- LOGIN
- MemberShip
- 2026-06-10 18:16:24
- Policy
- INN prescribing bill may be resubmitted to April subcommittee
- by Lee, Jeong-Hwan Apr 06, 2026 03:51pm
- 소병훈 보건복지위원장Attention is mounting within the healthcare and pharmaceutical sectors on whether a bill mandating limited international nonproprietary name (INN) prescribing for essential medicines or drugs with unstable supply will be reviewed by the National Assembly’s Health and Welfare Committee’s legislative subcommittee in April.The bill was included on the agenda of the Health and Welfare Committee’s legislative subcommittee last March, but did not get a chance to be reviewed as it was pushed back by other bills.On the 3rd, lawmakers from the Democratic Party of Korea have emphasized the need to convene the subcommittee in April to discuss bills under their purview that were not reviewed last month.As it is highly unlikely that the subcommittee will convene in May, just one month before the June 3 local elections, the Democratic Party of Korea members of the Welfare Committee saw a strong need to expedite the review of bills this month.Currently, the Health and Welfare Committee is led by Rep. Byung-hoon So (Gwangju-gap, Gyeonggi Province; third-term), who took over as the new chair following the resignation of former Chair Rep. Ju-min Park, who stepped down to run in the Democratic Party’s primary for the Seoul mayoral race.Accordingly, Chair Byung-hoon So, Democratic Party Executive Secretary Soo-jin Lee, and People Power Party Executive Secretary Mi-ae Kim are expected to begin discussions on the schedule for the April subcommittee meeting.Once the subcommittee meeting is confirmed, the bill mandating the limited use of INN prescribing, which was not reviewed last month, is highly likely to be placed on the agenda.Last month, on the day of the subcommittee meeting, the Korean Medical Association (KMA) held a “Rally to Block INN Prescriptions” on the steps in front of the National Assembly Main Building to pressure the Health and Welfare Committee.At that time, KMA President Taek-woo Kim made it clear that the association would take strong collective action if the bill were to be formally introduced.KMA President Taek-woo Kim led the rally to block the INN prescribing bill on the steps in front of the National Assembly Main Building on the 11th of last month.In particular, President Kim maintains a hardline stance that the bill must not be tabled at the April subcommittee meeting. The KMA is currently using the threat of a general doctors’ rally as leverage to block the review of the limited mandatory INN prescribing bill.However, the Democratic Party argues that the INN prescribing bill is part of President Lee’s campaign pledge and a national policy agenda adopted after his inauguration. They maintain that legislation is necessary to improve public access to medicines.A committee official stated, “The Democratic Party is calling for the need to convene the April subcommittee, but whether it will actually take place has not yet been finalized. It will be determined soon following consultations between the ruling and opposition party floor leaders, taking into account the schedule for the June 3 local elections.”There are three bills related to limited INN prescribing currently pending in the National Assembly: an amendment to the Pharmaceutical Affairs Act proposed by Rep. Yoon Kim of the Democratic Party of Korea, and amendments to the Medical Service Act and Pharmaceutical Affairs Act proposed by Rep. Jong-tae Jang of the same party.
- Policy
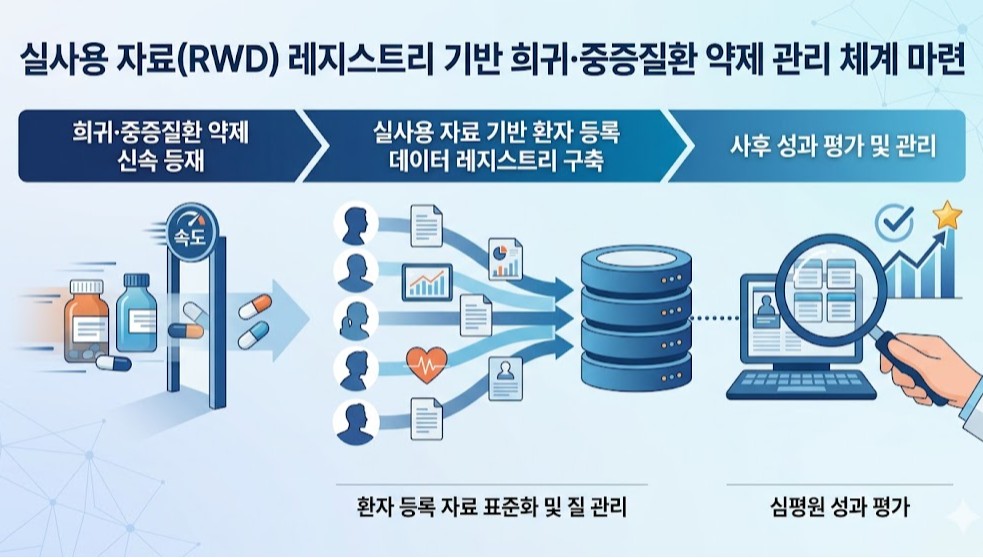
- Korea to use RWD for post-listing control of expedited drugs
- by Jung, Heung-Jun Apr 06, 2026 03:51pm
- A plan is being developed to utilize patient registry data, a subset of real-world data (RWD), to strengthen post-listing management of rare and severe disease treatments.This is an extension of the drug pricing system reform announced by the government last month through the National Health Insurance Policy Deliberation Committee. Previously, the government had revealed plans to expedite the listing of treatments for rare and severe diseases and then reevaluate their reimbursement status based on real-world data.According to industry sources on the 6th, HIRA and the Pharmacuetical Performance Assessment Department will conduct a study this year to establish an RWD registry-based management system. A registry refers to patient-level data collected by disease or drug.Although a call for proposals has not yet been issued, the research is scheduled to be completed by the end of this year.Following last year’s study on RWE (Real-World Evidence) guidelines for drug performance assessment, the government is now moving to establish a management system based on RWD registries.This research is significant as it moves beyond the conceptual definition stage towards building an actionable infrastructure. It also serves as a key follow-up measure for implementing the government’s drug pricing system reform.This is because a patient data management system must be established to implement measures such as performance-based reimbursement, adjustments to reimbursement scope, or drug price based on post-marketing evaluation results using real-world data.A HIRA official stated, “Building a registry is essential for managing rare disease treatments. We will conduct policy research on how to actually build such a system. I expect results to be available by the end of the year, following approximately 6 months of research.”The official further explained, “While this can be seen as an extension of the drug pricing system reform, there has long been a consensus that a registry is essential for establishing a management system for rare and severe disease drugs.”However, patient or disease-specific registration data in clinical settings have not yet been standardized, and concerns regarding proper quality control remain unresolved. These issues are expected to be key points of contention during the process of establishing a registry-based management system.An HIRA official stated, “We plan to commission the research soon. We ask researchers to show strong interest, given the high accessibility and utility of the registry data.”
