a candidate in the class of brain aminopeptidase A inhibitors. It is a mechanism that suppresses the production of angiotensin 3 in the brain's renin-angiotensin system (Brain RAS). Firibastat's efficacy with statistical significance was confirmed until the phase 2 clinical trial was conducted for patients with treatment-resistant hypertension. Afterward, it failed to achieve the primary evaluation index in phase 3 clinical trial (FRESH). Quantum Genomics is revising its development strategy to find new indications after the early termination of clinical trials. Professor Choi predicted, “There are still many cases of treatment-resistant hypertension, but if the development of a new drug for hypertension is successful, it will be possible to increase the treatment effect by adding it to existing drugs.”
- Company
- Forxiga price cut enforcement suspension extended
- by Jung, Sae-Im May 19, 2023 05:48am
- A decision on whether to suspend the execution of drug price cuts for AstraZeneca Korea’s diabetes treatments Forxiga and Xigduo is expected to be decided at the end of this month at the earliest. The temporary suspension period, originally until May 19, is also extended. On the 16th, the first division of the Seoul Administrative Court held an interrogation date for the suspension of drug price cuts filed by AstraZeneca Korea against the Ministry of Health and Welfare. The court, which conducted a private interrogation, decided to decide whether to quote a suspension of execution between the end of this month and the beginning of next month. As the court’s decision on whether to suspend enforcement is over 19 days away, the temporary suspension period for drug price cuts is also expected to be extended. Previously, the court had temporarily maintained the drug price until May 19, the scheduled date of the suspension trial. Forxiga and Xigduo, SGLT-2 inhibitors, are blockbuster products that raise outpatient prescriptions worth 90 billion won yearly as Dapagliflozin-based diabetes treatments. However, as a number of generics containing dapagliflozin were registered for reimbursement last month, they were subject to drug price cuts. The Ministry of Health and Welfare announced that it would cut the prices of Forxiga and Xigduo drugs by 30% from May 1 following the listing of generics. AstraZeneca Korea objected to this and filed an administrative lawsuit and at the same time applied for suspension of execution. As Forxiga and Xigduo also have indications for chronic heart failure and chronic nephropathy that have not expired patents, listing generics with only diabetes indications cannot be the basis for lowering original drug prices. It also argued that if the suspension of execution is not accepted, there is a risk of damage that is difficult to recover. If the court accepts the company's argument, the company can avoid losses of about 27 billion won a year until the prominent lawsuit is decided. In many cases, it takes more than three years from the citation of the suspension of execution to the cancellation of the drug price cut to the Supreme Court, so the company can prevent losses of tens of billions of won. However, it can be a burden that the judiciary's decision on the drug price cut enforcement suspension has been pointed out one after another it is fragmentary. In Korea's drug pricing system, which does not differentiate drug prices according to indications, questions are being raised as to whether the company's claims of inconsistency in price cuts due to inconsistency in drug prices are reasonable. This is because if the Ministry of Health and Welfare wins the prominent lawsuit after citing the suspension of execution, it will not be able to avoid criticism that the court cited the application for suspension of execution too broadly.
- Company
- HLB applies for liver cancer drug approval to FDA
- by Lee, Seok-Jun May 19, 2023 05:47am
- HLB submitted a New Drug Application (NDA) to the FDA for Rivoceranib, a targeted anti-cancer drug under development as a first-line liver cancer drug. This is the first time that a domestic bio company has completed its own clinical trials for its anti-cancer drug substance and proceeded with the new drug approval process in the global market. HLB started global clinical trials of Rivoceranib in 2011. Through its US subsidiary, Elevar Therapeutics, it has developed the combination of Rivoceranib and Camrelizumab as a first-line treatment for liver cancer. In the global phase 3 (CARES 310) study of 543 patients in 13 countries, compared to control sorafenib, 3 CR vs. 1 person, mOS 22.1 months vs. 15.2 months, mPFS 5.6 months vs. 3.7 months, ORR 25.4% vs. 5.9% was derived. It demonstrated therapeutic efficacy regardless of region (Asia vs. non-Asia) and cause (viral vs. non-viral). In particular, the Hazard Ratio was 0.62 for the overall survival period and 0.52 for the progression-free survival period, lowering the patient's risk of death by 40-50%. The FDA said at the pre-NDA (meeting before the application for new drug approval) that there was 'no problem' in the NDA submission for the Rivoceranib combination method. China already approved it as a first-line treatment for liver cancer in February this year. Rivoceranib is a TKI oral drug that effectively kills cancer by inhibiting VEGFR-2 and blocking the supply of oxygen and nutrients essential for cancer growth. To date, there is no approved first-line treatment for liver cancer that is a combination of a TKI anticancer drug with an angiogenesis inhibitory mechanism and an immune anticancer drug. HLB CEO Kim Dong-gun said, "We will do our best for the remaining procedures so that liver cancer patients and their families waiting for new treatment options, as well as employees and shareholders who have been with us on the long journey of new drug development can feel comfort and pride." On the other hand, HLB holds the global patent for Rivoceranib, HLB Life Science holds the Korean copyright and some profit rights in Europe and Japan, and Jiangsu Hengrui Medicine holds the Chinese copyright. All other global rights belong to Elevar Therapeutics.
