- LOGIN
- MemberShip
- 2026-06-11 00:25:13
- "Finished drug firms·API suppliers suffer from high exchange rate"
- by Chon, Seung-Hyun | translator Hong, Ji Yeon | 2026-04-03 08:02:47
The pharmaceutical industry is facing significant challenges conducting business amid high exchange rates and the war in the Middle East. As the KRW/USD exchange rate surpasses KRW 1,500, the cost pressure on imported Active Pharmaceutical Ingredients (APIs) is intensifying. Furthermore, with upcoming generic drug price cuts, seeking cheaper imported APIs is becoming increasingly difficult. The high exchange rate and price reductions are also major issues for API manufacturers.
According to industry sources on the 2nd, the KRW/USD exchange rate in the Seoul foreign exchange market reached 1,501.5 KRW on the 1st. Compared to KRW 1,352.6 on July 2 of last year, this represents an increase of more than KRW 150 in just eight months.
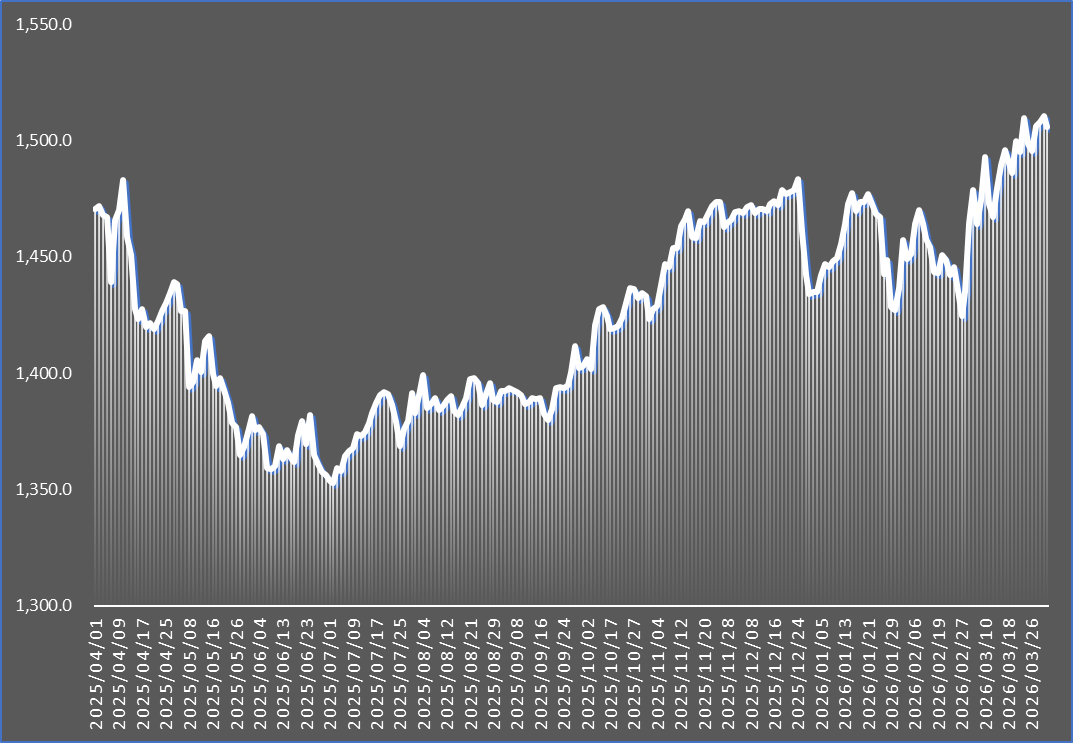
The exchange rate first surpassed KRW 1,500 on the 24th of last month and briefly stayed in the KRW 1,400 range for two days before surpassing KRW 1,500 again on March 29, continuing to increase. Amid concerns over a prolonged conflict stemming from the high-intensity standoff between the U.S. and Iran, the rate even surged past KRW 1,530 during intraday trading on the 31st, the highest level in 17 years since the global financial crisis in 2009.
The decline in the value of the Korean won directly translates to cost-push pressure for pharmaceutical companies. Since these companies are highly dependent on imported APIs, the core raw materials for medicines, the rise in the KRW/USD exchange rate directly increases production costs.
In 2024, the self-sufficiency rate for APIs was recorded at 31.4%, calculated using an average exchange rate of KRW 1,367. Self-sufficiency refers to the ratio of domestically produced products within the total market.
With 69.6% of APIs used domestically being imported, the reliance on foreign raw materials is absolute. Since US dollars are used even when purchasing APIs from China and India, the largest sources of imports, the impact of the rising exchange rate is unavoidable.
Recently, domestic pharmaceutical companies have also been considering switching API suppliers to reduce costs in response to the government's announced price cuts for generic drugs.
Under the reformed drug pricing system discussed by the Ministry of Health and Welfare (MOHW) at the Health Insurance Policy Review Committee on the 26th of last month, the price for both off-patent drugs and generics will decrease from 53.55% to 45% of the pre-patent-expiry price of the new drug. Mathematically, this is a 16.0% cut in generic prices.
