- LOGIN
- MemberShip
- 2026-05-16 19:14:52
- Company
- Imbruvica fails CDDC twice and reattempts 1st-line reimb.
- by Eo, Yun-Ho Apr 05, 2022 05:59am
- Once again, the blood cancer drug ‘Imbruvica’ is attempting to expand reimbursement to first-line treatment. According to industry sources, Janssen Korea has applied for the reimbursement extension of its Imbruvica (ibrutinib) as a first-line treatment for Chronic Lymphocytic Leukemia (CLL) and Small Lymphocytic Leukemia (SLL) to once again attempt at deliberations by the Health Insurance Review and Assessment Service’s Cancer Disease Deliberation Committee. Imbruvica’s first-line indication was unable to pass deliberation by the CDDC twice, one of which was held in October last year. The company was able to receive reimbursement extensions to the second line after being listed through the PE exemption pathway but is having trouble extending its indication further into the first line. Therefore, how Janssen will progress discussions on reimbursement based on what adjustments remain to be seen. Imbruvica is a first-in-class oral Bruton's Tyrosine Kinase (BTK) inhibitor that is taken once daily. With its oral formulation, the drug has the strength of being able to be administered in the outpatient setting. Since it was approved in April 2018, the drug is being used as ▲ a second-line treatment for adult patients with CLL, ▲monotherapy for the treatment of adult patients over the age of 65 with previously untreated CLL, ▲in combination with obinutuzumab for the treatment of adult CLL patients over the age of 65 or those who have comorbidities or at high-risk and those under the age of 65 with previously untreated CLL. Meanwhile, CLL is a type of blood cancer that occurs mainly in adults over the age of 60, characterized by increased production of mature but dysfunctional B lymphocytes Around 120 to 130 patients are newly diagnosed with CLL every year, and 60-70% of the CLL patients in Korea are discovered in a state that does not require treatment. Like its name, this “chronic” condition progresses slowly, sometimes insignificantly for several years.
- Opinion
- [Reporter's view] Commercialization of innovative new drugs
- by Lee, Tak-Sun Apr 05, 2022 05:58am
- #iLast month, immuno-cancer drug Keytruda was listed as the primary treatment for non-small cell lung cancer patients, and this month, the first chemical antigen receptor-T cell therapy (CAR-T) Kymirah received insurance benefits. Innovative treatments using patient immunity have succeeded in commercializing them in Korea one after another. The registration of the two drugs is significant. In terms of health insurance finance, the entry of ultra-high-priced drugs became a reality, leaving homework on rational financial management. From the perspective of medical staff and patients, the emergence of new paradigm drugs that go beyond the limits of existing treatments is raising expectations to increase the treatment effect. Immuno-cancer drugs such as Keytruda block the interaction of certain proteins that deactivate immune cells (T cells), making T cells more aware of cancer cells. CAR-T collects the patient's immune cells, mounts special receptors, puts them back into the patient's body, and finds and kills cancer cells that avoid attacks from T cells. While existing anticancer drugs directly attack cancer cells, there is a big difference in that immuno-cancer drugs or CAR-T treat cancer using the immune system. Moreover, it is in the spotlight as a next-generation drug that will lead chemotherapy in the future as it proves its high therapeutic effect in certain areas. Although next-generation drugs with such high therapeutic effects are expensive, it is encouraging that they have been commercialized in Korea. It is very unfortunate that the technology gap with the global market is gradually widening when looking back on the domestic pharmaceutical industry, which the state promotes as a future growth engine. Domestic pharmaceutical and bio companies are also developing immuno-cancer drugs and CAR-T, but they are still in the stage of seeking commercialization success. There is a big difference in level from Big Pharma, which has already completed commercialization and has grown into the highest-ranked drug. Contrary to investors' expectations, which are driven to pharmaceutical and bio stocks only by the possibility of development, it is necessary to calmly realize that the domestic pharmaceutical and bio industries are falling further behind. The new government also intends to actively invest in the pharmaceutical and bio industries. However, there seems to be no specific way to invest in an industry with any competitiveness. The current government and the new government also seem to focus solely on finding domestic pharmaceutical companies as a consignment production destination for overseas development COVID-19 vaccines, with biosimilars jumping into large companies. The government's diagnosis of the domestic pharmaceutical industry is to realize that it lacks thorough reflection than cheers and expectations for short-term performance. If there were mid- to long-term support for cell therapy when the world's first cell therapy was born in the early 2000s, wouldn't it have imitated CAR-T?
