- LOGIN
- MemberShip
- 2026-06-10 21:10:43
- Policy
- COVID-19 pill now limited to 'Paxlovid'… Lagevrio stock depleted
- by Lee, Jeong-Hwan Feb 27, 2026 08:36am
- From now on, oral COVID-19 treatments will be limited to 'Paxlovid.' This is due to the stock exhaustion of Lagevrio, another oral antiviral, which will lead to its discontinuation starting on the 17th of next month.On the 26th, the Korea Disease Control and Prevention Agency (KDCA, Commissioner Seung-Kwan Lim) announced that, since the government-supplied stock of Lagevrio has been depleted, the available oral treatments for COVID-19 will be restricted to Paxlovid only.Originally, among the three COVID-19 treatments supplied by the government (Paxlovid, Lagevrio, and Veklury), Paxlovid and Veklury received marketing authorization and have been covered by Nationa Health Insurance since October 25, 2024.Lagevrio's marketing authorization process has not been completed and has maintained only Emergency Use Authorization (EUA) to date. While it continues to be supplied on a limited basis using government inventory, its shelf life has expired, and its use will be discontinued starting from the 17th of next month.Paxlovid is used for mild-to-moderate patients among the elderly (aged 60 and over), those with underlying medical conditions, and immunocompromised individuals. Patients for whom Paxlovid administration is restricted typically use Lagevrio or Veklury.Once Lagevrio use is discontinued, patients previously eligible for Lagevrio can use Veklury. Clinics will need to refer patients restricted from Paxlovid to hospitals where Veklury can be administered.Meanwhile, the approval scope of Paxlovid was recently expanded by the Ministry of Food and Drug Safety (MFDS) to include patients with severe renal impairment, including those on dialysis (as of January 14).Patients with severe renal impairment, who were previously used to taking Lagevrio because Paxlovid was not recommended, can now be administered Paxlovid through dose adjustment. Consequently, it is expected that a significant number of patients who previously received Lagevrio due to difficulties in prescribing Paxlovid will now be able to transition to Paxlovid.The government is preparing measures to improve prescribing convenience at medical institutions to facilitate the use of Paxlovid, which will become the sole oral treatment available.Paxlovid involves some inconvenience in clinical settings because of the need to verify the patient's use of contraindicated drugs (40 types).To expand the use of Paxlovid among those taking contraindicated drugs, the KDCA plans to distribute pamphlets providing detailed guidance based on the marketing authorization (such as suspending the drug in question or prescribing alternative medications).
- Policy
- Novo Nordisk’s next-gen obesity drug enters Korean trials
- by Lee, Tak-Sun Feb 27, 2026 08:35am
- AI-generated imageNovo Nordisk, the developer of the obesity treatment Wegovy (semaglutide), will conduct multinational Phase III clinical trials for its next-generation obesity therapy in Korea.The candidate is amycretin, the company’s next-generation obesity treatment candidate , with the company aiming for global commercialization through multinational trials.On the 24th, the Ministry of Food and Drug Safety (MFDS) approved two Investigational New Drug (IND) applications for Novo Nordisk’s NNC0487-0111.One is a Phase IIIa trial (AMAZE 2) evaluating the efficacy and safety of once-weekly NNC0487-0111 s.c. in subjects with overweight or obesity and type 2 diabetes. The other is a Phase IIIb clinical trial (HF-POLARIS) evaluating the efficacy and safety of NNC0487-0111 versus placebo on morbidity and mortality in patients with obesity and heart failure with preserved or mildly reduced ejection fraction.NNC0487-0111 is the development code for amycretin, which is being developed by Novo Nordisk.Amycretin is a next-generation obesity treatment with a dual mechanism of action: it stimulates the GLP-1 receptor like Wegovy, while also acting on the pancreatic hormone amylin, which is involved in appetite regulation.Novo Nordisk is developing amycretin in both a once-weekly injectable formulation and a once-daily oral formulation. In earlier clinical trials, amycretin demonstrated up to 24.3% weight reduction over 36 weeks of subcutaneous administration.Based on these results, Novo Nordisk plans to conduct a multinational Phase III trial starting this year. Commercialization is expected after 2028. South Korea is included in the global, multi-national Phase