- Policy
- Study to improve ICER threshold underway
- by Jung, Heung-Jun Apr 02, 2026 08:46am
- With the government announcing plans to raise the ICER threshold next year, detailed reform measures are expected to be prepared by October this year.For this, research aimed at ensuring fiscal sustainability while realigning cost-effectiveness evaluation criteria to become more realistic will be conducted over approximately 6 months.On the 31st, the Health Insurance Review and Assessment Service (HIRA) issued a request for proposal for a research project titled “Establishing rational cost-effectiveness evaluation criteria for listing new drugs under the national health insurance system.”This study was launched in response to criticism that the current ICER threshold fails to reflect ▲rising national income and inflation ▲demands for enhanced patient coverage ▲trends in innovative drug development.Through this research, a model for calculating the ICER threshold that incorporates socioeconomic factors such as income and inflation will be developed. A weighting system that takes disease severity, therapeutic benefits, and fiscal impact into account will also be designed.The study will also explore ways to systematize and categorize ICER thresholds and develop models for applying evaluation criteria to reimbursement decisions through pilot implementation.Additionally, the government will review the introduction of a system for periodically adjusting ICER thresholds by referencing major countries. It also plans to gather input from stakeholders, including the pharmaceutical industry, patient groups, and academia.HIRA plans to sign the research service agreement between March and April, conduct an interim review, and complete the study by October. The total budget for the project is KRW 60 million.Through this study, the agency expects to establish rational cost-effectiveness evaluation criteria, thereby improving patient access to treatment and incentivizing new drug development. It also aims to build trust among pharmaceutical companies, patients, and the government by establishing clear standards and procedures.Previously, the Ministry of Health and Welfare announced plans to raise the ICER threshold next year through reforms to the drug pricing system. It decided to introduce a weighting system to allow for the flexible application of ICER values. The ministry plans to conduct policy research this year and implement a rational plan in 2027 based on the results.The government is expected to finalize detailed implementation plans based on the research results, which are due around October, and proceed with full-scale adjustments to the ICER threshold sometime next year.
- Policy
- Hair loss drug finasteride, sexual dysfunction-linked suicidal ideation warning
- by Lee, Tak-Sun Apr 02, 2026 08:46am
- AI-generated imageThe labeling for finasteride 1mg tablets, used in the treatment of hair loss, will now include warnings regarding sexual dysfunction side effects.For dutasteride, another hair loss treatment, a new "General Precautions" section will be added to include information on conditions such as depression.The Ministry of Food and Drug Safety (MFDS) recently announced that it has prepared these proposed label changes based on European Medicines Agency (EMA)'s results of a safety information review and has requested feedback from companies by the 10th.According to the revised labeling, a new statement will be added to the warnings section for finasteride 1mg tablets as "Sexual dysfunction that may affect mood changes, including suicidal ideation, has been reported in some patients. Patients should be advised to seek medical consultation if sexual dysfunction occurs. Consideration should be given to whether treatment should be discontinued."While the existing warnings section already mentioned reports of depression, it did not include specific details such as suicidal ideation resulting from sexual dysfunction, as specified in the newly added text.For dutasteride formulations, information regarding 'mood changes and depression' was added to the "General Precautions" section, which is a lower-tier alert than a warning.The newly added information states: "Mood changes (including depressed mood, depression, and rarely, suicidal ideation) have been reported in patients treated with other oral 5-alpha reductase inhibitors. Patients should be advised to seek medical consultation if such symptoms occur."For finasteride 1mg tablets, sexual dysfunction-related adverse events such as decreased libido and erectile dysfunction have already been frequently reported. They are specified in the "Adverse Reactions" section of the labeling. However, details regarding suicidal ideation linked to sexual dysfunction were previously absent.The target products for this label change include 94 items of finasteride 1mg, including the original drug Propecia 1mg.Additionally, for dutasteride, 98 items are subjected to changes, including the original drug Avodart Soft Cap 0.5mg.Meanwhile, in May of last year, the EMA specified suicidal ideation as a new adverse effect in the product information for finasteride. The EMA stated, "Cases have been included where patients complained of suicidal ideation alongside sexual dysfunction such as depression, decreased libido, and erectile dysfunction."The EMA recommended adding a warning statement regarding mood changes and suicidal ideation to dutasteride, a 5-alpha reductase inhibitor of the same class, as a precautionary measure. The EMA stated that, for dutasteride, the evidence does not currently provide a clear causal relationship, unlike that of finasteride.