- Product
- Lagevrio prescriptions exceed 2,000… Paxlovid's up 25.6%
- by Kang, Shin-Kook Apr 05, 2022 05:58am
- The number of oral COVID-19 treatment Largevrio’s prescriptions has exceeded 2,000 in only 5 days since it was supplied to pharmacies in Korea. Also, the number of total Paxlovid prescriptions has exceeded 150,000 courses. According to the Korea Disease Control and Prevention Agency on the 1st, a total of 150,321 prescriptions were made from January 14th to the 31st of this month. This is a 25.6% increase from 118,738 prescriptions made the previous week (week of March 24th). By subjects of Paxlovid use, ▲121,676 were at-home treatment cases, ▲1,312 residential treatment center cases, ▲1,312 Infectious Disease Hospital cases, ▲1,052 others (military camp support, etc.) The more newly introduced Largevrio was prescribed 2,110 times. Among those, 1,835 were used at home, followed by 261 at Infectious Disease Hospitals and 14 at residential treatment centers. 100,969 courses of Paxlovid and 98,690 courses of Lagevrio remain in stock in Korea. The courses of Paxlovid left in stock in reach region are as follows: ▲Seoul 11,231 ▲Busan 3,987 ▲Daegu 2,542 ▲Incheon 3,685 ▲Gwangju 1,455 ▲Daejeon 1,271 ▲Ulsan 1,077 ▲Sejong 164 ▲Gyeongi-do 10,449 ▲Gangwon-do 2,679 ▲Chungcheongbuk-do 1,891 ▲Chungcheongnam-do 2,623 ▲Jelloabuk-do 1,998 ▲ Jelloanam-do 2,693 ▲Gyeongsangbuk-do 2,268 ▲Gyeongsangnam-do 3565 ▲Jeju-do 971 ▲Central 46,420 The courses of Lagevrio left in stock in each region are as follows: ▲Seoul 5,267 ▲Busan 1,779 ▲Daegu 874 ▲Incheon 1,552 ▲Gwangju 461 ▲Daejeon 693 ▲Ulsan 359 ▲Sejong 187 ▲Gyeongi-do 4,806 ▲Gangwon-do 1,230 ▲Chungcheongbuk-do 1,259 ▲Chungcheongnam-do 1,590 ▲Jelloabuk-do 755 ▲Jelloanam-do 1,276 ▲Gyeongsangbuk-do 1,089 ▲Gyeongsangnam-do 1,262 Jeju-do 345 ▲Central 73,906. Meanwhile, 460,000 courses among the 1,004,000 courses of oral COVID-19 drugs procured by the government will be introduced within this month. 184,000 courses have been already introduced to Korea in March, and the other 276,000 courses are to arrive within April.