- Company
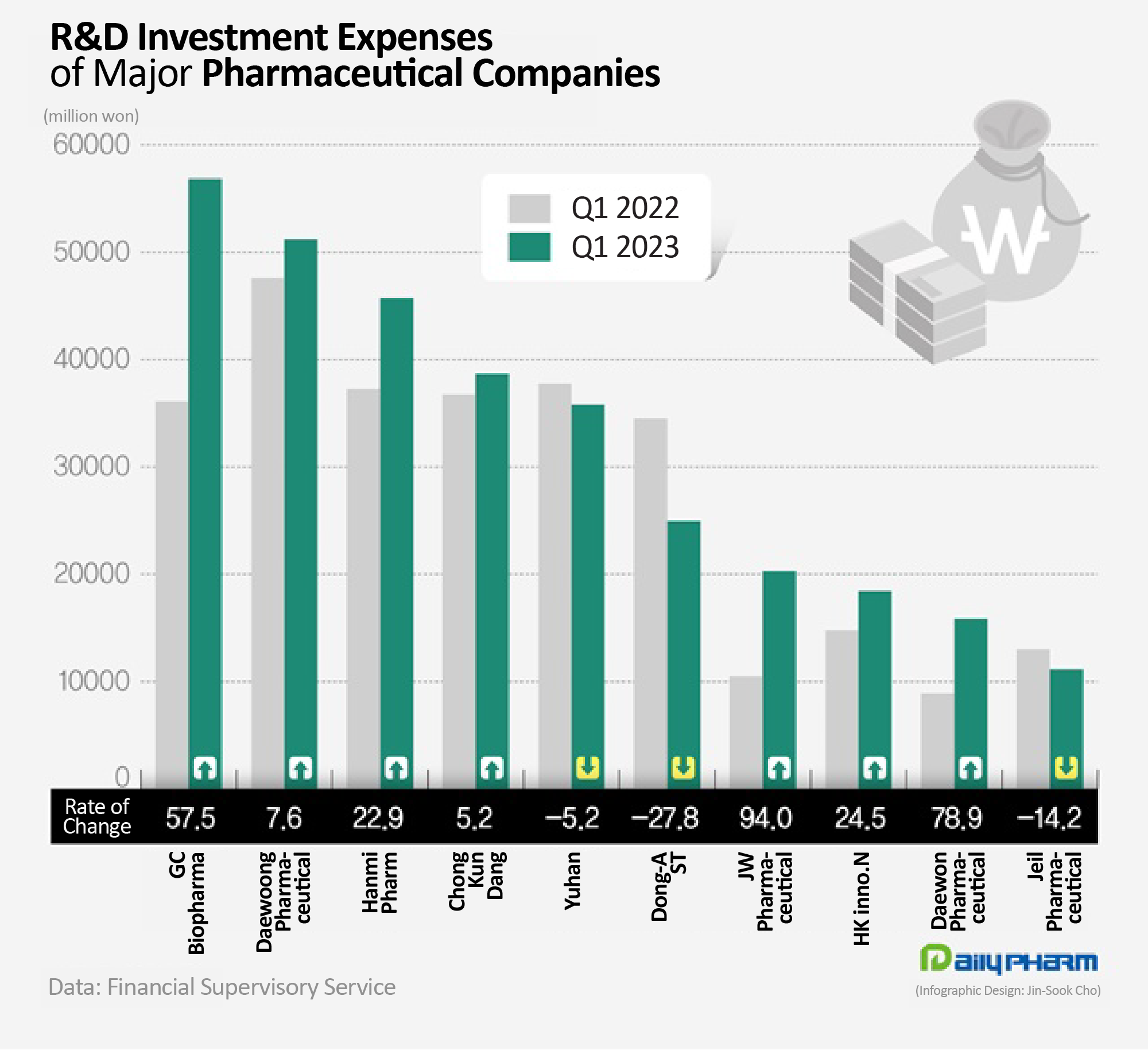
- Korean pharma industry expands R&D investments
- by Chon, Seung-Hyun May 19, 2023 05:47am
- Pharmaceutical companies have vigorously engaged in research and development (R&D) activities to discover future foods. 7 out of 10 major traditional pharmaceutical companies increased their R&D investment compared to last year. R&D expenditures have increased significantly due to the development of new drugs and the introduction of new R&D pipelines. In particular, R&D investments by GC Biopharma and JW Pharmaceuticals soared. According to the Financial Supervisory Service on the 25th, the R&D investment made by 20 major biopharmaceutical companies in Q1 totaled up to KRW 390.6 billion, which was 13.4% increase from the previous year. The top 20 sales of traditional pharmaceutical companies that submitted quarterly reports were counted. 14 out of 20 major pharmaceutical companies saw an increase in their R&D investment expense in Q1 this year compared to last year. Among traditional pharmaceutical companies in Korea, GC Biopharma was found to have spent the most on R&D. GC Biopharma’s R&D expenses in Q1 amounted to KRW 56.9 billion, which is a 57.5% YoY increase. GC Biopharma’s R&D expenditure increased because it recently introduced a new pipeline from a foreign company. In February, GC Biopharma signed an Asset Purchase Agreement with Catalyst Biosciences, a US new drug developer, for a pipeline related to rare blood clotting disorder. GC Biopharma acquired a total of 3 programs, including ‘Marzeptacog alfa (MarzAA)’ that the company is developing in the Global Phase III trial. In March, GC Biopharma exercised its license option to the Canadian company, Acuitas Therapeutics, for its lipid nanoparticle (LNP) delivery system to develop a messenger RNA (mRNA)-based therapeutics. LNP is a delivery system that safely transports nanoparticles to cells in the body to enable mRNA to function. The ratio of R&D investment to sales at GC Biopharma increased twofold from 8.7% to 16.3% in one year. Daewoong Pharmaceutical's sales increased 7.6% YoY to reach KRW 51 billion in Q1 this year. Daewoong Pharmaceutical