- Policy
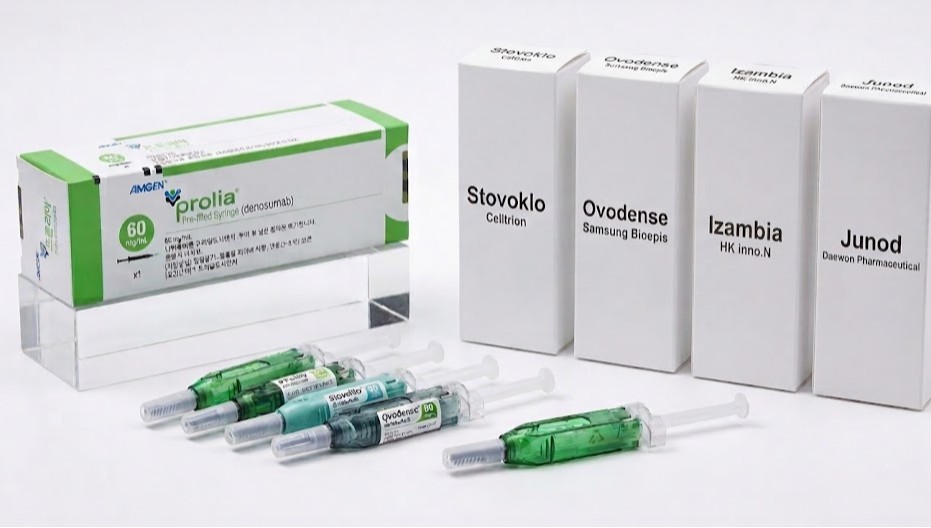
- Daewon Pharm joins the Prolia biosimilar competition
- by Lee, Tak-Sun Apr 01, 2026 08:15am
- AI-generated image (Prolia, Stoboclo, Obodence, Izambia, Junod)A series of biosimilars with the same active ingredient as Amgen’s osteoporosis drug Prolia are being approved. With Daewon Pharm joining the race, 5 products have now received marketing authorization in Korea.Prolia is an osteoporosis treatment administered as a subcutaneous injection once every 6 months, offering strong bone density improvement and fracture prevention effects.In Korea, it has dominated the market with annual sales of approximately KRW 180 billion through copromotion with Chong Kun Dang.On the 31st, the Ministry of Food and Drug Safety approved Daewon Pharma’s Prolia biosimilar, ‘Junod Prefilled Syringe.’This product is indicated for: ▲ the treatment of postmenopausal women with osteoporosis, ▲ the treatment of men with osteoporosis to increase bone density, ▲ the treatment of glucocorticoid-induced osteoporosis, ▲ the treatment of bone loss in patients with non-metastatic prostate cancer receiving androgen deprivation therapy, and ▲ the treatment of bone loss in women with breast cancer receiving adjuvant aromatase inhibitor therapy.It is administered via subcutaneous injection 6 six months in the upper arm, upper thigh, or abdomen, identical to Amgen’s Prolia. The product demonstrated equivalence to Prolia through Phase I and Phase III trials.Juno is a biosimilar developed by Gedeon Richter, headquartered in Budapest, Hungary.Gedeon Richter is a multinational pharmaceutical company with a global presence.In addition to Junod, Daewon Pharm has also introduced the Forteo biosimilar ‘Terrosa Inj’ through an agreement with Gedeon Richter. Terrosa was developed by Richter-Helm Biotec, a joint venture between Gedeon Richter and the German company Helm.With Daewon Pharm obtaining approval for Junod, there are now 5 Prolia biosimilars available in the Korean market.Celltrion’s ‘Stoboclo Prefilled Syringe’ was the first biosimilar approved in November 2024, followed by Samsung Bioepis’ ‘Obodence Prefilled Syringe,’ Meditip’s ‘Meditip Denosumab Prefilled Syringe,’ and HK inno.N’s ‘Izambia Prefilled Syringe,’ all of which have obtained marketing approval.Meditip Denosumab Prefilled Syringe was approved in Korea through a regulatory affairs agency, and Novartis has been identified as the contract manufacturer. There is a possibility that Novartis will handle domestic sales in the future.HK inno.N’s Izambia was developed by Spain-based mAbxience. HK inno.N has also signed a contract with mAbxience to introduce a nivolumab (brand name: Opdivo) biosimilar in addition to this denosumab biosimilar.Meanwhile, Celltrion’s Stoboclo is being co-marketed by Daewoong Pharmaceutical, and Samba’s Obodence is being co-marketed by Hanmi Pharmaceutical.The denosumab biosimilar market is rapidly becoming a battleground among major domestic pharmaceutical companies.Currently, Celltrion’s Stoboclo and Samsung Bioepis’ Obodence are listed for reimbursement. Full-scale competition is expected once the remaining approved products are also listed. All patents for Prolia listed with the MFDS expired as of March 17 last year.