III trial network.In Korea, the planned enrollment is 155 patients for HF-POLARIS (global total: 5,610) and 60 patients for AMAZE 2 (global total: 630).HF-POLARIS will be conducted at Wonju Severance Christian Hospital, Seoul National University Hospital, Chungnam National University Hospital, Gachon University Gil Medical Center, Pusan National University Yangsan Hospital, Inha University Hospital, Ajou University Hospital, Samsung Medical Center, Severance Hospital (Sinchon), Seoul National University Bundang Hospital, Keimyung University Dongsan Medical Center, Seoul St. Mary’s Hospital, Korea University Ansan Hospital, Seoul Asan Medical Center, and Hallym University Sacred Heart Hospital.AMAZE 2 will take place at Kyung Hee University Medical Center, Chonnam National University Hospital, Seoul Asan Medical Center, Seoul National University Hospital, Seoul National University Bundang Hospital, Yeouido St. Mary’s Hospital, and Severance Hospital (Sinchon).Novo Nordisk’s development of amycretin is viewed as a strategic move to maintain competitiveness in the GLP-1 obesity treatment market.In Korea, Wegovy and Eli Lilly’s Mounjaro have formed a two-horse race. Wegovy, approved by the MFDS in 2023, recorded domestic sales of KRW 467 billion last year, according to IQVIA.Mounjaro, which targets both GIP and GLP-1 receptors, was launched in Korea in the second half of last year and generated KRW 215.5 billion in sales. The two companies continue to compete globally, including through the development of oral obesity treatments.
- Policy
- Opposition boycott puts pharmacy and drug bills on hold
- by Lee, Jeong-Hwan Feb 26, 2026 07:48am
- On the 24th, the main opposition party, the People Power Party, decided to launch a filibuster on all bills submitted for consideration, along with a full boycott of standing committee proceedings, in response to the Democratic Party’s unilateral push to proceed with a plenary session.As a result, the meetings of the 1st and 2nd Subcommittees of the Legislation and Judiciary Committee of the National Assembly’s Health and Welfare Committee, originally scheduled for the 25th and 26th this week, now face uncertainty regarding whether they will convene.Particularly for Subcommittee 1, chaired by People Power Party lawmaker Miae Kim, the atmosphere suggests it is highly unlikely to convene.While some within the ruling party have suggested holding a plenary session alone to review Subcommittee 1's bills, unilaterally reviewing and passing livelihood bills would inevitably invite criticism for bypassing established inter-party consensus procedures.If Subcommittee 1 fails to convene, bills expected to be tabled, including legislation on limited international nonproprietary name (INN) prescribing for drugs facing supply instability or designated as nationally essential medicines, as well as bills regulating the labeling, advertising, promotion, and establishment scale of warehouse-style pharmacies, would lose their opportunity for review.This would effectively delay the examination of livelihood-related legislation within the Health and Welfare Committee due to ongoing partisan conflict.Meanwhile, the 2nd Subcommittee of the Legislation and Judiciary Committee, chaired by Democratic Party lawmaker Sujin Lee, could theoretically proceed without participation from the People Power Party. Among its jurisdiction is the bill proposing the establishment of a public medical school, a key component of the Lee Jae-myung administration’s policy agenda to strengthen regional, essential, and public healthcare.However, even this remains uncertain. Holding a subcommittee meeting solely by the ruling party to review and pass bills could face opposition from the opposition party later and could escalate conflict between the ruling and opposition parties within the Welfare Committee.As a result, the schedule for the Welfare Committee's bill subcommittees has been disrupted due to clashes between the ruling and opposition parties over the third amendment to the Commercial Act, which primarily concerns mandatory retirement of treasury shares, judicial reform bills, and the Special Act on Administrative Integration.An official from the Democratic Party's Welfare Committee office said, “While the Subcommittee 2 (chaired by the Democratic Party) might be able to convene with the ruling party alone, Subcommittee 1 appears difficult to hold. There have been suggestions to conduct an article-by-article review of Subcommittee 1 bills at the plenary session level, but the political burden is considerable.”