is currently developing new drugs for ulcerative colitis, idiopathic pulmonary fibrosis, obesity, autoimmune diseases, and infectious diseases. It is also conducting joint research with HanAll Biopharma, Daewoong Therapeutics, Oncocross, and D&D Pharmatech. Daewoong Pharmaceutical received approval for its gastroesophageal reflux disease treatment Fexclu in 2021 and succeeded in commercializing Envlo, a new diabetes SGLT-2 inhibitor class drug last year. Hanmi Pharmaceutical’s R&D investment also increased 22.9% YoY to reach KRW 45.7 billion in Q1 this year. Hanmi Pharmaceutical is developing new drugs for nonalcoholic steatohepatitis and idiopathic pulmonary fibrosis in the field of new biological agents. The company is also developing new combination drugs for diabetes and antithrombosis. Among major pharmaceutical companies, JW Pharmaceutical’s R&D expenditure increased the greatest. JW Pharmaceutical’s Q1 R&D expenditure was KRW 20.3 billion, increasing 94.0% YoY from the KRW 10.5 billion in Q1 previous year. JW Pharmaceutical started a Phase III clinical trial of 'URC-102', a gout treatment, at the end of last year. URC-102 is a uric acid excretion promoter that inhibits Urate transporter 1 (URAT)-1, which allows uric acid to be absorbed back into the body. It is effective for gout disease caused by hyperuricemia in which the concentration of uric acid in the blood is abnormally high. The Phase III trial will compare URC-102 with a total of 588 gout patients with the existing treatment febuxostat. Daewon Pharmaceutical and Handok’s Q1 R&D investment increased 50% from the previous year. R&D expenditures increased by over 20% at Hugel, HK Inno.N, and Dong Wha Pharm. On the other hand, the R&D investment amount of companies including Dong-A ST, Il-Yang Pharmacuetical, Jeil Pharmaceutical, Boryung Pharmaceutical, United Pharm, and Yuhan Corp decreased YoY. In terms of the R&D-to-sales ratio, Daewoong Pharmaceutical’s rate was highest at 17.5%. GC Biopharma, Dong-A ST, Daewon Pharmaceutical, Hanmi Pharmaceutical, JW Pharmaceutical, United Pharm, Samjin Pharm, Chong Kun Dang, and Ilyang Pharm invested more than 10% of their sales in R&D.