- Policy
- Premiums for incrementally modified drugs remain uncertain
- by Jung, Heung-Jun Mar 31, 2026 08:45am
- As the government has finalized the overall framework of the drug pricing system reform, discussions are expected to move into detailed areas, including premiums for incrementally modified drugs.The core of this drug pricing system reform is the innovative level of companies. However, decisions regarding product-level premium measures, such as those for incrementally modified drugs, have been left out.According to industry sources on the 27th, since the premium rate for incrementally modified drugs was not clearly determined at this Health Insurance Policy Deliberation Committee meeting, follow-up discussions are expected to continue.The reform plan discussed at the Health Insurance Policy Deliberation Committee meeting last November had tentatively decided to maintain the current premium system for “incrementally modified drugs, incrementally modified combination drugs, and biosimilars.”However, this wording was deleted at the latest HIPDC meeting. With the addition of a new “quasi-innovative company” category, the drug price premium scheme has been significantly revised. The disclosed premium preferential measures consist solely of a 60% premium for innovative drugs, a 50% premium for quasi-innovative drugs, and preferential treatment for pharmaceutical companies and drugs that ensure supply stability.Under the current pricing calculation system, incrementally modified drugs receive premiums on top of the base price. For salt/formulation changes after original patent expiry, the price is set at 70% with a premium from the base price (53.55%). The price is set at 77% with a premium from the base price (58.9%). The premium is generally applied for 1 year after listing, but can be extended up to 3 years if there are three or fewer generic manufacturers listed for reimbursment.The key question is whether the government will maintain these premium rates. Since the base calculation rate has been lowered, there is a possibility that the premium rates will also be readjusted. If the government attempts to lower the premium rates for incrementally modified drugs in line with the reduced base rate, strong industry backlash is expected. The prevailing view in the industry is that if the government attempts to adjust the premium rate as well, it will dampen the motivation to develop incrementally modified drugs.There is also the possibility of reforming premiums for incrementally modified combination drugs. Currently, their price is calculated as the sum of 53.55% of each component drug’s pre-patent-expiry price. In this case, innovative pharmaceutical companies receive preferential treatment with a 68% sum, while general pharmaceutical companies receive a 59.5% sum.Given that drug price premium tiers are currently divided into innovative, quasi-innovative, and non-innovative categories, changes are needed in the calculation rates for incrementally modified drugs as well.In particular, since the “quasi-innovative” category is a newly established preferential pricing bracket, discussions must also address whether to apply differential premium rates within this category.Industry insiders agreed that, as the basic calculation rates and drug price premiums had not been finalized until now, concrete discussions must begin immediately.A pricing manager at a pharmaceutical company stated, “Until now, the focus was on establishing the overall framework, so detailed discussions were not possible. Now, discussions on incrementally modified drug premium rates must begin.”Another industry official added, “The content announced at this Health Insurance Review and Assessment Service (HIRA) meeting only covers the broad framework, so significant detailed adjustments are needed. The same applies to the premium rate for incrementally modified drugs. While each pharmaceutical company may focus on different aspects of the reform, since the base calculation rate has decreased, it is necessary to ensure that premiums are maintained as much as possible.”
- Policy
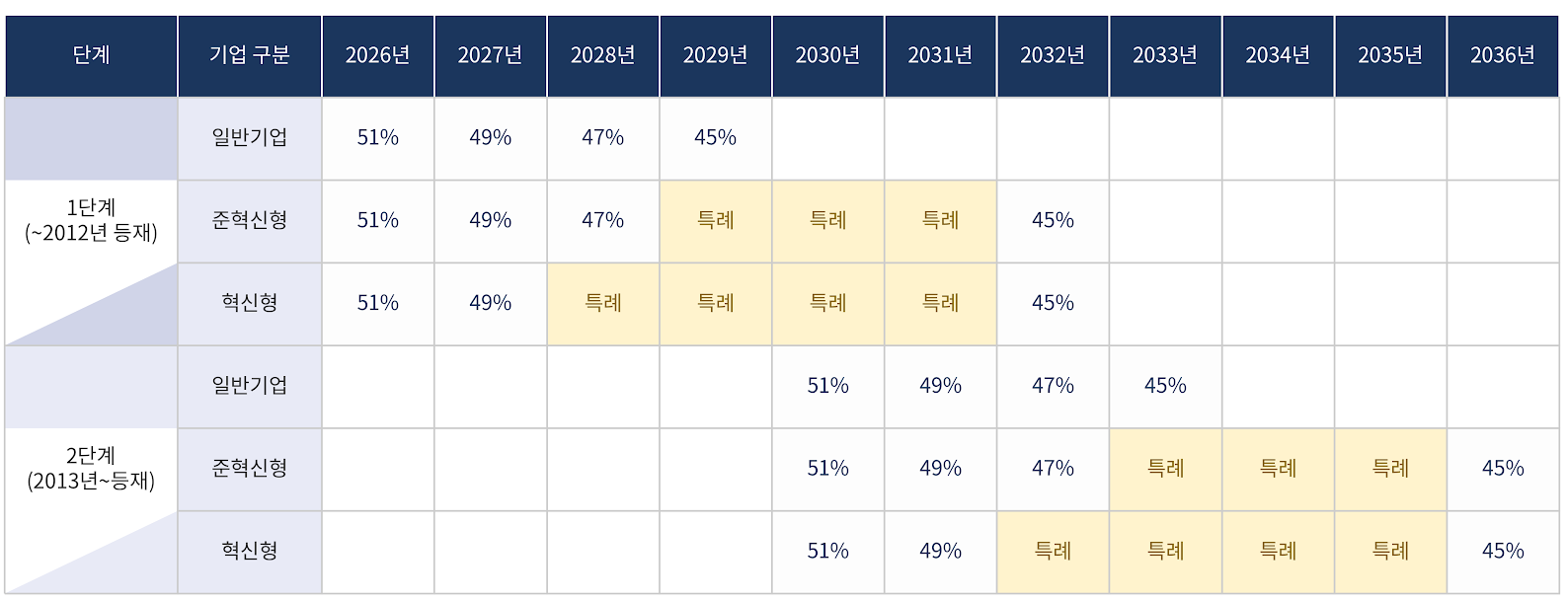
- Generic drug price for non-innovative firms set to 45% by 2029
- by Jung, Heung-Jun Mar 30, 2026 09:12am
- The government will implement price cuts for already-listed drugs, including those of non-innovative pharmaceutical companies, over a period of up to 10 years. With a gradual annual reduction of 2%, the drug price of innovative and quasi-innovative companies is expected to reach a rate of 45% by 2032, while non-innovative companies will reach the same level by 2029.Additionally, for products listed after 2013, gradual reductions will begin in 2030. Non-innovative companies will converge to 45% by 2033, while innovative and quasi-innovative companies will do so by 2036.The aim is to mitigate the shock of a sudden drop in sales for the pharmaceutical industry. Under this structure, if pharmaceutical companies fail to generate new revenue, the scale of their sales decline will gradually increase over the 10-year period.Price reductions for already-listed drugs will be implemented over a maximum of 10 years, including for non-innovative pharmaceutical companies.According to the government’s finalized plan announced on the 26th, the timing of reaching the 45% pricing rate will differ depending on ▲the listing date ▲whether special provisions apply.There are also conditional clauses for drugs with the same active ingredient, those failing to meet requirements, combination drugs, and