- LOGIN
- MemberShip
- 2026-05-15 22:34:47
- Policy
- ↑Rosuvastatin when combined with Brilinta/Rosuvastatin
- by Lee, Hye-Kyung Jul 11, 2022 06:04am
- According to a recent review of EMA safety information, the MFDS decided to establish a new clause on the interaction that combination of Ticagrelor and Rosuvastatin can affect kidney excretion and increase the risk of Rosuvastatin accumulation. According to the new clause, the exact mechanism of action for side effects was not known, but combination of Ticagrelor and Rosuvastatin resulted in decreased kidney function, increased CPK levels, and rhabdomyolysis. Ticagrelor, including Brilinta, is an ingredient that has been approved as a Ticagrelor in Korea. There are 76 items including 60 mg inno.N, Ahngook Ticagrelor, Brigrelor, Boryung Ticagrelor, Ticavix, Tirelor, Ticagrin, Hurorinta, Ticagen, and Brelor. The full-fledged launch is November, when Brilinta's material patent expires, and 25 items, including Boryung Ticagreler, have acquired generic for exclusivity from November 21 this year to August 20 next year. Brilinta, a representative item of Ticagrelor, was licensed for a new drug in 2011 to reduce the incidence of thrombotic cardiovascular events. The MFDS plans to change the permission after conducting an opinion inquiry by the 22nd.
- Company
- 4th JAKi Cibinqo to be prescribed in general hospitals
- by Eo, Yun-Ho Jul 11, 2022 06:03am
- The fourth JAK inhibitor ‘Cibinqo ‘ has landed at general hospitals in Korea. According to industry sources, Pfizer Korea’s new Janus kinases (JAK) inhibitor ‘Cibinqo (abrocitinib)’ passed the drug committees (DCs) of various medical institutions in Korea, including the Seoul National University Hospital. The drug, which is approved for atopic dermatitis, is Pfizer’s follow-up of ‘Xeljanz (tofacitinib)’ that is set to compete with ‘’ Lilly Korea’s ‘Olumiant (baricitinib),’ ‘Abbvie’s ‘Rinvoq (upadacitinib)' for the same indication. Olumiant and Rinvoq were approved for reimbursement in May this year, and the reimbursement listing process for Cibinqo is also in progress. Cibinqo received marketing authorization in November last year when the reimbursement listing for Olumiant and Rinvoq was under review. As a JAK1 inhibitor, the drug modulates multiple cytokines involved in the pathophysiology of atopic dermatitis, including interleukin IL-4, IL-13, IL-31, IL-22, and thymic stromal lymphopoietin (TSLP). It is indicated for the treatment of adults and adolescents over the age of 12 with moderate-to-severe atopic dermatitis. Cibinqo demonstrated its efficacy through the Phase III trials JADE MONO-1, MONO-2, COMPARE, etc. The drug reduced the Eczema Area and Severity Index (EASI) by over 70% at Week 12 and demonstrated an improvement in itch relief 2 weeks after initiating treatment. Its pivotal study, JADE Mono-1, was designed to evaluate the efficacy and safety of two doses (100 mg and 200 mg once daily) of Cibinqo monotherapy in patients 12 years of age and older with moderate-to-severe AD for 12 weeks. Results showed that 63% of the Cibinqo 200 mg administered group had achieved an EASI-75 (improvement of at least 75% in lesion extent and severity) at Week 12, which was a significant improvement compared to the 12% in the placebo group. Also, the rate of patients that achieved EASI-90 at week 12 had been 39% in the Cibinqo group and was higher than the 5% in the placebo group.