- Company
- 90% of pricing managers unsatisfied with new drug price
- by Eo, Yun-Ho May 19, 2023 05:46am
- Study results showed that about 90% of the drug pricing managers in Korea are not satisfied with the value recognized for new drugs. Recently, a study on ‘'An Industry Survey on Unmet Needs in South Korea’s New Drug Listing System' was published on the online version of the medical science journal Springer (https://link.springer.com/article/10.1007/s43441-023-00531-3). 6 authors including Professor Jong-Hyuk Lee of the Chung-Ang University College of Pharmacy, expert advisory member Sungju Kim from Lee&Ko participated in the study. The study was conducted with the cooperation of 3 industry associations: Korea Pharmaceutical and Bio-Pharma Manufacturers Association, the Korean Research-based Pharmaceutical Industry Association, and the Korea Biomedicine Industry Association. Members of the associations that in charge of insurance drug pricing participated in the study. The total number of respondents was 56, 34% of which were from domestic companies and 66% from multinational pharmaceutical companies. The survey consisted of questions that study the industry's satisfaction with the current insurance system, requests for improvements in the new drug reimbursement listing system, including the pharmacoeconomic evaluation system, pharmacoeconomic evaluation exemption system, and risk-sharing agreement scheme, and the need to introduce systems that have not been introduced to Korea. According to the results, 64.3% and 89.3% of respondents answered that they were dissatisfied with the patient accessibility and value recognition of new drugs, respectively, and answered that institutional improvement for rare diseases was needed the most (41.1%). Regarding the pharmacoeconomic evaluation system, 92.9% said that the ICER threshold needs to be improved. In the risk-sharing system, the reimbursement standard expansion system required improvement (91.1%), and in the risk-sharing agreement scheme, the expansion of target diseases (89.3%) was needed. Also, regarding the reimbursement listing of anticancer drugs, 83.9% answered that the Cancer Disease Review Committee, which determines the reimbursement standards, needs to be improved. In the case of general drugs, the majority of respondents said that the drug price negotiation system needs to be improved. When asked about the need to introduce a system that is yet to be introduced, respondents expressed a high need to introduce a drug pricing system by indication and insisted that the system should be introduced regardless of the severity of the disease, whereas the pre-listing post-evaluation system should be limitedly applied to life-threatening diseases. Sungju Kim said, “In general, study results showed low satisfaction with the current system and a great need for its improvement. As pharmaceutical companies are also important stakeholders, their opinions should also be considered in the process of pricing and reimbursement policy reforms.”
- Company
- Ibrance emerges as a new drug partner for metastatic breast
- by Jung, Sae-Im May 19, 2023 05:46am
- Professor Joo-Hyeok Son, Department of Oncology, Yonsei Cancer Hospital CDK4/6 inhibitor Ibrance is emerging as a combination partner for metastatic breast cancer drug developers based on its long-accumulated treatment experience. Even if the dose is increased, there is little concern about side effects, so it is expected that it will be used as a variety of combination drugs. Ibrance is the first CDK4/6 inhibitor developed by Pfizer and was launched in Korea in the fourth quarter of 2016. It provided a new treatment option for patients with HR+/HER2- metastatic breast cancer who had to use anti-hormonal drugs such as aromatase inhibitors or chemotherapy if not managed with these drugs. Ibrance, which held the top spot in the CDK4/6 market for five years, faced a decline for the first time last year. According to IQVIA, a pharmaceutical market research institute, Ibrance sales decreased 14% from 65.6 billion won in 2021 to 56.2 billion won last year. The rapid growth of generics has affected Ibrance's sales. Generics such as Verzenio and Kisqali are breaking down Ibrance's dominance through more sophisticated clinical trials and new field development. In particular, with generic drugs expanding their scope to early breast cancer, prospects are raised that Ibrance's position, which is limited to metastatic breast cancer, will narrow. In this situation, Iran is trying to turn around by emerging as a new drug combination partner for metastatic breast cancer. In an interview with Daily