- LOGIN
- MemberShip
- 2026-06-11 00:26:35
- En masse designation of quasi-innovative pharmas?
- by Kim, Jin-Gu | translator Alice Kang | 2026-03-20 08:55:29
The government is reviewing a pricing incentive scheme for companies deemed equivalent to innovative pharmaceutical firms (“quasi-innovative” companies). Although more than 30 companies are expected to meet the criteria, the actual level of preferential treatment granted to the companies appears limited, leading to criticism that the policy is merely a publicity stunt.
According to industry sources on the 19th, the government is considering establishing a new preferential pricing track for “companies equivalent to innovative pharmaceutical firms (quasi-innovative firms).”
The government’s policy is to review price preferences to enable solid pharmaceutical companies with potential to advance to the level of innovative pharmaceutical firms at an early stage. These details were revealed during the Health Insurance Policy Deliberation Committee subcommittee meeting on the 11th.
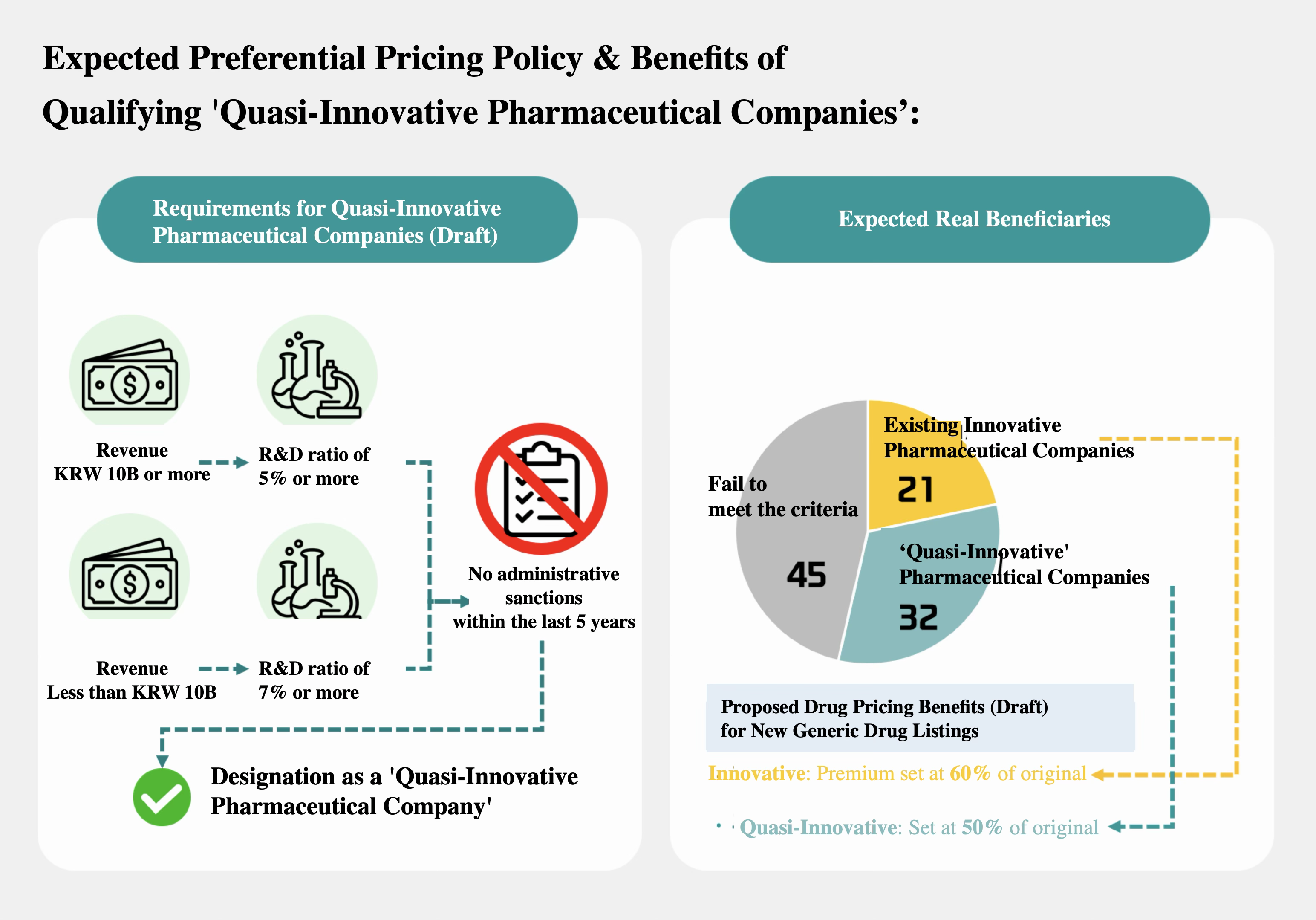
The government has proposed R&D investment ratios relative to revenue as the key criterion. Companies with revenue above KRW 100 billion must have an R&D ratio of at least 5%, and companies below KRW 100 billion must have at least 7%. Companies that have received administrative sanctions within the past 5 years under the Pharmaceutical Affairs Act, Fair Trade Act, or Pharmaceutical Industry Act due to rebate-related issues will be excluded.