- Policy
- DPK moves to hold Welfare Committee plenary session unilaterally
- by Lee, Jeong-Hwan Feb 26, 2026 07:47am
- The Democratic Party of Korea has decided to convene a plenary session of the National Assembly's Health and Welfare Committee on the 26th, solely with its own members, to receive a report from the Ministry of Health and Welfare on current issues, including medical school enrollment expansion and regional, essential, and public healthcare policies.This move comes in response to the main opposition party, the People Power Party, conducting a filibuster (unlimited debate) and deciding to boycott all standing committee proceedings, citing the Democratic Party's unilateral handling of the plenary session agenda.Notably, some Democratic Party members of the committee have also raised the need to review and pass bills assigned to the first Subcommittee of the Legislation and Judiciary Committee during the plenary session.However, such a move is expected to trigger significant friction with the People Power Party over future standing committee operations and bill deliberations.According to a Democratic Party official on the 25th, the Health and Welfare Committee (Chair Jumin Park) has tentatively decided to hold a plenary session on the afternoon of the 26th. During the session, the Ministry of Health and Welfare will present reports on key pending issues, including medical school quota expansion, emergency medical care, and the regional physician initiative, and adopt the plan for a public hearing on the Patient Basic Act bill.The second Subcommittee of the Legislation and Judiciary Committee will convene as scheduled at 10 a.m. on the 27th.This decision to proceed with the Welfare Committee plenary session and the second Subcommittee of the Legislation and Judiciary Committee's meeting was made solely by the Democratic Party, without involvement from the People Power Party.It is reported that the decision to hold the plenary session solely by the Democratic Party came after the first Subcommittee of the Legislation and Judiciary Committee meeting, originally scheduled for the 26th, could not be held due to a boycott by the People Power Party.A key point of attention is whether the Democratic Party will attempt to deliberate and pass bills assigned to the first Subcommittee of the Legislation and Judiciary Committee during the plenary session.This necessity arose because some ruling party welfare committee members have strongly criticized the fact that the subcommittee has not convened for several months and argued that it may now be necessary to process pending legislation at the plenary level.The problem is that if the Democratic Party reviews and votes on bills under the jurisdiction of the first subcommittee at the plenary session without the opposition parties, including the People Power Party, the Welfare Committee could face disruptions in its normal operations due to clashes between the ruling and opposition parties.A Democratic Party welfare committee official hinted, “Due to the opposition party's lack of cooperation, bill subcommittee meetings failed to take place in December and January, and there is now a risk that February will follow the same pattern. There are strong voices from some lawmakers stating the necessity to review past livelihood-related bills during the plenary session.”