Pharm, Professor Sohn Joo-hyeok of the Department of Oncology at Yonsei Cancer Hospital said, "To use an oral SERD instead of an aromatase inhibitor, it must be used in combination with CDK4/6, but Irance is widely adopted as a combination drug." It means being recognized." Recently, oral SERD development is in full swing as a CDK4/6 inhibitor combination therapy for breast cancer. Many global pharmaceutical companies such as Pfizer, AstraZeneca, and Menarini jumped into the market. Big pharma is in the midst of studying combination therapy with CDK4/6 inhibitors as well as oral SERD monotherapy, and most of them chose Ibrance as the combination drug. Professor Sohn cited safety as the reason why Ibrance received much love calls as a combination drug. Professor Sohn said, "Ibrance's strength is that it is stable as it has accumulated the longest treatment experience among CDK4/6 inhibitors." It's less, so I'll consider it first," he explained. This evaluation was proven with real-world data. The Ibrance P-REALITY X study conducted by Pfizer is a large-scale real-world study that retrospectively analyzed the data of 2888 patients with HR+/HER2- metastatic breast cancer enrolled from February 2015 to March 2020. Patients receiving Ibrance plus Letrozole combination therapy as a first-line treatment option were compared with patients on Letrozole monotherapy. As a result of matching the baseline characteristics of the two groups similarly, the median overall survival (mOS) of the Ibrance group was 49.1 months, which was significantly prolonged compared to 43.2 months of the Letrozole single group, reducing the risk of death by 24%. It is less toxic, so even elderly patients can use it without burden. As a result of a sub-analysis examining Ibrance and Letrozole combination therapy in elderly patients aged 65 years or older, the median progression-free survival (mPFS) was 30.6 months, compared to 19.1 months in the control group. Professor Sohn said, "Recently, I prescribed Ibrance to an 80-year-old elderly patient with an anti-hormone drug. The treatment went well, and people around me said, 'How can you correct this when you are receiving chemotherapy at an advanced age?' I always worry about prescribing medications for cancer, but Ibrance greatly eases that burden."
- Policy
- Revival of omega-3 fatty acids...4g high-dose recommended
- by Choi, sun May 18, 2023 05:45am
- The Korean Society of Lipid and Atherosclerosis (KSoLA) disclosed the full version of its 5th edition of the Korean Guidelines for the Management of Dyslipidemia, in which the use of omega 3 was subdivided into the use of 'high dose and refined ingredients'. Although there has been controversy over its efficacy, the new guideline puts weight on the fact that the benefits of its use still outweigh its non-use. Annual Scientific Conference of the KSC with Affiliated Cardiac Societies On the 22nd, the Annual Scientific Conference of the KSC with Affiliated Cardiac Societies that was organized by 8 academic societies including the Korean Society of Cardiology, the Korean Heart Rhythm Society, the Korean Society of Heart Failure, and KSoLA, announced the ‘2023 KSoLA Guideline Update,’ ‘Evidence of Guidelines,’ and ‘Limitations in the Evidence’ were announced. A simplified version of the revised guidelines was announced last year, but this year, the full version was released to support its details and rationale. The new changes made in the 5th version were ▲the Method of diagnosis and standards, ▲Treatment standards. First, in the new diagnosis method and standard, the KSoLA showed the result that the non-fasting triglycerides level showed a high correlation with the risk of cardiovascular disease. In the standards, patients with coronary artery disease were recommended to lower their LDL cholesterol target level from less than 70 mg/dL to less than 55 mg/dL and lower it by more than 50% from baseline. In the case of patients with diabetes, the target goal is less than 70 mg/dL for patients with a disease duration of 10 years or more, one or more additional risk factors, or target organ damage, depending on the risk level. In addition, diabetic patients with 3 or more target organ damage or major cardiovascular disease risk factors can selectively consider reducing LDL cholesterol to less than 55 mg/dL. Furthermore, as exercise therapy, the recommendation grade for the use of wearables for fitness was newly presented as IIa, and additionally methods for using wearable equipment and a strategy for promoting physical activity were suggested. In the full version, society took a somewhat reserved position on the use of