Companies that meet these requirements will receive preferential drug pricing. Preferential pricing will be applied to newly listed generics, and the duration of this preferential treatment is currently under review to be similar to that for innovative pharmaceutical companies. The government also plans to grant temporary special exemptions when adjusting the prices of already listed drugs.
This plan is interpreted as a response to the pharmaceutical industry’s backlash against the drug pricing system reform draft released last November. At that time, the government proposed applying price discounts of 68%, 60%, and 55% to innovative pharmaceutical companies based on their R&D ratios. However, criticism arose that the benefits would be concentrated among a select few top companies, while most companies would find it difficult to avoid price reductions.
Applying the new criteria would significantly increase the number of companies eligible for preferential pricing.
An analysis of 98 listed pharmaceutical companies classified as pharmaceutical firms on the KOSPI and KOSDAQ that will be affected by price cuts found that 32 companies would newly qualify. This excludes holding companies, R&D-focused biotech ventures, and medical device firms that are not affected by price cuts.
Conversely, 45 companies failed to meet the criteria. The remaining 21 are companies that had previously been designated as innovative pharmaceutical companies.
Among companies with revenue above KRW 100 billion, those meeting the ≥5% R&D ratio include firms such as ▲CMG Pharmaceutical, J▲W Pharmaceutical, ▲SK Bioscience, ▲SK Biopharmaceuticals, ▲Kyungdong Pharm, ▲Kyungbo Pharm, ▲Daehan New Pharm, ▲Dong-A ST, ▲Myung-In Pharm, ▲Samjin Pharm, ▲Sam Chung Dang Pharm, ▲Ahn-Gook Pharmaceutical, ▲Withus Pharmaceutical, ▲Korea United Pharm, ▲Il-Yang Pharm, ▲Jeil Pharmaceuticval, ▲Chong Kun Dang, ▲Kolon Life Science, ▲Hana Pharm, ▲ Hanall Biopharma, ▲Whanin Pharm, ▲Humedix, and ▲Huons. These companies will likely be included as quasi-innovative pharmaceutical companies.
On the other hand, companies such as ▲HLB Pharma, ▲Kwang Dong Pharmaceutical, ▲Kukjeon Pharmaceutical, ▲Kukje Pharm, ▲Dai Han Pharm, ▲Dongkook Life Science, ▲Myungmoon Pharm, ▲Binex, ▲Samsung Biologics, ▲Samil Pharm, ▲Celltrion Pharm, ▲Sinsin Pharmaceutical, ▲CTC Bio, ▲Arlico Pharm, ▲RP Bio, ▲Yungjin Pharm, ▲Yuyu Pharma, ▲Reyon Pharmacuetical, ▲ Jinyang Pharm, ▲Kips Pharma, ▲Theragen Etex, ▲Pharmgen Science, ▲Hawil Pharm, ▲Hugel have R&D rations belos 5% as of the end of last year or Q3 last year, and fail to meet the threshold. It will be unlikely for these companies to receive preferential treatment during price cuts.
Among companies with revenue below KRW 100 billion, relatively few meet the ≥7% R&D threshold, with only a handful such as ▲Samsung Pharmaceutical, ▲Sam-A Pharmaceutical, ▲Samyang Biopharm, ▲Cellbion, ▲Onconic Therapeutics, ▲ENCell, ▲GL PharmTech, and ▲Prestige Biologics.
In fact, within the same revenue bracket, many companies do not meet the criteria for quasi-innovative pharmaceutical firms. These include ▲JW Shinyak, ▲Kyungnam Pharm, ▲Korean Drug Pharm, ▲Dongsung Pharmaceutical, ▲DuChemBio, ▲Vaskan Bio Pharm, ▲Vivozon Pharmaceutical, ▲Samik Pharmaceutical, ▲Seoul Pharma, ▲Sinil Pharmacuetical, ▲Icure, ▲Aprogen Biologics, ▲L&C Bio, ▲Optus Pharmacuetical, ▲Ilsung IS, ▲Cho-A Pharm, ▲Telcon TF Pharmaceutical, ▲TDS Pharm, ▲BNC Korea, ▲ Union Korea Pharm, ▲ Korea Pharma, among others, which have R&D ratios below 7%, making it highly likely they will not receive preferential treatment in the event of drug price reductions.
Industry observers emphasize that the actual level of pricing incentives matters more than the number of eligible companies. Even if many firms qualify, limited incentives could render the policy merely symbolic.
In the revised reform plan, the government proposed a 60% pricing premium for innovative pharmaceutical companies when listing new generics, with the preferential period extended to up to 4 years. For quasi-innovative companies, a lower level of benefit is expected.
It is reported that the government is considering a preferential level of around 50% for quasi-innovative companies. Given that generic prices are currently set at around 58.55%, this implies that price reductions would still be unavoidable even with preferential treatment. The relatively short preferential period of 1+3 years is also criticized as insufficient to provide meaningful incentives.
An industry official commented, “The introduction of criteria for quasi-innovative pharmaceutical companies and pricing incentives is positive in itself. However, in reality, it is more of a structure that reduces the extent of price cuts and is far from being a true incentive. It is a scheme designed to include a large number of companies for the sake of appearances, while in reality, it forces them to lower drug prices.”
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.







