- LOGIN
- MemberShip
- 2026-04-27 17:31:21
- Generic DOACs take over half of Xarelto mkt
- by Kim, Jin-Gu | translator Alice Kang | 2026-04-27 09:03:50
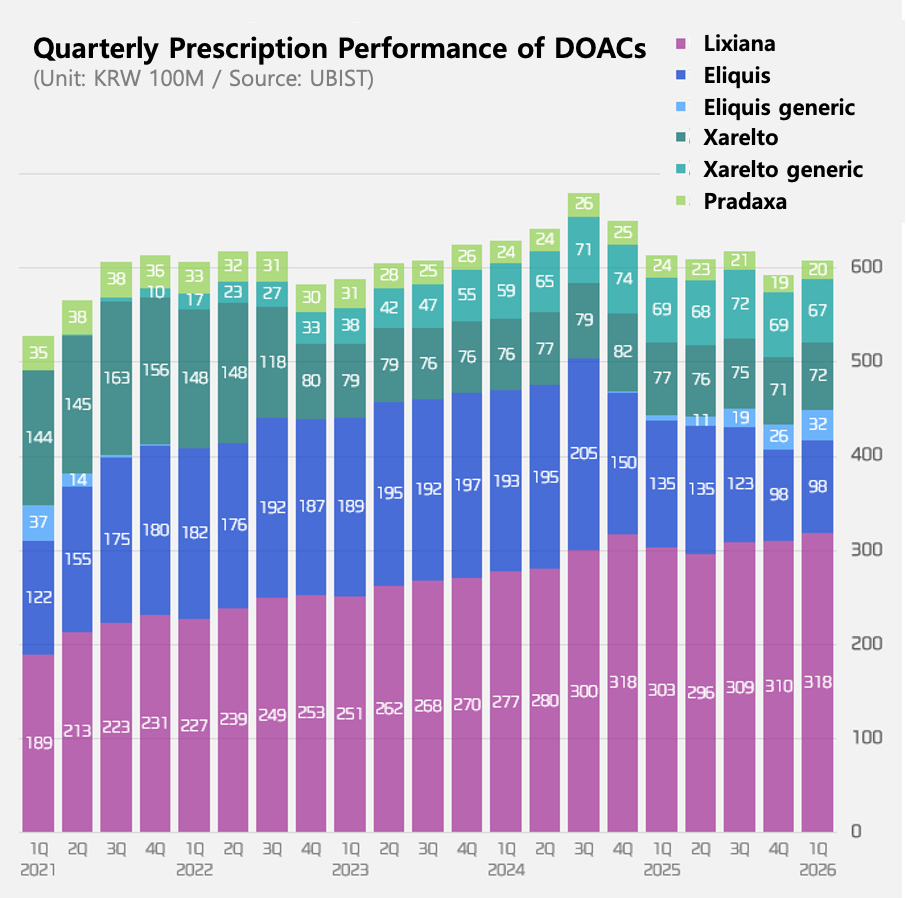
The KRW 260 billion direct oral anticoagulant (DOAC) market is seeing rapid generic penetration. Generic share has reached 49% for Xarelto (rivaroxaban) and 25% for Eliquis (apixaban).
Industry attention is now focused on Lixiana (edoxaban), whose generics are expected to launch later this year. As the top-prescribed product, its generic entry is expected to reshape the entire DOAC market. Fifteen companies have already received approvals and are waiting to launch.
DOAC market at KRW 60.8 billion in Q1…decline since Q3 2024
According to market research firm UBIST, outpatient prescriptions in the domestic DOAC market totaled KRW 60.8 billion in Q1.
Overall, the market has been declining since Q3 2024, due to patent expirations, generic launches, and the resulting price cuts.
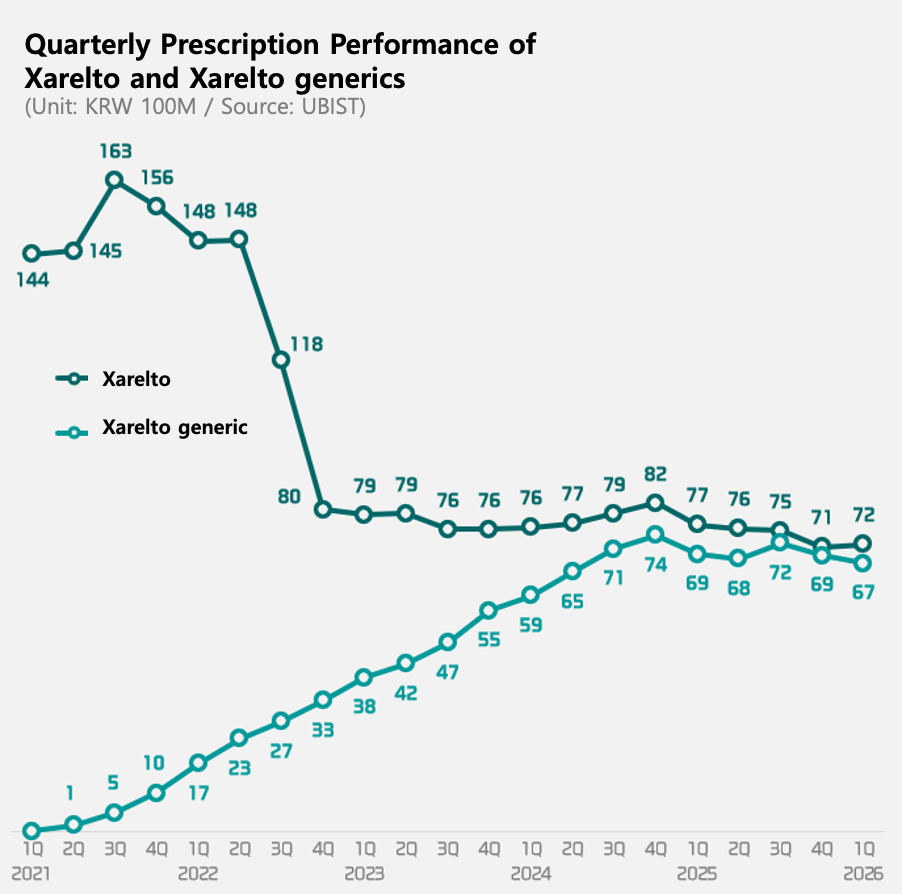
Xarelto’s patent expired in Q2 2021. Since then, 65 companies have launched Xarelto generics. These generics have rapidly increased their prescription volume since then. Since Q3 last year, they have maintained a market share of around 49% in the rivaroxaban market. This means that, just over four years after the launch of generics, their market share has expanded to a level comparable to that of the original product.