Omega 3, which was plagued by controversy over its cardiovascular protective effect. Professor Ye-Seul Yang (Endoctrioinology, SNUH), who presented on the 'New Changes on the KSoLA Guidelines,’ said, “The 5th edition of the revised guidelines segmented and reinforced the treatment criteria and treatment targets for dyslipidemia. The new guideline recommended different treatment standards by subdividing diabetes by risk group." Yang said, “Individualized guidelines were set according to specific groups such as those with stroke, chronic kidney disease, the elderly, adolescents, and familial hypercholesterolemia. In terms of drug therapy, the recommended grade was raised to emphasize that statins are a first-line treatment drug, and introduced icosapent ethyl(IPE) and the role of fibrate and omega-3 fatty acids in managing triglyceride.” As for whether omega 3 actually shows a protective effect against cardiovascular disease, large-scale studies have come to different conclusions. The most recent study that proved its efficacy observed an effect when only the IPE component was isolated from the omega-3 and used in high dosages. The KSoLA newly included the use of IPE in the recommendation for patients with atherosclerotic cardiovascular disease or diabetes who still have hypertriglyceridemia even after LDL cholesterol is controlled below the target level with statins. In the treatment of hypertriglyceridemia, "Use of fibric acid derivatives or omega-3 fatty acids that mainly lower triglycerides first are recommended as a priority (IIa, A), and even after achieving LDL cholesterol below the target level through therapeutic lifestyle improvement and statin drug treatment, if the triglyceride is 200 mg/dL or higher or the non-HDL cholesterol level is higher than the target value, drug treatments to lower the triglyceride can be considered (IIa, B)." KSoLA said, "In the recent REDUCE-IT study, 4g of IPE was administered per day in patients at high risk of atherosclerotic cardiovascular disease or diabetes, which reduced the incidence of cardiovascular disease by 26% compared to placebo. If hypertriglyceridemia persists in high-risk patients at a 200 mg/dL or higher level even after lifestyle improvement and statin administration, additional IPE (4g per day) can be administered to prevent cardiovascular disease (IIb, B)." However, KSoLA added a provision on account of the controversy in place. KSoLA added, “The effect of omega-3 fatty acids on reducing the risk of cardiovascular disease is still in controversy because of the varying results shown depending on formulation or dosage. In the recent REDUCE-IT study that was conducted in patients at high risk of cardiovascular disease whose triglyceride levels were higher than 135-499 mg/dL, the use of IPE, a high-purity EPA, twice a day significantly reduced death from cardiovascular disease and the occurrence of ischemic disease." “In the European practice guidelines that were revised in 2019, control of triglyceride using IPE was recommended for patients at high risk of cardiovascular disease, however, IPE is not being sold in the market yet. Also, in another recent study, the STRENGTH study, the combination of EPA and DHA did not show any benefit in cardiovascular disease, but rather increased the risk of atrial fibrillation compared to the control group." Also, the guidelines emphasized the need to use a high-dose high-purity ingredient for the combined use of statin and Omega 3. KSoLA said, “Combination therapy can be used to reduce LDL cholesterol and triglyceride at the same time. "The combination of 4 g of Omega 3 per day with simvastatin significantly decreased triglyceride and slightly increased HDL cholesterol."
- Product
- Diabetes Association also paid attention to 'zero' drinks
- by Choi, sun May 18, 2023 05:45am
- Possible inhibition of glycemic response and increased risk of cardiovascular events This year, while the American Diabetes Association recognized intermittent fasting and time-restricted eating as part of a meal pattern based on research results that help reduce weight and improve blood sugar, the Korean Diabetes Association also reviewed low-carbohydrate diets and non-nutritive sweeteners. The Diabetes Association acknowledged the Mediterranean diet and the DASH diet, which have accumulated some evidence, while asking for a cautious approach in that non-nutritive sweeteners have mixed results. On the 12th, the Korean Diabetes Association held a spring academic conference at the Kimdaejung Convention Center in Gwangju, and disclosed the results of the review of evidence and revision work conducted by the Food