- Policy
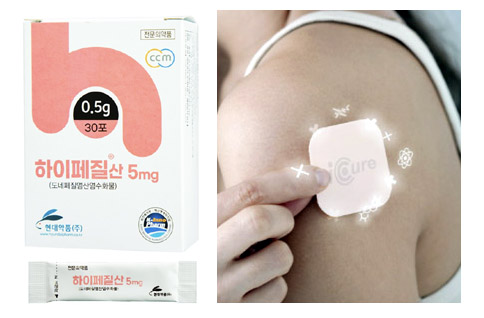
- New Donepezil & a dilemma over low drug prices
- by Lee, Tak-Sun Jul 11, 2022 06:03am
- Hypezil Powder 5mg (left) and IcureDomestic pharmaceutical companies are releasing a new form of Donepezil, a component of dementia treatment, but existing low-cost drugs are acting as a dilemma. Donepezil accounts for about 80% of dementia treatments, so competition among the same products is fierce. As there are 150 pharmaceutical companies with the same ingredient products, the drug price is breaking the lowest price every time. According to the industry on the 7th, Hyundai's Donepezil, Hypezil Powder 5mg, has been applied to the market since this month. This product is expected to be useful for patients who have difficulty swallowing existing tablets because it can be taken in water. The upper limit is 1,700 won. The MFDS recognized this product as a new formulation and approved it as a drug that submitted data. Therefore, if the drug price is calculated based on the standard for calculating the drug price, there is a possibility that the upper limit amount will be determined at 90% of the highest price of the existing product with the same ingredient. As the highest price of Donepezil 5mg is 2,060 won, 90% of this price is 1,854 won. However, Hyundai voluntarily lowered the price from the calculation standard and registered it as 1,700 won. In fact, 1700 won is also a high price based on the Donpezil 5mg. Because the current lowest price is 500 won. As 127 pharmaceutical companies compete for Donepezil 5mg alone, the lowest price is breaking every time a generic comes out. Therefore, it is analyzed that Hyundai, which is releasing new products, had no choice but to take into account the price competitiveness of the Donepezil market. Among the new Donpezil formulations, the patch system is expected to be released as early as the next day. This is because Donerion Patch (Celltrion) was conditionally passed by the Committee of the HIRA in May, are at the end of drug price negotiations. Since Daewoong Pharmaceutical's Fexuclu, which passed the committee's review on the same day, released this month, Celltrion and Icure's Donpezil defeat are also expected to be listed soon. The Committee judged that the benefit was appropriate when accepting less than the evaluation amount, but the industry believes that the committee calculated the drug price lower than the pharmaceutical company's expectations. Developer Icure was also initially in a position to release the drug at a lower price than the original drug Aricept, but it is said that a lower drug price was suggested. The industry analyzes that if alternative drugs were included, they would fall short of the company's expectations. For example, the average price of the original and generic 10mg is 2,040 won, which is cheaper than the original Aricept 10mg (2,265 won) as well as the developer Icure's Penedo 10mg (2,460 won). Considering the compliance of dementia patients, it is analyzed that the use of patch drugs is better, so even if the drug price is low, it can record high performance due to its high usage. The market seems to be at a disadvantage for product developers because Donepezil prices are too low.
- Company
- ER contraceptive market battles over original & generics
- by Nho, Byung Chul Jul 11, 2022 06:03am
- Norevo (Levonorostrel 0.75mg) and Elaone (Ulipristal Acetate 30mg), which Hyundai Pharmaceutical received import licenses from HRA Pharma in 2001 and 2011, by a 10-year gap, are emerging as icons of changes in the emergency contraceptive market. Emergency contraceptives have been submitted to the Central Pharmaceutical Review Committee of the MFDS and classified and maintained as Rx drugs during the reclassification process of Rx drugs vs OTC. In 2014, seven years after Norevo, the first product of this series, was approved, the emergency contraceptive market showed box-office performance after forming about 8.7 billion won in appearance. In the past five years, the CAGR has been showing a gradual decline, with -2.6% based on market value and -4.9% based on prescription quantity. It is observed that the market of Levonorgestrel, where each brand's products were licensed and generic appeared less than a year after patents were not protected, has been bisected by the launch of Ulipristal in 2011. The share of Ulipristal products, which was 21% in 2013, the third year of its launch, has remained at 45% until last year since entering the 40% range in 2016. Ulipristal preparations have the advantage of extending the emergency contraceptive effect from 72 hours (3 days) to 120 hours (5 days) after the relationship. As mentioned above, the size of the oral emergency contraceptive market itself is stagnant, but new changes are also expected under the surface. Pharmaceutical companies that have oral hormone preparation facilities include Myungmoon Pharm, Dalim Biotech, and GL Pharma. GL Pharma, which entered the market last, is expanding its market share to 28% by manufacturing a total of six items, including its own product, Sexcon One. Hyundai Pharmaceutical's 5-year CAGR of Norevo one was -6.5%, and GI Medics' Phostino-1 sold by Bayer Korea had a CAGR of -20.4% during the same period, and its market share also decreased by more than half from 22% to 10%. Given this situation, the challenge of emergency contraception by Ulipristal's first generic, Ellaoil (authorized on June 13, 2022/GL Pharma), is drawing attention from the industry. Attention is being paid to THEU's move, which quickly rose to second place (23%) in market share from Levonorgestrel. The price change due to the shift in market flow from monopoly to oligopoly is also another point to watch. Four first generics companies, including GL Pharma, are competing with HRA Pharma, a French emergency contraceptive developer, to determine the scope of rights and to sue for patent infringement.
