- LOGIN
- MemberShip
- 2026-06-11 00:26:15
- Medical device companies gather for KIMES2026
- by Hwang, byoung woo | translator Hong, Ji Yeon | 2026-03-20 08:55:00
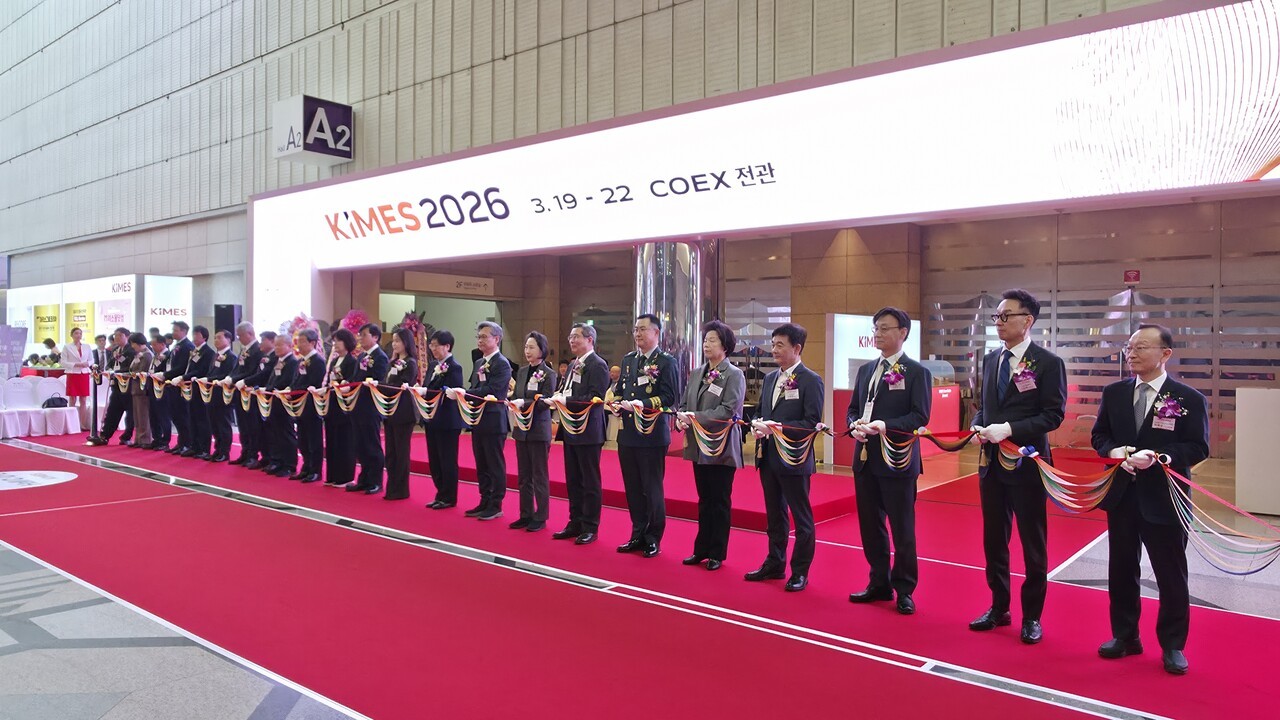
At the medical device exhibition, the primary topic has emerged from the 'whether to adopt Artificial Intelligence (AI)' to the question of 'how to implement it.'
At 'KIMES 2026,' which opened on the 19th, a clear shift in hospital views toward AI acceptance was observed, signaling a rapid migration of the industry's competitive axis.
KIMES, celebrating its 41st anniversary, is South Korea's largest medical device exhibition, reflecting global healthcare trends and encompassing the entire industrial ecosystem. This year's event featured 1,490 manufacturers from 41 countries, including 846 domestic and 644 international companies.
In this exhibition, major players in ultrasound, diagnostic imaging, endoscopy, and patient monitoring moved beyond standalone hardware to showcase integrated solutions linking diagnosis, treatment, and management. Notably, a 'platform war' is intensifying, with AI serving as the nexus connecting medical data and clinical workflows.
Beyond hardware to 'platform' competition…AI-Driven industrial restructuring
First, global medical device companies presented 'Life-cycle Patient Management' as their core strategy this year.
GE HealthCare Korea emphasized a structural continuum from diagnosis to treatment and monitoring by unveiling integrated solutions that combine ultrasound, MRI, and patient monitoring technologies. By presenting ultrasound for quantitative fatty liver analysis, AI-based MRI image reconstruction, and pain-response monitoring during surgery, the company showcased its direction toward data-driven precision medicine.
Samsung Medison also placed its platform strategy at the forefront by revealing its next-generation ultrasound brand, 'One Platform.' This structure ensures consistency across the entire examination process through a unified User Interface (UI) and automated features through AI to enhance diagnostic efficiency.
These industry leaders are moving past performance competition of individual devices to engage in a full-scale platform battle, one that connects diverse medical data points to facilitate clinical decision-making.
The direction of change was also distinct in the diagnostic imaging sector. While the transition from film-based X-rays to digital detectors is complete, advanced equipment such as CT and CBCT (Cone Beam Computed Tomography) is now becoming the standard for medical institutions.
Specifically in dentistry, CBCT has effectively become the standard, making 3D-based diagnostics a common practice.
Furthermore, the utility of portable X-rays and C-arm systems is expanding into operating rooms and emergency sites, evolving from simple diagnostic tools into procedural support equipment.
The most notable change is the improvement in diagnostic efficiency enabled by AI.
In the MRI field, there is a clear trend of using AI-based image reconstruction technology to shorten scan times. This is considered a critical factor that not only reduces patient wait times but also increases the number of scans, thereby directly impacting hospital operational efficiency and profitability.
An official from SG HealthCare stated, "Reducing MRI scan times is more than just a convenience. It is directly linked to hospital operational efficiency," and "With the introduction of AI, demand for simultaneously improving scan speed and quality is rising rapidly."