and Nutrition Committee of the society. Professor Lim Jeong-hyeon, Seoul National University Hospital, Food and Nutrition Division (Cancer Committee, Korean Society of Clinical Nutrition) Numerous studies have already shown that healthy eating habits, including the Mediterranean, vegetarian, low-fat, low-carbohydrate, and DASH (salt-restricted diets designed to reduce blood pressure in people with hypertension), improve blood sugar, reduce weight, and reduce the risk of cardiovascular disease. being confirmed Because of this, the American Diabetes Association and other major treatment guidelines recommend individualizing various dietary patties and using them appropriately. In particular, according to the research results that time-restricted meals are helpful for weight loss and blood sugar improvement, in 2023 the American Diabetes Association included intermittent fasting and time-restricted meals as one type of meal pattern, and whether or not this was reflected in Korea has emerged as a matter of interest. This month, the Korean Society for Obesity published guidelines for low-carbohydrate diets that limit carbohydrate intake to 10-45% of total energy intake. situation you are doing. In the midst of this, the Korean Diabetes Association also presented an appropriate answer. The revised guidelines of the Korean Diabetes Association are largely divided into ▲ intake of carbohydrates in a form rich in dietary fiber ▲ refrain from sugar-sweetened beverages to minimize the intake of added sugars ▲ limited use of non-nutritive sweeteners for a short period of time. “The 2021 recommendation did not recommend an extreme diet method that did not prove long-term benefits,” said Lim Jeong-hyun, professor of nutrition at Seoul National University Hospital (Cancer Committee of the Korean Society of Clinical Nutrition). The method was observed and reflected in the 2023 recommendation.” "Mediterranean, vegetarian, low-fat, DASH, and low-carbohydrate eating patterns have demonstrated long-term benefits," he said. "Therefore, these eating patterns can be adapted according to goals and preferences (limited recommendations)." He said, “The American Diabetes Association revised the carbohydrate section of medical nutrition therapy in 2022 to emphasize the quality intake of carbohydrates. We presented the rationale for this and added an ultra-low-carbohydrate diet to the dietary pattern session.” In fact, as a result of analyzing 10 RCTs involving 1376 type 2 diabetes patients, it was found that glycated hemoglobin and body mass index decreased according to carbohydrate restriction. Professor Lim said, "In a 2022 meta-analysis study of 50 RCTs for patients with type 2 diabetes, reducing carbohydrate intake to less than 45% of total energy intake showed improvements in several indicators such as body weight, lipids, and glycated hemoglobin." For each 10% reduction, A1C decreased by 0.2%, FPG by 0.34mmol/L, and body weight by 1.44kg, and all serum lipid indicators were improved.” He said, "Up to 6 months, the rest of the indicators showed a linear decrease in the 40% carbohydrate diet, but triglycerides and LDL cholesterol showed a U-shaped shape." Although they decreased, their weight showed a U-shaped pattern." "Because a meal is a combination of various foods, there can be many patterns in a weight control diet," he said. It is important to increase individual compliance, such as using a Mediterranean diet or Mediterranean diet, and to allow patients to choose individually according to their goals and preferences, and to provide evidence for that.” In view of the consistent increase in the risk of developing diabetes when consuming sugar-sweetened beverages, the society recommended refraining from sugar-sweetened beverages while urging caution against consuming beverages that have been replaced with non-nutritive sweeteners. Professor Lim said, “The risk of diabetes rises by 51% when you consume one or more sugary drinks a day, and a 10% reduction in sugar-sweetened beverage intake consistently reduces the risk of diabetes.” It should be recommended to replace it with coffee.” He said, “The problem is that the effect of replacing sugar-sweetened beverages or sugar-sweetened beverages with artificial sweeteners is inconsistent. "I haven't made a clear statement," he said. He said, “In a 2021 study, sucralose and saccharin inhibited the blood sugar response, and a study published in NATURE this year showed that erythritol increased the risk of major cardiovascular events.” For this purpose, the use of non-nutritive sweeteners for a short period of time can be considered on a limited basis.”