- Policy
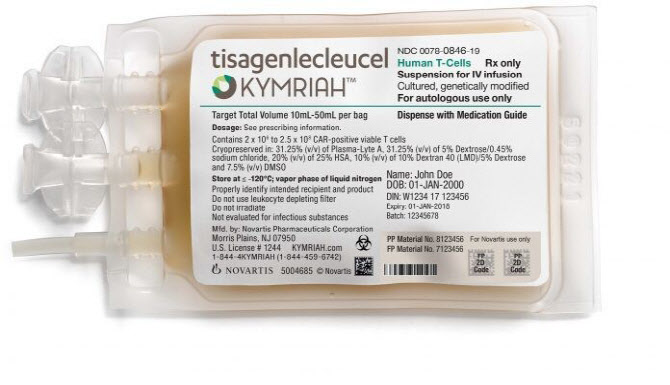
- Kymriah has been listed, hope of cure
- by Lee, Jeong-Hwan Apr 05, 2022 05:57am
- Patients immediately welcomed the confirmation of health insurance benefits of the acute lymphocytic leukemia and lymphoma CAR-T treatment Kymriah (Tisagencleucel). The patients urged the transition committee of the next presidential post and Yoon Seok-yeol to quickly register new drugs directly related to life as health insurance benefits. On the 31st, Korea Leukemia patient organization said, "The cost of one-time non-reimbursement payment is 460 million won, and the ultra-high-priced Kymriah passed the Health Policy Review Committee and acquired health insurance benefits to even hope for cure." Kymriah will be listed with an upper limit of 360 million won for one-time health insurance from the 1st. The Korea Leuchemia patient organization believes that the appeal of Cha Eun-chan's mother, Lee Bo-yeon, who left for heaven while preparing for Kymriah treatment, and the consent of tens of thousands of Cheongwadae petitioners influenced Kymriah's health insurance registration in a year and a month. The National Human Rights Commission of Korea expressed its opinion to the MOHW to establish a rapid registration system for Kymriah's health insurance system, which was raised by the Korea Leukemia patient organization, also positively affected benefits. The Korea Association of Patients urged the government to manage the risk sharing based on patient-level performance-based risk sharing that applies only to lymphoma, as well as Refund risk sharing and Extended Cap risk sharing, which are conditions for health insurance registration for Kymriah. The Association of Patients argued, "The next presidential transition committee and the Yoon Seok-yeol government should also introduce a rapid registration system for new drugs that show excellent effects without alternative drugs for life-threatening patients such as Kymriah."
- Company
- AZ starts next-gen ADC dev with confidence from Enhertu
- by Apr 04, 2022 06:07am
- AstraZeneca is leading the next-generation ADC development environment. The company, which has successfully commercialized ‘Enhertu’ in partnership with Daiichi Sankyo, is targeting areas slow in development such as triple-negative breast cancer. On the 31st, AstraZeneca received approval for its Phase III trial of a new ADC drug last month. The new ADC drug subject to the trial is ‘datopotamab deruxtecan (DS-1062, hereafter datopotamab),’ a TROP2 targeting ADC that is being co-developed by AstraZeneca and Daiichi Sankyo. The Phase III trial for datopotamab will be conducted on patients with unresectable locally advanced or metastatic triple-negative breast cancer. The trial will evaluate the drug’s efficacy as a first-line treatment in patients who are not eligible for treatment using PD-(L)1 immunotherapies in comparison to chemotherapy. MoA of datopotamab(Source: Daiichi Sankyo) ADC is a next-generation drug that is produced by coupling a potent cytotoxic agent to a monoclonal antibody that binds to a specific antigen on the surface of a tumor cell. It minimizes the side effects while increasing the treatment effect by selectively working on cancer cells. Although ‘Kadcyla’ marked the start of the commercialization of ADC drugs, the drugs were unable to make significant performance in the earlier stages due to technical limitations. Since then, the development of next-generation ADCs, such as linkers that control the drug-antibody ratio (DAR) or enhance blood stability, is being developed in earnest. AstraZeneca already has experience commercializing an HER-2 targeted ADC, ‘Enhertu’ after signing a joint development agreement with Daiichi Sankyo. Enhertu had demonstrated superior efficacy over the early-generation ADC drug ‘Kadcyla’ as a second-line treatment in HER2-positive breast cancer. According to results from the DESTINY-Breast03 that was presented last year, Enhertu reduced the risk of disease progression and deaths by 72% compared to Kadcyla in the head-to-head trial. Also, the objective response rate (ORR) was significantly higher, 80% in the Enhertu group as compared to 34% in the Kadcyla group. The new ADC therapy that will be presented by AstraZeneca will target the field of triple-negative breast cancer that has poor prognosis. In the Phase I trial, datopotamab showed positive results with an ORR of 43% and DCR of 95%. If significant results continue on in the follow-up trials, AstraZeneca’s new ADC will likely compete with Gilead’s ADC therapy. Like datopotamab, Gilead’s ADC drug ‘Trodelvy’ also targets TROP2. Trodelvy was approved by the US FDA as a treatment for triple-negative breast cancer last April. AstraZeneca paid Daiichi Sankyo $1 billion (₩1.22 trillion) in upfront payment for the development rights of datopotamab. AstraZeneca will pay additional conditional amounts up to $6 billion according to the company’s achievement of development and commercialization milestones. In the past, AstraZeneca had signed a $6.9 billion (₩8.41 trillion) agreement with Daiichi Sankyo for the development rights of Enhertu. The company is also investigating using datopotamab in combination with its immunotherapy ‘Imfinzi’ in addition to another trial that assesses the effect of datopotamab in NSCLC. In addition to the ADC technology the company had bought from other companies, AstraZeneca is also developing a next-generation ADC using its linker technology, 'AZD8205.’ 'AZD8205 is a new drug candidate that targets overexpression of B7-H4 found in various solid cancers. The company plans to first disclose study results for AZD8205 at the ‘AACR 2022’ that will be held from the 8th.