- Company
- The pharmaceutical union of 2,500 people has been launched
- by Jul 11, 2022 06:03am
- The first chairman is Ahn Deok-hwan, head of the Korea Novartis union. Fifteen multinational companies and one domestic company participated in the NPU launch ceremony after three years of discussion "Working conditions are deteriorating despite increased sales in the pharmaceutical industry. Active response to common issues" The nation's largest pharmaceutical labor union has been newly created. The new labor union, which consists of a total of 2,500 pharmaceutical workers, expressed its willingness to jointly respond to the pharmaceutical company's restructuring moves. The NPU held a launching ceremony at the FKTU on the morning of the 5th. Kim Dong-myung, chairman of the FKTU, Hwang In-seok, chairman of the FKTU, Kim Ki-chul, chairman of FKTU's Seoul regional headquarters, Nam Sung-hee, chairman of FKCU's pharmaceutical cosmetics division, and Yoo Byung-jae, CEO of Novartis Korea, attended the meeting. A total of 16 domestic and foreign pharmaceutical labor unions, including ▲Novonodisc Pharmaceutical, ▲Bayer Korea, ▲Amgen Korea, ▲Ipsen Korea, ▲Novatis Korea, ▲Viatris Korea, ▲ Janssen Korea, ▲ Pfizer Pharmaceutical, ▲ Sanofi Aventis Korea, ▲Alvogen Korea, ▲Opella Healthcare Korea, ▲Merck, ▲Beringer Ingelheim, ▲AstraZeneca, ▲GSK, and ▲Hyundai Pharmaceutical. Eight unions, including Novonodisc Pharmaceutical and Pfizer Pharmaceutical Korea, have already decided to switch to industrial unions. Eight unions, including the AstraZeneca Korea Union, are preparing to switch industries. The total number of union members was about 2,500. Those who have been preparing for a new union of pharmaceutical unions for about two years since 2019 formed an integrated promotion committee for some unions and non-affiliated unions that left the KDPU, and officially established the NPU last year. In a declaration of launch, Ahn Deok-hwan, the first chairman of Novartis Korea, said, "The Korean pharmaceutical industry is suffering from job insecurity and a real decrease in wage income through the fourth revolution and COVID-19." "I felt the need for solidarity as I recognized these problems as the same problems that appeared in the pharmaceutical industry as a whole, not in a specific company," he said. He then stressed, "The NPU will firmly maintain employment stability, raise wages and welfare, and respond intensively to common problems arising from low growth and non-face-to-face operations in the name of voluntary retirement." Yoo Byung-jae, CEO of pharmaceutical industry, said in a congratulatory speech, "Congratulations on the launch of NPU. The pharmaceutical bio industry is facing a reorganization of its organizational structure to improve innovation and productivity, he said. "I think trust between employees and companies is more important than anything else for companies to grow continuously and stably." There may be conflicts of interest between each subject, but we hope to establish a constructive relationship with the NPU," he said. The NPU is closely watching the reduction of personnel due to the recent reorganization at the global level. For example, Pfizer, which is in the process of reorganizing its sales department, is likely to implement additional Voluntary Early Retirement Programs. In addition, Novartis recently announced plans to reduce the number of people worldwide by integrating the organization at a global level. In addition to the overlapping number of people due to department integration, additional personnel reduction can be made in Korea. Chairman Ahn said, "The Voluntary Early Retirement Programs have been doing ERP due to a lack of trust that the union can protect itself, even though the subject has to be a worker. The NPU will try to gain confidence in its members that it can protect them."