- Policy
- Expanded reimb for Imfinzi will be available next month
- by Jung, Heung-Jun Feb 25, 2026 05:46pm
- AstraZeneca Korea's Imfinzi New reimbursement criteria for anticancer drugs will be established ahead of the expanded insurance coverage for AstraZeneca Korea's Imfinzi (durvalumab), an immunotherapy used to treat cancer.Starting in March, Imfinzi combination therapy will be added for both liver cancer and biliary tract cancer. Notably, in liver cancer, the drug achieved dual success by simultaneously proving its combination therapy with Imjudo (tremelimumab).On the 23rd, the Health Insurance Review and Assessment Service (HIRA) announced that it is currently conducting an opinion survey regarding the revision of the "Application criteria for National Health Insurance reimbursement for drugs prescribed and administered to cancer patients." After the survey ends on the 25th, the revised contents will be applied starting in March.For liver cancer, the combination therapy with Imjudo will be newly established. Reimbursement is limited to patients with advanced hepatocellular carcinoma who are ineligible for surgery or local treatment and who meet specific criteria.The reimbursement criteria are appliable up to 1 year of treatment. However, if clinical results for the administration period within that year have not been published, reimbursement will be automatically extended for up to 2 years.For biliary tract cancer, the combination therapy of Imfinzi (durvalumab) + gemcitabine + cisplatin will be newly established. Insurance coverage will apply to patients with unresectable locally advanced or metastatic biliary tract cancer. This is limited to adenocarcinoma and excludes ampullary Vater carcinoma.Gemcitabine and cisplatin will not be administered after the initial 8 cycles of combination therapy. The reimbursement period is the same as that for the liver cancer combination therapy.Imfinzi is becoming reimbursed for two combination therapies. Consequently, Imfinzi prescriptions, which had been concentrated on lung cancer, are expected to expand into liver cancer and biliary tract cancer.In particular, it will emerge as a new treatment option for biliary tract cancer. Imfinzi is the second drug to receive a flexible Incremental Cost-Effectiveness Ratio (ICER) for an innovative new drug.In this revision, Janssen Korea's Balversa (erdafitinib) monotherapy for urothelial carcinoma was also newly established. It is indicated for second-line or later use, and the target population is "patients with unresectable locally advanced or metastatic urothelial carcinoma with FGFR3 genetic alterations whose disease has progressed during or following at least one prior systemic therapy, including a PD-1 or PD-L1 inhibitor."For multiple myeloma, a second-line or later combination therapy of Antengene's Xpovio (selinexor) + bortezomib + dexamethasone was newly established. It can be administered to patients with multiple myeloma who have failed previous treatments.Xpovio received a decision on reimbursement expansion conditions from the Drug Benefit Evaluation Committee meeting last November, which required the company to accept a price below the evaluated amount.
- Policy
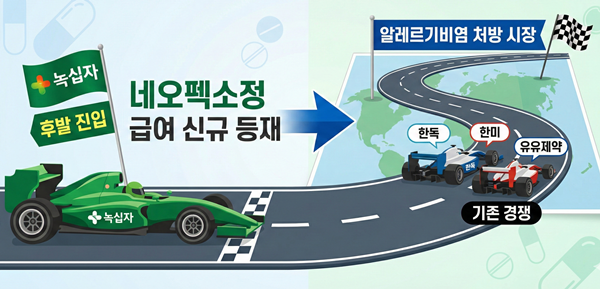
- GC Biopharma expands lineup to target antihistamine market
- by Jung, Heung-Jun Feb 25, 2026 05:45pm
- GC Biopharma is expanding its reimbursed lineup of its fexofenadine-based Neofexo Tablets this year to strengthen its position in the prescription market for allergic rhinitis.By increasing the number of reimbursed items, it is entering the antihistamine prescription market as a latecomer, where companies like Handok, Hanmi, and Yuyu are competing.According to industry sources on the 24th, following the listing of Neofexo Tab 120mg (fexofenadine hydrochloride) in January, GC Biopharma will add Neofexo Tab 180mg to the reimbursement list in March.GC Biopharma plans to list Neofexo Tab 180mg for reimbursement in March. AI-generated image.The reimbursement ceiling price for Neofexo Tab 180mg has been set at KRW 267. Neofexo Tab 120mg, listed in January, previously received a ceiling price of KRW 220.Fexofenadine, a third-generation antihistamine, is marketed as both an over-the-counter (OTC) and prescription drug. The 120mg formulation is available in both categories, while the 180mg strength is prescribed exclusively.OTC formulations are indicated solely for the relief of seasonal allergic rhinitis, whereas prescription products carry broader indications, including symptom relief in chronic idiopathic urticaria.Neofexo Tab 120mg was approved as an OTC product, while the newly listed Neofexo Tab 180mg can only be sold as a prescription drug.A review of previously reimbursed products shows that companies including Handok, Hanmi Pharm, Yuyu Pharma, Chong Kun Dang, Il-Yang Pharm, and Hutecs Korea are actively competing in the prescription segment.Meanwhile, Allegra Tab, the original fexofenadine product, continues to demonstrate steady sales growth. According to UBIST, Allegra recorded sales of KRW 7.9 billion last year, marking a 9% increase year-on-year. Hanmi Pharm’s Fexonadine Tab also posted modest growth, rising 2% to KRW 3.3 billion.GC Biopharma already markets Allerjet Soft Cap, a 60mg OTC fexofenadine hydrochloride product approved in 2023.With reimbursement coverage now for Neofexo Tab, the company is expected to accelerate its strategic push into the allergic rhinitis market.