- Policy
- Phase 3 of Jetema the toxin will be conducted in Korea
- by Lee, Hye-Kyung Apr 04, 2022 06:07am
- Jetema the toxin 100U, which has been approved for export botulinum toxin, will conduct phase 3 clinical trials in Korea. The MFDS recently approved Jetema the toxin for a "multi-organization, double-blind, random assignment, parallel design, noninferiority trials, active control groups, and phase 3 clinical trials" for JTM201. The clinical trial will be conducted at Asan Medical Center in Seoul, Konkuk University Hospital, and Chung-Ang University Hospital. Jetema is conducting a clinical trial of Jetema the toxin after obtaining a drug manufacturing license in January 2019, and is expected to officially apply for an item license in Korea when the phase 3 clinical trial is completed. In June 2020, Jetema received permission from the MFDS to export Jetema the toxin for full-fledged overseas expansion and commercialization. It signed a local clinical and supply contract worth 144 billion won with Brazil even before applying for clinical trials and acquiring KGMP, and signed an exclusive sales and supply contract with HUADONG MEDICAL AESTHETICS as a Chinese market supply partner in February this year. Jetema aims to conduct phase 1 of Jetema the toxin in China by the end of this year along with phase 3 clinical trials in Korea. Since the beginning of this year, news of phase 3 clinical trials for botulinum toxin, a treatment for improving wrinkles in adults, has continued in Korea. On the 1st, the phase 3 clinical trial of Protox, a botulinum toxin A drug applied by the MFDS, was approved. Clinical trials of Protox are conducted at Chung-Ang University Hospital.
- Policy
- 20 ultra-high-priced drugs over ₩5 mil sold in Korea
- by Lee, Tak-Sun Apr 04, 2022 06:07am
- Survey results have shown that 20 high-priced drugs over ₩5 million are approved for reimbursement 10 years after, Soliris, which had been then the most expensive drug in the world, was listed for reimbursement in Korea With ‘Kymriah,’ the one-shot treatment that was listed for reimbursement on April 1st, recording the highest price at ₩360,030,000, the top 4 drugs with regards to their price were listed by the Moon’s administration. Looking at the price caps set in the reimbursement list, a total of 20 drugs were found to cost over ₩5 million. When lowering the standard to ₩10 million, the number of drugs increased to 84. The drug in 50th place is the PNH treatment Soliris which was listed 10 years ago in 2012. At the time, Soliris’s price was set at ₩5 million per year, sparking heated debate on the reimbursement of the drug. However, 49 more drugs with a higher price cap had been introduced to the market in 10 years. Of course, the price cap cannot determine the cost of each drug as their treatment period differs. For example, Kymriah is administered once in a lifetime, and in this sense, the drug’s ₩360,030,000 is cheaper than the ₩500,000,000 that was set as the annual cost for Soliris 10 years ago. By price cap alone, Kymriah is the only drug priced at the 100 million level. The second in line is the SMA treatment Spinraza, which costs ₩92,350,000. In third place is the neuroendocrine tumor treatment Lutathera set at ₩22,100,000, followed by the immunotherapy cancer drug Yervoy set at ₩14,000,000. Fourth is the stem cell therapy for Crohn’s Fistula, Cupistem at ₩13,490,000. What is unusual is that the top 4 most-expensive drugs on the list were approved for reimbursement under Moon’s administration. As new drugs with better efficacy in rare diseases are being introduced, the fact that the latest drugs have a higher price is, in a sense, natural. Spinraza was listed for reimbursement in April 2019, Yervoy in September last year, Lutathera from March this year, and Kymriah from April, after which the NHI will be supporting most of their costs. Such essential drugs that are ultra-high-priced will continue to be released in the future. The increased number of such drugs can also burden NHI finances, therefore, the government’s concern over effective fiscal sharing will also continue to deepen.