data-submitted products. This means that even if a product was listed in 2013, it may still be included in the first-stage reduction.First, even general pharmaceutical companies that are not classified as innovative will undergo price cuts over 4 years. Prices will fall to 49% next year, then to 47% in 2028, and to 45% in 2029. Compared to innovative firms, they will reach the 45% level three years earlier.Innovative companies will be given a four-year grace period, and quasi-innovative companies a three-year grace period, reaching 45% by 2032. For innovative firms, the pricing rate will remain at 49% until 2027 and then drop directly to 45% in 2032.Drugs listed from 2013 onward will start at 51% in 2030 and decrease by 2% annually. General pharmaceutical companies will reach 45% in 2033, while innovative and non-innovative companies will similarly receive a 3- or 4-year grace period, with the 45% rate applying in 2036.A key feature is the classification of drugs listed by 2012 and those listed from 2013 into two stages (Stage 1 and Stage 2). However, drugs with the same active ingredient are classified into the same group based on the timing of the first generic entry.For example, even if a drug was listed in 2013, if the first generic for that active ingredient was listed in 2012, it falls under the first stage price reduction. ‘In addition, drugs that do not meet requirements, such as those lacking in-house bioequivalence testing or registered active pharmaceutical ingredients, will be subject to the 45% pricing rate. The same applies to combination drugs and data-submitted drugs that are essentially based on off-patent, generic substances.This means that even if a drug is a combination drug or a data-submission drug, if its active ingredients are equivalent to off-patent generics, it is subject to the 45% adjustment without exception.
- Policy
- Government busy cutting drug prices without volume control
- by Lee, Jeong-Hwan Mar 30, 2026 09:11am
- Health insurance drug expenditure is calculated by multiplying the reimbursement price by the prescription and utilization volume. No matter how much the government repeatedly lowers drug prices, it is difficult to achieve the policy goal of reducing pharmaceutical expenditure or rationally managing the health insurance budget unless physicians’ prescribing behavior and real-world usage are also controlled.In other words, even if the government lowers the generic pricing rate from 53.55% to 45% (a reduction of about 8.55 percentage points), failure to control prescription and utilization volume could paradoxically lead to higher drug spending than before the price cuts.If not handled carefully, this could lead to a worst-case scenario where the price cut creates an environment in which generics of lower quality than in the past are produced, while at the same time, some companies intensify their efforts to offer illegal rebates to increase prescription volumes in an attempt to mitigate the shock of price cuts.This is why, on the 27th, academia and the pharmaceutical industry urged the Ministry of Health and Welfare not to focus solely on the reform plan for generic drug price cuts, but to establish mechanisms to reasonably control prescription volumes.Academia “Government must lead generic competition through policies such as mandatory lowest-price substitution”Academia argues that attempting to reduce health insurance expenditure through price cuts alone without addressing volume control is fundamentally misguided.In particular, the prevailing view among academics is that while the Ministry’s efforts are needed to create an environment where pharmaceutical companies can compete to dominate the market through “low-cost generic” strategies, its repeated exclusion of this aspect is ultimately reinforcing an industry structure that leaves companies with no choice but to generate sales through illegal rebate competition.Criticism has been raised that the Ministry has virtually no plans for policies designed to enable low-cost generics to gain a competitive edge in the market, such as promoting the substitution of the lowest-priced generics, establishing a competitive landscape for generics based on international nonproprietary name prescriptions, or shifting the environment for generic selection through a Korean-style reference pricing system.Health economists emphasize that once bioequivalence is established, generics should be allowed to compete solely on price.Professor Hye-young Kwon of Mokwon University stated, “The conflict between the industry and the government over the generic drug pricing rate is meaningless. If drug prices fall, companies are more likely to increase prescription volume through rebates, which may maintain or even increase overall drug spending. Ultimately, cheaper generics must be prescribed more and sold more in order to achieve both health insurance savings and pharmaceutical industry development.”Professor Kwon added, "The Ministry of Health and Welfare must devise administrative measures to create a competitive market where pharmaceutical companies that lower prices the most for each ingredient dominate the market. A pharmaceutical company that wins on price in the domestic market has a good chance of succeeding in the global market as well. The government must work with doctors and patients to devise policies such as mandating the substitution of the lowest-priced generic, using the cheapest drug as a reference price, and providing incentives when that drug is used, or imposing disincentives