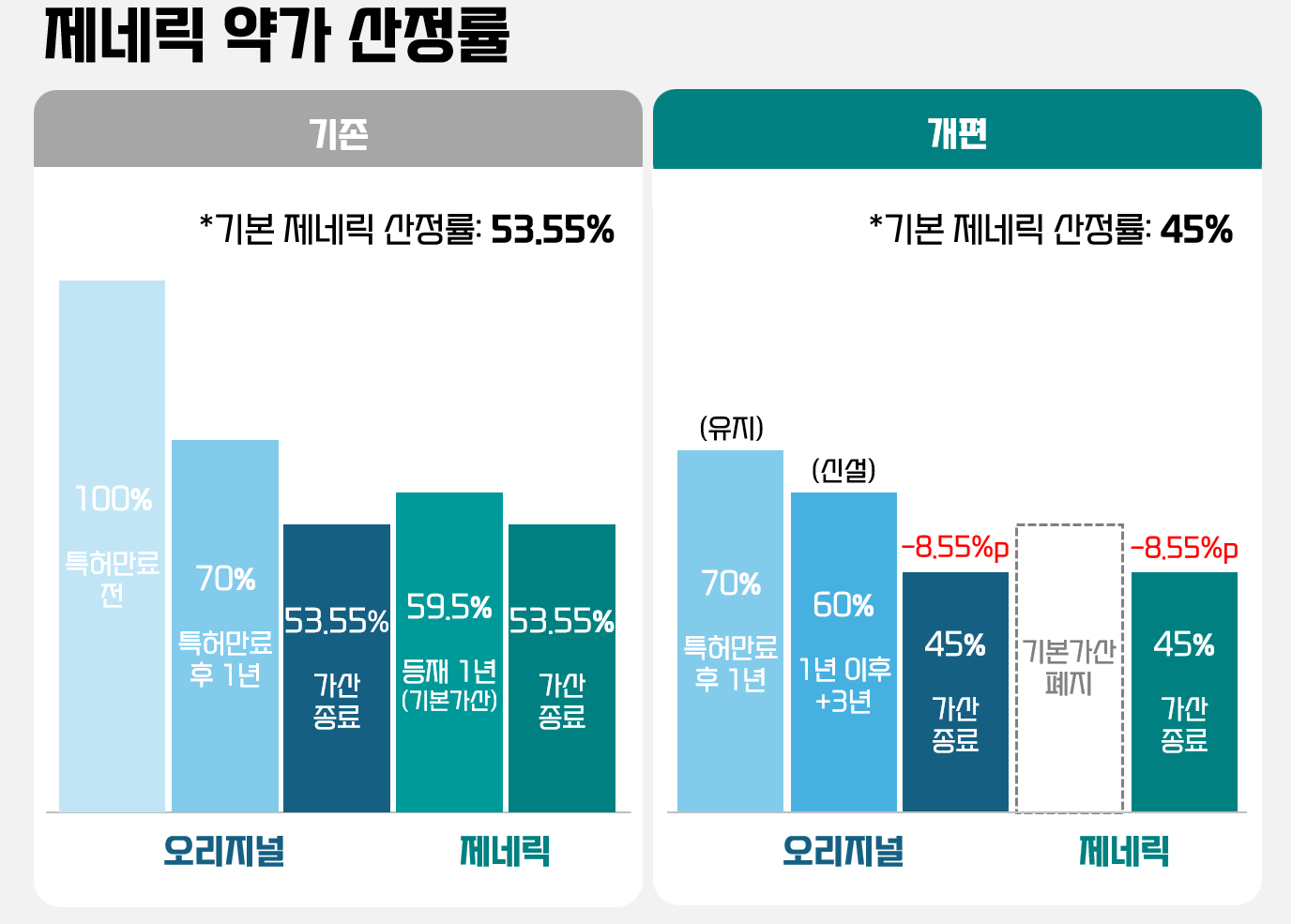
The price reduction range increases further when "top-tier price requirements," such as conducting bioequivalence (BE) studies and using registered drug substances (DMF), are applied to existing listed generics. Under the reform, the penalty for failing to meet these requirements will expand from 15% to 20%. Since July 2020, requirements have been in place that generics can receive the 53.55% maximum price only if they meet both the direct BE study and DMF criteria. For each unmet requirement, the ceiling price drops by 15%.
Applying the new 45% standard and the 20% cut for unmet requirements, generics failing one requirement drop to 36%, and those failing both drop to 28.8%. This means the price for a generic failing one requirement will be 20.9% lower than current levels, while those failing both will see a 25.6% decrease.
Due to these price-reduction pressures, pharmaceutical companies are forced to seek cheaper imported products rather than relatively expensive domestic APIs to save costs.
The value of API imports from China is already increasing. In 2024, Chinese API imports reached USD 816.32 million, up 110.2% from USD 388.31 million in 2014. In 2014, China was the 6th-largest source of drug imports for Korea, but it rose to 3rd place by 2024.