- Company
- Keytruda, the primary treatment for esophageal cancer
- by Apr 04, 2022 06:07am
- Keytruda, an immuno-cancer drug of MSD, has become the primary option in esophageal cancer, where treatments have been limited. The medical team predicted that an immuno-cancer drug-oriented treatment strategy using Keytruda or Opdivo will be established depending on the PD-L1 expression rate. At a seminar to commemorate the expansion of Keytruda indications held online by MSD Korea on the 31st, Sun Jong-moo, a professor of hematological oncology at Samsung Medical Center, pointed out the meaning of the launch of Keytruda in esophageal cancer. He said, "As immuno-cancer drugs appear in esophageal cancer, treatment strategies are changing in the direction of considering using immuno-cancer drugs from the earliest stage possible depending on the PD-L1 expression rate of patients." Professor Sun Jong-moo of the Dept. of Hematology at SMC, who is presenting at the MSD Online Seminar in KoreaOn the 7th, the MFDS expanded indications for Keytruda in combination with chemotherapy in metastatic esophageal cancer and gastroesophageal junction cancer, which cannot be operated. Keytruda is the first immuno-cancer drug to be released in the primary treatment. Keytruda targets patients with positive PD-L1 expression. Esophageal cancer is largely divided into squamous epithelial cell cancer and adenocarcinoma, of which squamous epithelial cell cancer accounts for 90%. According to Professor Sun, esophageal cancer is considered a very difficult cancer, and if surgery is impossible, it should be treated with chemotherapy. However, when chemotherapy is used, mOS is only about 10 months. Although treatments have developed dramatically in various cancers over the past decade, esophageal cancer has continued for nearly 40 years. Keytruda significantly improved the therapeutic effect through a study on KEYNOTE-590, a licensed clinical trial. The mOS of Keytruda+anti-cancer chemotherapy group was 13.5 months, which was significantly longer than the control group (anti-cancer chemotherapy alone) of 5.5 months. Keytruda reduced the risk of death by 38%. mPFS also reduced the risk of disease progression or death by 49% compared to 5.5 months in the control group to 7.5 months in the Keytruda group. Keytruda previously put forward a distinctly different strategy from BMS's immuno-cancer drug Opdivo, which entered esophageal cancer. Opdivo can be used regardless of the PD-L1 expression rate, but can be used as a secondary treatment in patients who first used chemotherapy. Conversely, Keytruda was limited to PD-L1 positive patients, but acquired the status of primary treatment. Professor Sun said, "The current treatment that can be used in the primary esophageal cancer is a drug that has been used since the early 80s, and the demand for unmet was high." Professor Sun said, "Now that immuno-cancer drugs can be used in the first round, the treatment effect is expected to increase significantly," adding, "Medical staff also experienced that adding immuno-cancer drugs to chemotherapy does not significantly increase side effects, and the experience has been proven by clinical data."