when more expensive generics or originals are used.”She emphasized, “If we establish a generic price competition policy that goes beyond the government and pharmaceutical companies to include doctors, pharmacists, and patients, pharmacists and patients will be able to intervene in the decision-making process to ensure cheaper drugs are used, even if a doctor unilaterally prescribes a specific drug This would allow rationalization of health insurance finances without excluding physicians’ clinical judgment. The government needs to make a decisive move and establish the necessary framework.”Professor Seung-jin Bae of Ewha Womans University College of Pharmacy also believes that the Ministry of Health and Welfare has not given sufficient consideration to usage policies beyond generic price adjustment policies. In particular, the professor stated that administrative measures by the Ministry are needed to address the current reality where doctors are bound to brand-name prescriptions despite the government recognizing generics as therapeutically equivalent.Specifically, Bae suggested activating lowest-price substitution or implementing INN prescribing to enable genuine price competition among generics.Bae said, “We need policies that allow cheaper generics to expand market share. The issue of brand-name prescribing must be addressed first. It makes no sense for the government to acknowledge bioequivalence and still tolerate brand-name prescribing practices.”Bae emphasized, “We should not view this (the development of health insurance cost-saving policies) as a conflict between professions. Instead, we must consider measures such as substituting the lowest-priced generic to allow the market for chemical generics to be organized through price competition. Korea’s health insurance resources are not unlimited. In the case of biosimilars, there may be differing opinions regarding INN prescribing or substitution. However, for chemical drugs, we must take a more proactive approach from the perspective of health insurance finances.”The domestic pharmaceutical industry also maintains that, alongside price cuts, addressing physician prescribing behavior and patient overuse is essential to achieving effective cost savings. If the government pays attention to volume control, it could move away from its current administrative pattern of repeatedly relying on price cuts.An official from a leading domestic pharmaceutical company pointed out, “The government establishes policies claiming it will reduce drug costs, maintain generic drug quality, ensure a stable supply of medicines, develop innovative new drugs, and foster robust pharmaceutical companies, all within limited health insurance funds, but the conclusion always boils down to drug price cuts. It is time to consider the root causes of why we face criticism that medical institutions prescribe more drugs than necessary and why pharmaceutical companies are forced to boost sales through rebate competition.”“If we merely suppress drug prices while neglecting the increase in usage, we cannot achieve fiscal savings. If we only cut drug prices, companies will find it difficult to avoid decisions such as increasing prescriptions for their own drugs through aggressive marketing using CSOs, or producing and supplying low-quality generics by cutting costs and reducing employment.”Calls grow for governance involving government, industry, and academia in drug pricing policyWhenever drug price reduction reform plans are discussed, some in the pharmaceutical industry offer a self-deprecating assessment that “pharmaceutical companies are always in the position of the weakest party.”This reflects the reality that pharmaceutical companies find it difficult to openly and freely express their opinions or arguments, as they must constantly gauge the reactions of both the Ministry of Health and Welfare, which seeks to lower drug prices, and doctors, who hold the power to prescribe medications.Accordingly, the pharmaceutical industry is requesting that, starting with this drug pricing system reform proposal, a governance structure be established where government agencies, the pharmaceutical industry, and academia can discuss and design drug pricing policies together.The intent is to ensure that the pharmaceutical industry is granted the authority to participate in the decision-making structure for establishing drug pricing policies, including drug price reductions and revisions to post-approval drug price management systems, both administratively and legally.In the recent reform process, since the Ministry’s proposal was announced on November 28 last year, the pharmaceutical industry has been limited to merely proposing amendments and improvements to the Ministry’s draft, rather than actively voicing its own opinions, from the time the Ministry’s revised proposal was submitted to the subcommittee of the Health Insurance Policy Deliberation Committee on March 11 of this year until its final approval at the plenary session on the 26th.In particular, since the Ministry’s proposal was not made public until just before it was reported to the HIPDC, submitted to the subcommittee, and finally approved at the plenary session, pharmaceutical companies’ drug pricing officials had no choice but to work tirelessly trying to interpret the government’s intentions.In contrast, advanced countries overseas guarantee the pharmaceutical industry’s right to submit opinions on the direction, implementation timeline, and detailed regulations of drug pricing system reform