"The necessity of AI adoption has grown"…a shift in field
The most significant change from this exhibition is the shift in how hospitals perceive medical AI. Industry representatives on-site unanimously noted that the nature of visitor inquiries has changed.
In the past, questions primarily focused on hardware specifications or pricing. This year, however, there was a surge in inquiries regarding "whether AI features are applied," "where they are used in practice," and "what effects are observed."
Analysis suggests that interest in AI has moved beyond the exploratory phase to the actual implementation review stage. A consensus on the necessity of AI adoption is spreading quickly, particularly among large-scale tertiary hospitals.
Questions were raised regarding actual use cases and efficacy in various fields, including AI endoscopy, patient monitoring, and clinical support software, shifting the focus from technical validation to 'practical utility.'
However, the market is still in its early stages, with variations in adoption rates among institutions. While some hospitals show aggressive intent to adopt, many others maintain a cautious approach, weighing costs against clinical utility.
An official from an AI firm at the site commented, "The necessity for AI is becoming clear, starting with large hospitals. Once this trend solidifies, it is highly likely to influence small and medium-sized hospitals."

Platform competition intensifies…reshaping hospital operations
KIMES 2026 demonstrated that the medical device industry is transitioning from a hardware-centric industry to a 'data-driven platform industry.'
While performance and price were the core competitive factors in the past, ▲the new competitive axes include AI-based automation ▲data integration, workflow optimization ▲ensuring continuity of patient care.
InBody showed a trend of expanding into primary points of contact, such as pharmacies, by combining digital technology with its body composition analysis solutions. They introduced cases in which measurement data is analyzed in real time and results are presented through visualization, a structure that connects simple measurement to consultation and health management.

This is evaluated as an example of medical devices evolving beyond diagnostic tools into 'data-driven health management platforms.'
In particular, AI technology has become a key factor influencing not only diagnostic assistance but also the operational efficiency and revenue structures of hospitals and pharmacies.
The industry anticipates that future competition in the medical device market will likely be reorganized around 'platform units' rather than individual equipment.
An industry insider stated, "AI is not just a technology. It is an element that changes how hospitals operate," and concluded, "Corporate competitiveness will be determined by how effectively a company can connect and utilize data."
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.