- Policy
- Based on PVA exclusion, the arithmetic average is 90%
- by Lee, Tak-Sun Apr 04, 2022 06:07am
- Drugs subject to PVA with an arithmetic average of less than 90% of the same product group are excluded. Previously, only drugs below the arithmetic average were excluded, but the target was further narrowed to less than 90%. Products with annual claims of less than 2 billion won are also excluded from PVA drugs. Previously, products worth less than 1.5 billion won were excluded, but the revision will expand the drugs subject to exclusion. The NHIS announced on the 28th that it will revise detailed operating guidelines for PVA negotiations to enhance the role of drug expenditure management and promote efficiency in operating the system. The NHIS explained that the PVA negotiation system is a system that adjusts drug prices as the usage increases after listing drugs, and plays a key role in the follow-up management of drug prices. The need to improve effectiveness has been steadily raised, and the guidelines have been revised this time. The revision of the guidelines focused on revising the drugs subject to negotiation (Article 6 of the Guidelines) for efficient operation of the system and financial management. First, in order to select the top drug of the amount of claims excluded due to reasons below the arithmetic average, the rule excluding "less than the arithmetic average" was revised to the "less than 90% arithmetic average" rule. As of 2020, the average claim for drugs subject to PVA negotiations in 2021 was 12.7 billion won, while those subject to exclusion below the arithmetic average were 22.3 billion won, much more than this. Most of the drugs subject to exclusion below the arithmetic mean were between 90% and 100% of the arithmetic mean. An official from the NHIS explained, "In the past, it was difficult to efficiently operate the system by excluding drugs below the arithmetic average price from the PVA negotiations." The revision will revise the existing regulations to less than 2 billion won in order to exclude small claims with low fiscal impact from negotiations. Among the subjects of the 2021 negotiations, drugs in the section between 1.5 billion won and 2 billion won in claims account for 35.6% of the total. Jeong Hae-min, head of the NHIS' Pharmaceutical Management Office, explained, "The revision of the detailed operation guidelines for PVA negotiations will strengthen the follow-up management of drugs that affect insurance finances by strengthening drug management." The revised detailed operation guidelines for PVA negotiations will be implemented from April 1, and they will also be applied to drugs undergoing PVA monitoring and negotiations at the time of implementation of the guidelines.
- Policy
- To commercialize innovative new drugs/supply essential drugs
- by Lee, Jeong-Hwan Apr 03, 2022 04:25pm
- The MFDS reports discuss ways to become a bio-health powerhouse The Presidential Acquisition Committee Yoon Seok-yeol and the MFDS agreed to systematically support the commercialization of high-tech and innovative medical products and establish a stable supply environment for rare essential drugs with low profitability. The transition committee plans to take the lead in the development of domestic treatments for COVID-19 to establish sovereignty over vaccines and treatments, a major pledge of Yoon Seok-yeol. On the 28th, Yoon Seok-yeol's transition committee's social welfare and culture division made the announcement after reporting to the MFDS. Lim Yi-ja, secretary-general of the MFDS, as well as standing expert committee members Kim Mi-ae and Seo Jung-sook of Ahn Sang-hoon, Baek Kyung-ran, and Kim Do-sik were present in the report. From the MFDS, deputy director Kim Jin-seok, evaluation director Seo Kyung-won, and directors attended. The development of domestic treatment for COVID-19 is a major pledge related to COVID-19 by President-elect Yoon Seok-yeol. Yoon promised to expand full national R&D support to establish sovereignty over vaccines and treatments and build a global vaccine hub. It will also come up with policies to open Yoon's pledge, and the MFDS emphasized the importance of ▲preemptive preparation of predictable screening criteria,▲ systematic commercialization support for advanced and innovative medical products, ▲ training of human resources with global level of regulatory response capabilities, and▲ the role of the MFDS to leap forward as a bio-health powerhouse, such as strengthening international cooperation to secure global competitiveness. Discussions on preparation for the outbreak of new infectious diseases were also held. The acquisition committee and the MFDS agreed that rare and essential medical products, which are difficult to supply to the market due to lack of profitability, should play their role in the country. The transition committee said, "We need to discuss with various experts and closely cooperate with related ministries in the stage of reviewing the safety and effectiveness of medical products." The transition committee said, "We should respond quickly with forward-looking judgments in crisis situations such as COVID-19. The MFDS should make efforts to ensure that domestic medical products have international competitiveness."