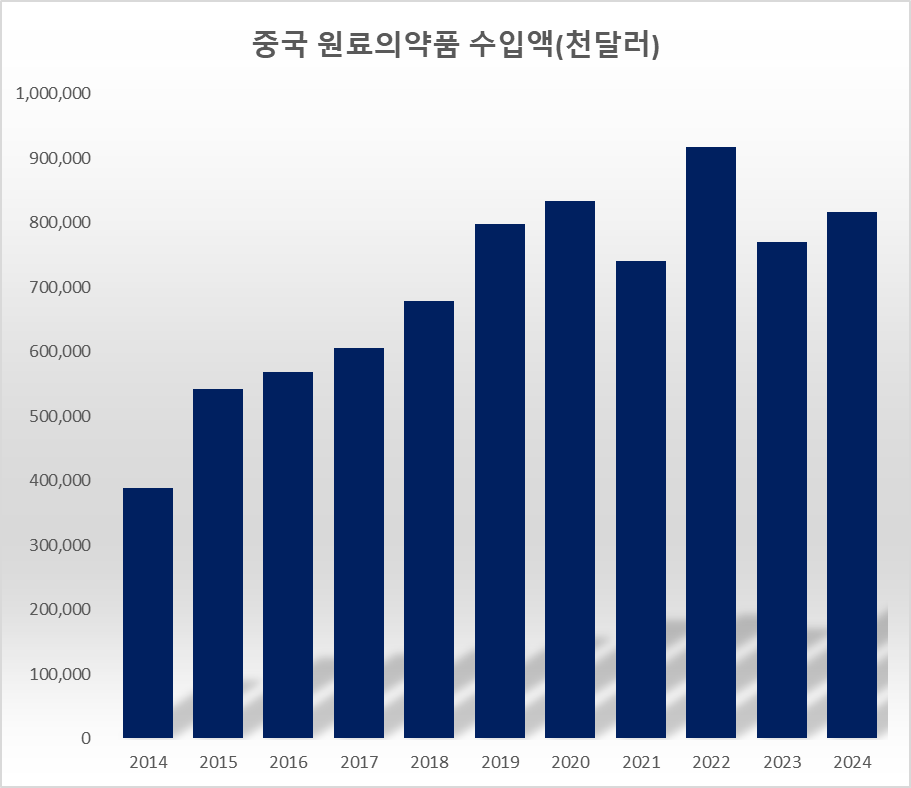
In 2024, the value of Chinese APIs used in Korea was KRW 1.1159 trillion. Of the KRW 4.4007 trillion in APIs produced in Korea, only KRW 1.43 trillion was used in the domestic market. This indicates that the amount of Korean and Chinese APIs used in the local market is roughly equal. Considering that Chinese APIs are generally cheaper than domestic ones, this suggests that the actual volume of Chinese APIs used by domestic companies overwhelms that of domestic APIs.
Under these circumstances, there are concerns that if generic prices drop further, more companies would refrain from using relatively expensive domestic APIs. Both finished drug manufacturers and API suppliers are structured in a way that could lead to simultaneous losses under pressure to reduce drug prices.
For API manufacturers, the high exchange rate and price cuts act as major setbacks. Even for domestically produced APIs, starting materials are often imported, so they must worry about rising costs due to exchange rates. As pharmaceutical companies search for cheaper imported alternatives, the concerns of domestic API firms are further compounded.
A pharmaceutical industry official stated, "If generic prices fall further, the movement to replace raw materials with cheaper alternatives to save costs will spread. As the dependency on imported APIs increases, domestic API companies find themselves in a position where they must worry about survival."
Critics pointed out the ineffectiveness of the government's API preferential pricing policy. The government plans to apply price preferences to listed "essential national medicines" that use domestic APIs. This involves granting a price preference of up to 68% of the pre-patent-expiry price of new drugs for essential medicines made with domestic raw materials.
However, pharmaceutical companies complain that the proportion of essential medicines within total drug sales is negligible, and even if prices are raised, there is insufficient incentive to switch to domestic APIs. To receive the preferential designation for domestic raw materials, a company must prove that all raw materials were synthesized at a domestic manufacturing site. Required documents include the ▲API Registration Certificate ▲Common Technical Document (CTD) ▲Manufacturing Instructions and Records.
During a parliamentary audit last October, Rep. Baek Jonghean of the People Power Party pointed out, "The fact that not a single pharmaceutical company has applied for the price preference for essential national medicines using domestic raw materials for seven months is proof that the system exists in name only," adding, "Despite industry-wide complaints that the application criteria are too stringent, the MOHW's failure to recognize that the regulations will undermine the policy's ability to foster the domestic API industry."
An industry source stated, "Due to the recent aftermath of the war in the Middle East, there are concerns over the supply instability of various raw materials, and with the added burden of costs from the high exchange rate, it is difficult to predict business plans for this year," adding, "In addition to the government's drug price reduction policy, it has become difficult to guarantee the business sustainability of both domestic finished drug and API manufacturers."
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.