- Policy
- MFDS offering regulatory support for orphan drug discovery
- by Lee, Tak-Sun Feb 25, 2026 05:45pm
- The Ministry of Food and Drug Safety (MFDS) plans to accelerate the commercialization of government-led orphan drug (treating rare diseases) development by providing regulatory support from the early stages of development.The MFDS has initiated support for the orphan drug sector through the "Overcoming Unconquered Diseases" project, the Korean ARPA-H initiative led by the government.Through this support, the MFDS aims to accelerate the commercialization of orphan drugs and improve patient access to treatment for ultra-rare diseases.In observance of "Overcoming Rare Disease Day" on the 28th, the MFDS held a briefing for the medical and pharmaceutical press to explain the current status of support and policies for rare disease treatments.In observance of "Overcoming Rare Disease Day" on the 28th, the MFDS held a briefing for the medical and pharmaceutical press. (from left) Project Manager Misun Park of the Korea Health Industry Development Institute (KHIDI); Hyeon Jin Yim, Head of the Regulatory Science Policy Promotion Division; and Division Heads Chun-rae Kim, Jae-hyun Park, and Mi-ryeong Ahn."World Rare Disease Day" falls on February 29th, the rarest day of the year that occurs once every four years. Under the Rare Disease Management Act, the Korean government commemorates the last day of February each year (typically February 28th or 29th) as Rare Disease Day. The goal is to enhance public understanding of rare diseases and improve access to treatment and medical support for patients and their families.While South Korea has designated 1,380 rare diseases, drug development remains challenging due to the small patient population and the difficulty of attracting corporate investment.The government began treatment development through the Korean-style ARPA-H project. Benchmarked after the U.S. "ARPA-H (Advanced Research Projects Agency for Health)," this project is a "high-risk, high-return" national R&D initiative that began last year.In the field of rare diseases, projects currently underway include the development of a tailored, innovative treatment platform for pediatric rare disease patients, the N-of-1 (single-patient) clinical trial project (HEART), and patient-customized gene therapy to overcome visual impairment in hereditary eye diseases (BEACON). A national budget of KRW 17.5 billion is allocated for investment over 4.5 years.Typically, national R&D projects often conclude with researchers registering papers or patents. However, this project aims for commercialization. Consequently, regulatory support from the MFDS is considered crucial.Misun Park, Project Manager of the K-Health Future Promotion Division at KHIDI, explained, "To meet regulatory requirements after development has significantly progressed can lead to irreversible losses in time and cost. It is essential to communicate with the MFDS from the early stages of development to collaborate on clinical trial design and the direction of data preparation."Furthermore, due to the nature of rare diseases, patient administration cannot be cancelled even if any issues arise during the Investigational New Drug (IND) process. Therefore, the aim is to minimize trial-and-error by consulting with the MFDS from the start of development.The MFDS provides early-stage regulatory support through the "Regulatory Adequacy Review System," which has been in place since last year. This system analyzes and supports regulatory requirements during the initial stages of national R&D projects.The system reviews how products under development are classified and which laws apply to them, and provides consultations on evaluation criteria and methods for demonstrating safety and efficacy. Through this, regulatory response strategies and the necessity of joint research with the MFDS are also reviewed.Last year, the system supported commercialization by classifying stem-cell-based artificial blood projects as advanced biopharmaceuticals and recommending GRADE changes for digital therapeutic development projects for developmental disabilities.Hyeon Jin Yim, Head of the Regulatory Science Policy Division at the MFDS, stated regarding the system, "Researchers often develop innovative technologies but face difficulties at the commercialization stage because they do not fully understand regulatory procedures. The Ministry provides guidance from the beginning on which laws apply, what data needs to be prepared, and what strategies are necessary to increase