proposals through mandatory agreements or legal commitments at the executive and industry levels.France operates under a framework agreement between CEPS and the pharmaceutical industry association, while the UK mandates stakeholder participation in pricing and reimbursement reforms through NHS England and the Department of Health.Japan also determines pricing through the Central Social Insurance Medical Council under the Ministry of Health, Labour and Welfare.An official from a mid-sized pharmaceutical company stated, “This drug price reform should serve as an opportunity to prepare various measures for establishing public-private consultative governance and to submit industry-level opinions to the government and the National Assembly. Even if we cannot exert a substantial influence on the establishment of the system, wouldn’t this create a forum to publicly convey the pharmaceutical industry’s well-founded opinions to the government, academia, and the public?”Academics have also suggested that improving the operational standards of the Health Insurance Policy Deliberation Committee (HIPDC), which is already run by the government, could ensure transparency in the establishment of the drug pricing system and the national health insurance system.Professor Bae said, “Regarding the governance of the drug pricing system and national health insurance policies, there is a need to ensure transparency by disclosing meeting minutes and other details discussed within the HIPDC. National health insurance finances and policies are funded using premiums paid by the public. Since the system is funded by public contributions, citizens have the right to know what is discussed during the Health Insurance Policy Deliberation Committee’s proceedings.”He added, “Ultimately, for everyone to accept the health insurance policies decided by the government, we must improve the process so that it is clear who expressed what opinions at the committee, rather than suddenly bringing items to a vote as is currently done. Creating additional consultative bodies outside the Health Insurance Policy Deliberation Committee framework risks creating unnecessary layers of bureaucracy.”
- Policy
- Will only trivalent vaccines be available this season?
- by Lee, Tak-Sun Mar 27, 2026 08:33am
- AI 이미지 생성 활용With the WHO recommending a full shift back to trivalent influenza vaccines, supply suspensions of quadrivalent vaccines are continuing one after another.There is even a possibility that quadrivalent vaccines may disappear from the domestic market in the 2026–2027 season. In fact, only trivalent vaccines have been included in the National Immunization Program (NIP) since last year.According to industry sources on the 25th, Boryung BioPharma recently reported to the MFDS that it would discontinue the supply of Boryung Flu V Tetra Vaccine.Boryung Flu V Tetra Vaccine is a quadrivalent vaccine approved in 2016, designed to protect against four influenza virus strains.The company explained, “As the WHO’s recommended influenza strain composition for the 2026–27 season does not include quadrivalent influenza vaccines, we have decided to discontinue production and supply of this product.”It added, “We plan to produce and supply trivalent influenza vaccines instead, and there is no risk of supply shortages resulting from the discontinuation of this product.”Boryung Biopharma had already discontinued another quadrivalent vaccine, Boryung Flu VIII Tetra, last year in line with WHO recommendations.On the 12th, Seqirus Korea likewise reported to the MFDS that it would discontinue supply of its Fluad Quad Prefilled Syringe. This is also a quadrivalent influenza vaccine, and the decision was made in accordance with WHO guidance.The company stated, “We plan to supply the trivalent version of this vaccine, ‘Fluad Prefilled Syringe ((influenza surface antigen MF59C.1 adjuvanted vaccine),’ in the domestic market.”CSL Korea, on the 19th, obtained approval for a trivalent influenza vaccine, Flucelvax Prefilled Syringe. Flucelvax previously had an approved quadrivalent version, and this time an additional trivalent version has been approved. This is interpreted as the company obtaining approval for a new product to transition to a trivalent vaccine in accordance with WHO recommendations. Furthermore, a trivalent vaccine is now mandatory for pharmaceutical companies wishing to participate in the National Immunization Program (NIP).The WHO recommended a trivalent influenza vaccine in February of this year, following its recommendation last year, noting that not a single case of the B/Yamagata lineage virus has been detected worldwide since March 2020.In particular, the WHO determined that there is no longer a need to include a viral antigen that no longer exists in the vaccine. Consequently, it established the trivalent vaccine composition, which excludes the Yamagata antigen from the existing quadrivalent vaccine system, as the standard and recommended a complete return to trivalent vaccines.Following WHO guidance, the government already switched the NIP influenza vaccines from quadrivalent to trivalent last year. As a result, quadrivalent vaccines were sold only in the private market. However, some confusion arose as certain pharmaceutical companies continued promoting quadrivalent vaccines without clearly disclosing the WHO recommendation.This year, as pharmaceutical companies are gradually shifting back to the trivalent vaccine, there are predictions that the quadrivalent vaccine may completely disappear from the market.