- Policy
- Hemophilia Type A tx, Bayer's Jivi has been approved
- by Lee, Hye-Kyung Jul 10, 2022 03:35pm
- Bayer's hemophilia type A treatment Jivi has been approved for domestic items. The MFDS recently granted Jivi to routine prophylaxis for on-demand and suppression of bleeding episodes in adults and adolescents (blood coagulation factor VIII birth deficiency) who have been treated before, management of bleeding before and after surgery, and reducing the frequency of bleeding episodes. Jivi obtained permission from the EU Commission in November of the same year after being approved by the U.S. FDA as a routine preventive treatment for youth and adult hemophilia patients aged 12 and older with a history of treatment in August 2018. Since t1/2 is maintained at 17.9 hours, the initial recommended dose is administered twice a week once every five days, and is administered every seven days or twice a week depending on the clinical characteristics of the patient. Depending on the bleeding episode, the frequency of administration can be further adjusted for each individual. The most common side effects were headaches, coughs, nausea, and fever. Hemophilia A treatments currently prescribed in Korea include Advate (Takeda), Novoseven (Novonordisc), GreenMono (GC Pharma), Xyntha Solofuse (Pfizer), Adynovate (Takeda), Greengene F (GC Pharma), Faiva (Korean Red Cross), There are about 10 products including Kogenate FS (Bayer), Immune (Korean Red Cross), Elocate (Sanopy), Hemlibra (JW Pharmaceutical), Kogenate FS (Bayer), Immunate (Korean Red Cross), Eloctate (Sanofi), and Hemlibra (JW Pharmaceutical). Hemophilia is largely divided into hemophilia A ( factor Ⅷ deficiency), hemophilia B ( factor Ⅸ deficiency), hemophilia C (factor XI deficiency), parahemophilia ( factor V deficiency), pseudohemophilia or von Wilbrand factor deficiency. The frequency of hemophilia A patients is about 1 out of every 5,000 to 10,000 normal born boys, and hemophilia B is about one-fifth of hemophilia A.I n the case of von Willebrand Disease, it occurs in both men and women, and it is about one in 1,000 people. It is estimated that there are 600,000 hemophilia patients around the world, and about 12% of all hemophilia patients are type B hemophilia. In Korea, hemophilia A has the largest number of patients with hemophilia at about 2,000. Hemophilia B patients are estimated to be 400 patients, von Wilbrand Disease patients are estimated to be 100 patients, and hemophilia C patients are estimated to be 20.
- Product
- Sell Saxenda directly after non-face-to-face treatment?
- by Jung, Heung-Jun Jul 10, 2022 03:31pm
- Can hospitals sell Saxenda, known as diet injections, directly to patients after non-face-to-face treatment? It is an expensive drug worth more than 100,000 won per unit and there is a high demand for purchase, so it is not prescribed for outpatients. The problem is that some hospitals sell and deliver directly to patients even in non-face-to-face treatment. On blogs, people can find reviews of prescriptions that seem to have received Saxenda directly from the hospital. Pharmacists have increased their outpatient prescriptions since the controversy over abuse of Saxenda in the past, and it is pointed out that it is inappropriate to use non-face-to-face treatment to administer inpatients Rx. Pharmacist A in Seoul said, "After the controversy over misuse a few years ago, more doctors prescribed outpatient Rx. Since it is a self-injection and the price is high, there may be some places that sell it directly, but that doesn't mean that it can be sold through non-face-to-face treatment, the pharmacist said. He then expressed concern, saying, "There are many people who visit non-face-to-face treatment to get diet pills, but they will only repeat the problem of misuse again." The MOHW also says that the principle of self-injection, such as Saxenda, is to prescribe outpatient Rx. It was stated that the temporary notice of non-face-to-face treatment only stipulated that drug delivery should be done through pharmacies. An official from the MOHW said, "Whether it is face-to-face or non-face-to-face, self-injection should be prescribed outpatient Rx unless injected within a medical institution. It cannot be seen as an exception to the division of medicine. In addition, the official said, "There is no content on the delivery of drugs by medical institutions even in the announcement of temporary permission for non-face-to-face treatment. We are making sure that medicine is delivered through pharmacies," he said. He added that the related internal review is in the final stage as there have been questions recently received by the MOHW related to outpatient Rx of Saxenda. Meanwhile, in 2020, Rep. Nam In-soon of the Democratic Party of Korea also argued for mandatory outpatient Rx, saying that the frequency of non-reimbursed self-injection systems such as Saxenda is high.
- Company
- Byung-Jae Yoo to head integrated Novartis Korea
- by Eo, Yun-Ho Jul 08, 2022 06:25am
- General Manager Byung-Jae Yoo Byung-Jae Yoo (49) was appointed the head of the integrated Novartis Korea. Suhee Shin, General Manager of the Oncology Business Unit at Novartis submitted her resignation. According to industry sources, Novartis Korea has been integrating its Pharmaceuticals and Oncology Business Unit with the goal of completing the integration within the year. The company had been operating 2 business units independently under one name, but the two units have operated separate support departments, including marketing·sales as well as drug pricing·government relations·approval departments. In the integration process, the business units will be competing for the head position of each department. As Yoo was nominated as the head of the integrated business unit, how the structure of Novartis Korea would change remains to be seen. Novartis’s Asia-Pacific Region also appointed the former head of its Pharmaceutical Business Unit as Head of the integrated Novartis Asia-Pacific. Meanwhile, Yoo joined J&J in 2006 after completing his MBA at Harvard Business School through the company’s International Recruitment & Development Program. Since then, Yoo had held various roles in the company, including at North America’s Endovascular team, DePuy UK, and DePuy Australia. In Korea, he served at North Asia’s Cardiovascular Care Business Unit and was appointed the Managing Director of Johnson & Johnson North Asia to oversee Korea, Taiwan, and Hong Kong in 2017. In October last year, Yoo was appointed General Manager of Novartis Korea.