the likelihood of approval."Yim added, "Preventing delays in commercialization is possible if the time required for supplementation is shortened by analyzing regulatory targets in advance. We are currently in discussions regarding eight projects under the ARPA program."The MFDS is also working to improve accessibility by easing designation requirements for orphan drugs and implementing expedited reviews. Starting this year, drugs can receive orphan drug designation (ODD) even without submitting data demonstrating significantly improved safety or efficacy compared to existing alternatives.Chun-rae Kim, Head of the Pharmaceutical Policy Division at the MFDS, explained, "Previously, for rare disease drugs announced by the Korea Disease Control and Prevention Agency (KDCA), data proving improved safety and efficacy had to be submitted. However, through consultations with the pharmaceutical industry last year, we decided to omit this regulatory requirement."This year, the Ministry plans to discuss with the industry measures to ease regulations on orphan drug designation cancellations.When designated as an orphan drug, rapid approval can be expected through expedited regulatory reviews. The MFDS selects drugs for expedited review through the "GIFT" (Global Innovative products on Fast Track) system. Currently, of the 50 items approved as GIFT products, 42 are orphan drugs.Jae-hyun Park, Head of the Expedited Review Division at the MFDS, stated, "According to the disease groups of treatments designated as GIFT, half are recurrent or intractable cancers with small patient populations. For these patients, clinical trials themselves can be a treatment opportunity. In the future, we plan to consult with the public and private sectors to ensure that patient opinions are reflected in GIFT reviews."For orphan drugs not designated under GIFT, the MFDS also offers flexibility in reviews, granting approval based on Phase 2 clinical trials, with the requirement to submit Phase 3 clinical data post-market. Mi-ryeong Ahn, Head of the Oncology and Antibiotics Division, added, "We continue to develop guidelines related to clinical trial design to increase flexibility at the approval stage."
- Policy
- First matching-form Ofev generic listed at half price
- by Jung, Heung-Jun Feb 24, 2026 03:55pm
- A generic version of the chronic fibrotic interstitial lung disease treatment Ofev Soft Cap (nintedanib esylate) with the same formulation will be listed for reimbursement for the first time.With its reimbursement entry at a price more than 50% lower than the original, full-scale market competition is expected.According to industry sources on the 20th, Ildong Pharmaceutical’s Cuninta Soft Cap 100 mg and 150 mg will be added to the March reimbursement list.The substance patent for Boehringer Ingelheim Korea's Ofev Soft Capsules expired in January last year. Subsequently, generic companies have successively launched their versions.However, unlike the original drug, those products obtained approval in tablet formulations. From July last year, Yungjin Pharm, Ildong Pharmaceutical, Daewoong Pharmaceutical, Kolon Pharmaceutical, and others entered reimbursement. Whan In Pahrm also received approval for Ofenib Tab, but the drug is yet to be listed for reimbursement.Until now, no reimbursed generic matching Ofev’s soft capsule formulation has been available. Ildong Pharmaceutical is expected to be the first to list a soft capsule generic, Cuninta Soft Cap, following its release of Cuninta Tab.Cuninta Tab 150 mg was previously listed at KRW 13,500, roughly half the price of Ofev Soft Cap, which was priced at KRW 26,220.Although generics designated as orphan drugs could receive equivalent pricing to the original, manufacturers pursued lower prices to strengthen market competitiveness.The reimbursement ceiling prices for Cuninta Soft Cap 100 mg and 150 mg are set at KRW 8,500 and KRW 13,500, respectively. Notably, the lower dose represents about 40% of the originator’s 100 mg price of KRW 20,960.Where earlier competition strategies relied on tablet differentiation and lower pricing, manufacturers can now seek prescription expansion based on identical formulation at half the cost.Because switching prescriptions within the same formulation is expected to face less patient resistance, Ildong Pharmaceutical is anticipated to intensify its competitive positioning against Ofev.Given that Ofev entered reimbursement relatively late, 8 years after domestic approval, Ofev is expected to face intensifying competition from the later entrant generics.