- Policy
- Complete revision to the "Innovative Pharma Company" certification
- by Lee, Jeong-Hwan Mar 27, 2026 08:32am
- The Ministry of Health and Welfare (MOHW) will make administrative move to encourage drug investment by raising the R&D expenditure requirement for "Innovative Pharmaceutical Company" certification.The MOHW will improve administrative fairness by revising the disqualification criteria related to illegal rebates. Specifically, "rebate violations that concluded more than five years before the point of certification or renewal evaluation" will now be excluded from the review process. The new policy aims to address the irrationality of revoking certifications based on violations that occurred in the distant past. However, the proposed "points-based scoring system" for rebate-related cancellations will not be introduced.Additionally, the certification track for foreign pharmaceutical companies will be newly established and revised. To encourage foreign firms to host research and production facilities in Korea, attract overseas capital, and engage in joint research and open innovation, the score weightings for these categories will be increased.Transparency in the certification process will also be enhanced. The minimum passing score for certification (65 points) will be explicitly stated in the official notice, and companies that fail to gain certification will be formally notified of the specific reasons for their disqualification.The Ministry of Health and Welfare (MOHW) revises the "Innovative Pharmaceutical Company" certification. 1. R&D expenditure ratio will be raised 2. "Rebate violations that concluded more than five years before the point of certification or renewal evaluation" will be excluded from review 3. Foreign company track for Innovative Pharmaceutical Company certificationOn the 26th, the MOHW issued a legislative and administrative notice regarding amendments to the Enforcement Decree, Enforcement Rules, and related notices of the "Special Act on Promotion and Support of Pharmaceutical Industry" (the Pharmaceutical Industry Act). The public opinion engagement remain open until May 6.R&D Expenditure Ratio to Rise by 2%pFirst, the R&D expenditure ratios required for the certification and renewal of Innovative Pharmaceutical Companies will be increased. While the ratio relative to pharmaceutical sales will rise by 2 percentage points across the board, the application of this new standard will be 'deferred for three years from the date of enforcement' to allow companies sufficient time to prepare.Through this administrative move, the MOHW aims to encourage a continuous expansion of R&D investment among certified firms.Furthermore, the system will now categorize firms into "Innovative Pharmaceutical Companies" and "Foreign Innovative Pharmaceutical Companies" to provide a regulatory system that accounts for the unique characteristics of multinational corporations. The track for foreign firms will be implemented immediately upon announcement.For companies holding cGMP (current Good Manufacturing Practice) or EU GMP certification seeking renewal under the relaxed "R&D expenditure-to-sales" ratio, they must now submit evidentiary materials prepared within 3 years of the certification's expiration date. This administrative move resolves the current lack of a specific timeframe for GMP-related documentation.The amendments to Enforcement Rules will take effect on the date of announcement and are expected to apply to renewal applications starting in the second half of this year.Revising 'Rebate Checking Policies' and 'Detailed Evaluation Criteria'Regarding illegal rebates, the MOHW has decided to exclude violations that occurred more than 5 years before the certification or renewal review.In cases where administrative appeals or lawsuits are filed, certification can be revoked within one year of the date of dismissal of the appeal or a final ruling against the company.By maintaining the current revocation structure rather than switching to a points-based system, and excluding violations older than five years, the MOHW aims to eliminate the irrationality of penalizing firms for old actions. This addresses repeated parliamentary criticism that legal stability and predictability for pharmaceutical companies were undermined when certifications were revoked for ancient violations.The current standards state that administrative actions for rebates under the Pharmaceutical Affairs Act or the Fair Trade Act that occurred five years before the certification review are excluded. However, if a lawsuit is filed challenging the administrative action, the date the judgment becomes final is treated as the date of the administrative action.Consequently, cases have occurred in which "Innovative Pharmaceutical Company" certifications were revoked due to rebate violations that occurred a long time ago. Criticisms rose from the National Assembly that the predictability and legal stability for pharmaceutical companies are being undermined.Furthermore, the detailed evaluation criteria for certification will also be improved and will be noted in the appendix. The total score will be adjusted from 120 to 100 points, and the number of evaluation items will be reduced from 25 to 17.Objectivity in certification standards will be enhanced by converting evaluation items, such as R&D investment, the number of clinical trials, and export volume, into quantitative indicators (4 out of 17 items).Notably, a new category will be established to recognize excellence in social responsibility activities, such as the production and supply of medicines that help stabilize the supply chain.For the Domestic Innovative Pharmaceutical Company certification standards, the score weightings for items such as partnership·collaboration activities, non-clinical·clinical trials, candidate development, and transparency in corporate management will be increased. Conversely, items such as research personnel, research/production facilities, and R&D strategies will be adjusted downward.Establishment of Certification Standards for Foreign Innovative Pharmaceutical CompaniesFurthermore, based on the types of Innovative Pharmaceutical Companies classified under the Enforcement Decree of the Pharmaceutical Industry Act, detailed certification review criteria for Domestic Innovative Pharmaceutical Companies and Foreign Innovative Pharmaceutical Companies will be separately regulated.Foreign pharmaceutical companies will be permitted to choose and apply under either the Domestic Innovative Pharmaceutical Company certification standards or the Foreign Innovative Pharmaceutical Company certification standards.The certification standards for Foreign Innovative Pharmaceutical Companies will have increased weightings for specific items to encourage foreign pharmaceutical firms to establish domestic research and production facilities, as well as to attract foreign capital, facilitate joint research, and promote open innovation.Given the characteristics of foreign pharmaceutical companies, in which the global headquarters typically holds the technology and patents, the score weightings for items related to the development of non-clinical and clinical trial candidates and to pharmaceutical patent technology transfer performance will be reduced.Finally, the MOHW will state the minimum passing score of 65 points and mandate that the reasons for failure be specified in notifications to companies. These changes will be reflected starting with new and renewal applications in the second half of this year.Meanwhile, the MOHW plans to systematically analyze the types and capabilities of Korean pharmaceutical and biotech companies and establish a "Strategy for Fostering National Pharmaceutical & Biotech-Industry" within this year.