- Opinion
- [Reporter’s View] COVID-19 trials still in smooth progress?
- by Kim, Jin-Gu Jul 08, 2022 06:25am
- COVID-19 developers have been announcing that they will discontinue their clinical trials one after another. In just 10 days, Celltrion, Chong Kun Dang, and CrystalGenomics publicly announced that they will discontinue their COVID-19 clinical trials. When counting from January, the number increases to 5 with Genexine and HK Inno.N. The news of the discontinuation was received with the response that it was as expected. However, there is no reason for the companies to be criticized for discontinuing their clinical trials. Strictly speaking, only 10% of the drugs succeed in receiving final approval for their treatment or vaccine after initiating clinical trials. Rather, the companies can be regarded as being ‘honest,’ as they announced the discontinuation officially to their investors. If no official announcement is made on the discontinuation, there is no way for outsiders to know the progress made within a company. Even if a pharmaceutical company internally decides to abandon development, they can pretend that the trial is still ‘well in progress’ externally by not officially getting rid of their trials. There have been many cases where development has been silently scrapped this way. Pharmaceutical companies would sneakily announce their trial discontinuation after investors’ interest wanes. The same goes for COVID-19 trials. When pharmaceutical companies first rushed in to start clinical trials for COVID-19 treatments two years ago, there was skepticism on whether those were a ruse to ‘raise stock price.’ With this skepticism unresolved, many trials are still ‘well in progress.’ From the investor’s perspective, they can never know how many companies have been ‘serious’ in conducting their clinical trials. The Ministry of Food and Drug Safety discloses the progress of clinical trials as ▲ Completed ▲Recruiting ▲Completed Recruitment or ▲Terminated, but it is impossible to confirm how many patients were recruited after trial approval. In other words, there is no way to know how actively the company is recruiting patients for its trials. The clinical trial registry ClinicalTrials.gov operated by the US National Institutes of Health (NIH) offers a more detailed view on the progress of its clinical trials. The progress is categorized as ▲Not yet recruiting ▲Recruiting ▲Active, not recruiting)▲Suspended ▲Terminated ▲Completed ▲Withdrawn ▲Unknown. In particular, the ‘Active, not recruiting’ category deserves attention. This means the study has started, but its potential subjects have not been recruited or registered yet. It indirectly shows the company's degree of engagement in conducting its clinical trials. Starting next year, the Ministry of Food and Drug Safety plans to publicly disclose the results of the factual survey that was conducted to secure the transparency and reliability of clinical trials. However, the disclosed contents will not contain the progress made by each clinical trial. Therefore, referring to the US cases to secure transparency and trust in domestic clinical trials may also be a viable option.
- Company
- Dong-A ST prepares to make another leap with Actonel
- by Eo, Yun-Ho Jul 07, 2022 05:55am
- Dong-A ST is attempting to make another leap with ‘Actonel,’ a bisphosphonate drug that is now sold exclusively by the company. According to industry sources, Dong-A ST has been working to land Sanofi’s bisphosphonate osteoporosis treatment Actonel (risedronate) at major medical institutions in Korea after it started to exclusively supply the drug in October last year. As of now, the drug has passed the drug committees (DCs) of 64 medical institutions including the ‘Big 5 tertiary hospitals’ of Korea - the Seoul National University Hospital, Seoul Asan Medical Center, Sinchon Severance Hospital, etc. Actonel, which is one of the representative bisphosphonate bone resorption inhibitors, has ruled the ranks in the Korean osteoporosis treatment market after its release in 2003, but then lost its share to new drugs such as Amgen’s ‘Prolia (denosumab).’ Therefore, what performance Dong-A ST will make with Actonel remains to be seen. With additional landings made, Actonel’s various strengths such as the 150mg formulation have been newly or additionally listed at hospitals. Sanofi discontinued the supply of its 5mg and 35mg strengths last year, and Dong-A ST plans to attract prescriptions while stabilizing the supply. Actonel has demonstrated its efficacy by reducing the relative risk reduction (RRR) of osteoporotic fractures and the risk of vertebral, hip, and nonvertebral fractures over its competitors from six months after intake. Meanwhile, Actonel EC 35mg is an enteric-coated tablet that can be taken once a week regardless of meal intake to treat osteoporosis in postmenopausal women. Actonel 150mg is taken 30 minutes before or 2 hours after meals monthly to treat osteoporosis in postmenopausal women.