- Policy
- Government to establish bioequivalence standards for complex generics
- by Lee, Jeong-Hwan Feb 24, 2026 03:55pm
- The government is moving forward with the development of guidelines, including bioequivalence evaluation standards for complex generic drugs.This initiative aims to support the rapid development of complex generics as market entry for new technology and new concept drugs increases.Formulations targeted for new assessment criteria and testing methodologies include oral agents, injectables, inhalation products, and ophthalmic preparations.On the 20th, the National Institute of Food and Drug Safety Evaluation (NIFDS) under the Ministry of Food and Drug Safety (MFDS) announced the launch of a research project titled the “New Technology and New Concept Drug Guideline Development Project”.To develop bioequivalence assessment standards and testing method guidelines for complex generics, the Ministry of Health and Welfare (MOHW) will establish a multi-layered advisory framework involving industry, academia, and regulatory experts. Policy decisions will be made through an expert committee following internal review for stage-by-stage examination and verification.The research will define the concept of complex generics and conduct comparative analyses of domestic pharmaceutical industry demand alongside guideline frameworks from major global regulatory authorities to create new guidelines.Guidelines currently operated by the US FDA, European EMA, and Japan PMDA will be analyzed. Priorities for new guideline development will be determined based on technological impact, industry needs, and regulatory necessity.Subsequently, it will present general evaluation principles applicable to all dosage forms of complex generics and publish a reference manual to support development.In addition, bioequivalence guidelines specific to complex generics will be developed. It plans to create equivalence evaluation guidelines for oral, injectable, inhaled, and ophthalmic combination generics, validate them through a separate expert advisory body, including the Ministry of Food and Drug Safety (MFDS), and then finalize them.For this year, oral formulations selected for guideline development include pentosan polysulfate sodium products, ferric citrate hydrate capsules, and mesalamine extended-release tablets.Injectable products include dexamethasone extended-release implants and paliperidone palmitate long-acting injections. Inhalation products cover budesonide, formoterol fumarate inhalation aerosols, fluticasone propionate inhalers, and fluticasone propionate/salmeterol xinafoate inhalation powders.Ophthalmic formulations include brinzolamide suspension eye drops and prednisolone acetate suspension eye drops.Notably, authorities plan to establish a collaborative framework with the task force supporting rapid drug development guidelines. Once completed, newly developed guidelines will be promoted to industry and academic stakeholders, alongside implementation activities such as conferences.NIFDS stated, “We plan to develop more than 10 bioequivalence guidelines for complex generics and publish a project report. Through technological support for complex generics, we aim to accelerate the timely commercialization of high-quality generics while strengthening review reliability and transparency through science-based guidelines.”