- Policy
- Initial appvl rate of drugs subject to prior review varies
- by Lee, Tak-Sun May 18, 2023 05:45am
- As a result of analyzing the approval rate of prior authorization drugs over the past 10 years, the approval rate varied greatly according to the type of drug. However, unlike during the initial review, the review for maintenance therapy showed a high approval rate of 90%. Yong-Kyun Won, Professor of Radiation Oncology at Soonchunhyang University Cheonan Hospital, announced so through a retrospective record analysis study on the prior authorization drugs over the past 10 years (2021-2022).' The study was presented at the 22nd Annual Conference of the Korean Society of Insurance Medicine which was held on the 14th. The prior authorization system was implemented in 2012 to establish clear standards for the use of high-priced drugs and to prevent drug abuse. Soliris, Spinraza, Ultomiris, Strensiq, and Zolgensma, which are rare disease drugs and ultra-high-priced drugs that cost more than KRW 300 million won per year, receive health insurance reimbursement through the system. Crysvita was recently added as a drug that requires prior authorization. According to the study, drugs that were expensive but are essential for the treatment of rare diseases have been able to receive reimbursement through the system, and this pre-deliberation system has been successful, such as in managing the quality of treatment through patient monitoring (maintenance therapy), etc. However, the approval rate was different for each drug. In particular, the prior authorization approval rate for initial administrations ranged from 20% to 100% by product or indication. In comparison, the review approval rate for maintenance therapy exceeded 90%. The varying initial approval rate of prior authorization drugs (retrospective record analysis study on prior authorization drugs (2012~2022)) For example, in the case of patients who seek to use Soliris for aHUS, the initial approval rate was only 21.6%. On the other hand, drugs such as Ultomiris (77.8%) and Strensiq (100%) showed high approval rates. On the other hand, the number of acceptations of objections on the disapproval was low. Only 1 out of 17 objections in 2022 were accepted, and therefore the analysis was that it was a difficult environment for disapproved drugs to receive deliberations again. Professor Won expressed concerns about how the low approval rate may limit access to reimbursement. In addition, for drugs in need of urgent deliberations due to the child's age or disease type, Won explained that the system where institutions need to wait for announcements until the end of the month to see why their application was disapproved and what needs to be supplemented, should be improved as well. Professor Won said, “The prior authorization system is settling as an essential system in securing access to treatment for high-priced drugs that are being continuously introduced to the field. Doctors may feel it is difficult to use a drug if the approval rate is too low. Therefore, it is necessary to review whether the reimbursement standards are too strict and whether it needs revisions.”
- Opinion
- [Reporter's view] Are you ready to use
- by Lee, Hye-Kyung May 18, 2023 05:45am
- The 'Act on the Safety and Support of Advanced Recycles and Advanced Biomedicines' will be in effect for three years in August. The Advanced Recycled Bio Act prepares a system for securing the safety of advanced renewable medicine, provides a plan for technological innovation, and practical use, and stipulates the necessary matters to secure the quality, safety, and effectiveness of advanced biopharmaceuticals, and to support commercialization.The Advanced Recycled Bio Act has been implemented, and 36 cell treatment facilities and 32 human cell management institutions have been approved in Korea. Cell therapy is made in Korea to export technology to foreign countries, or the CMO/CDMO business of biopharmaceuticals is actively underway. As if proof of this, 'BioKorea 2023', which was held from May 10th to 12th, was presented by a high-tech renewable medical center for the first time, and various sessions were held on the theme of revitalizing clinical research on advanced renewable medicine and exploring the direction of the development of the high-tech biopharmaceutical industry. A speaker who attended the site at the time said that the regenerative medicine session was formed for the first time in Bio Korea, and that it was time to seek industrial development as well as domestic advanced regeneration bio research. However, it has only been three years since the High-Tech Recycled Bio Act was implemented, so it seems that there is still a long way to go. The Ministry of Health and Welfare has expressed its will to improve regulations to revitalize clinical research on cutting-edge bioregenerative medicine treatment technology. It is said that the clinical research application target will be expanded and the clinical research results can be linked with approval, but the position of the researchers conducting the actual research was a little different. It is said that it is urgent to prepare a legal basis for the activation of clinical research data based on the Temple of Heaven's Regenerative Bio Act. In fact, there have been various attempts to utilize the health care big data platform, but the linkage rate between each institution is low as it still hits the wall of personal information. Even if you use data from multiple platforms, you have no choice but to hit the limits of using it with your gender, name, and date of birth alone. In the end, this phenomenon seems to be bound to occur in the use of advanced regeneration bio clinical research data. There is a provision in the Advanced Recycled Bio Act that 'if you need a head of a safety management agency, you can request the provision of data such as unique identification information', but it is not enough to expand it to pharmaceutical companies and clinical research practitioners. In accordance with the Advanced Recycle Bio Act, the basic plan for advanced renewable medicine and advanced biopharmaceuticals is established every 5 years. Three years are coming, and the time is coming soon to come up with a second basic plan. When you try to take advantage of the accumulated endless health care big data, you have to remember the many hurdles. Data from high-tech regeneration and biopharmaceuticals that have just crossed 2/3 of the first basic plan is being collected one by one. In order to actively conduct clinical research with this data, it is necessary to come up with various strategies that can utilize individual unique identification information.