- Policy
- Backlash mounts to drug price reduction reform plan
- by Lee, Jeong-Hwan Feb 24, 2026 03:55pm
- The Korean pharmaceutical industry continues criticizing that the background of the Minister of Health and Welfare (MOHW)'s drug pricing system reform, which includes 'calculation criteria for impact of drug price reductions' and 'preferential drug price criteria,' is unreasonable.The MOHW calculated simulation results for losses incurred from drug price reductions based on total sales amount. However, the view is that the calculation standard itself is flawed, as sales amount includes all accounting elements involved in the manufacturing and production of medicines.Furthermore, the criticism is that preferential drug price clauses based on whether a company is certified as an Innovative Pharmaceutical Company or on its ratio of new drug Research and Development (R&D) expenditure to sales are inappropriate because they are arrived at unilaterally, without proper agreement between the government and the industry.On the 22nd, the pharmaceutical industry voiced concerns about MOHW's unilateral push for drug pricing reform, stating, "The justification for the price reduction is unclear, and such administration significantly undermines trust in the government, leading to results that run counter to the fostering of the global pharmaceutical industry."On the 20th, the Health Insurance Policy Deliberation Committee (HIPDC) subcommittee excluded the drug pricing system reform plan, including the price reduction for currently listed generics, from its agenda.The MOHW cited insufficient gathering of industry opinions and said it would continue communicating with the industry until next month's (March) HIPDC meeting.Despite the MOHW’s decision to defer or postpone the agenda, the pharmaceutical industry remains skeptical about the possibility of revisions to the reform plan. This is due to the observation that the MOHW has not shown a proactive negotiation attempts thus far.Korean pharmaceutical companies cite the MOHW's methodology for calculating the impact of drug price reductions as the biggest issue.It is reported that the MOHW, based on simulation results analyzing the financial impact after price reductions using the sales revenue of several domestic pharmaceutical companies operating the generic business, concluded that revenue losses would be minimal even if the generic price calculation rate were lowered to 40%.Furthermore, the ministry believes that its reform plan is sufficiently valid and consistent, having derived results showing that certified innovative pharmaceutical companies or those with a high proportion of new drug R&D relative to sales would remain relatively unaffected by the price reductions.Korea's domestic pharmaceutical companies maintain that if the MOHW simulates the impact of price reductions based solely on total sales revenue or simple operating profit, an accurate evaluation is impossible.The reason is that a simulation based on sales revenue calculates figures by including every accounting element without exception. In contrast, one based on operating profit excludes selling, general, and administrative (SG&A) expenses. This evaluation method is impossible to reflect the innovation of pharmaceutical companies that have substantially contributed to the development of the domestic industry.In particular, while the MOHW has stated multiple times that the purpose of this drug pricing system reform is to foster the pharmaceutical industry rather than reduce health insurance drug expenditures, the pharmaceutical industry argues that reducing generic drug prices significantly while claiming to foster the industry is contradictory.The criticism is that the ministry's administrative goal, which it seeks to achieve through price reductions, is unclear.An official from domestic Pharmaceutical Company A pointed out, "The sales revenue evaluation, which is the standard for the MOHW’s impact simulation, is flawed as it includes all accounting elements," and added, "They must divert from single standards like sales revenue, perform a value evaluation considering various factors, and then proceed with simulations in a direction that grows the pharmaceutical industry and the drug market."Then added, "The MOHW changed the health insurance listing method for drugs from a negative list to a positive list system," and added, "This implies that the governemtn will make value judgments on drug prices. Therefore, all-at-once reduction of drugs listed through this system means the government considers existing medicines to have no value and intends to suppress the market."An official from domestic Pharmaceutical Company B also expressed, "The MOHW can analyze the reduction in pharmaceutical companies' revenue quite precisely through health insurance claim amounts or supply history reports. It is puzzling why they designed the drug price reduction and the pricing system reform plan with a simulation that excludes such big data when it is available for use."The official stated, "The pharmaceutical industry will be able to trust the government again only if the MOHW discloses transparent data regarding the evidence and standards used to design the drug pricing system reform and shows a willingness to negotiate," and added, "For now, meetings between the MOHW and the pharmaceutical industry may become a superficial formality, aimed at filing a quota of superficial mutual consultations."