Amid the rapid growth of generics, prescriptions for the original Xarelto have been steadily declining. Q1 prescriptions were KRW 7.2 billion, down 6% YoY, roughly half of the KRW 16.3 billion recorded in Q3 2021.
Competition among generic products is also intensifying. As of Q1, Hanmi Pharm’s ‘Riroxban’ took the top spot among generics with prescription sales of KRW 2.2 billion, a 3% increase YoY. In contrast, the products ranked 2nd through 4th—Samjin Pharmaceutical’s ‘Rivoxaban,’ Chong Kun Dang’s ‘Riroxia’ and Daewoong Bio’s ‘Varelto’—all saw prescription sales decline by more than 10%.
As competition intensifies, the number of companies withdrawing from the market is also increasing. Last year, 14 products from six companies left the market due to the expiration of their validity periods. This year, market exit appears to be accelerating, with the validity periods of 38 products from 14 companies set to expire.
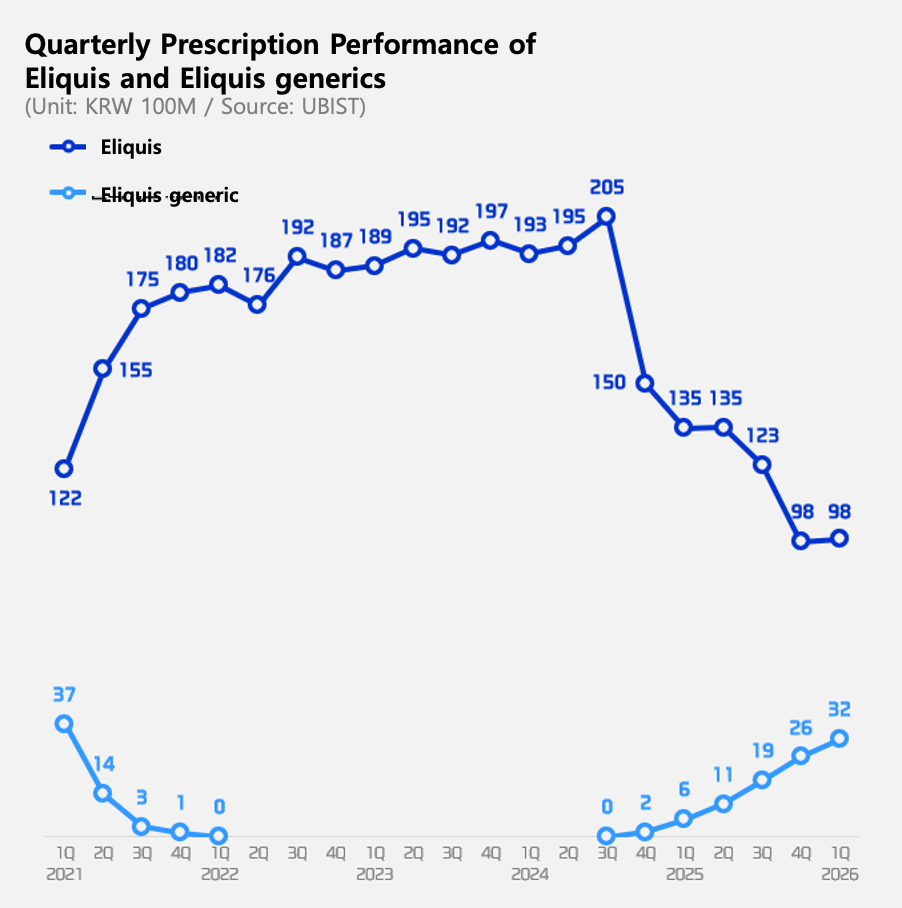
Eliquis generics reach 25% share just a year and a half after re-entry
Eliquis generics re-entered the market in Q4 2024. The generics were originally launched in June 2019. At the time, generic manufacturers released the products based on favorable rulings in the first and second instances of patent litigation. However, the situation reversed in April 2021 when the Supreme Court overturned the lower court rulings and ruled in favor of the originator, BMS. Generic manufacturers immediately withdrew from the market. This created a three-and-a-half-year gap in the market until the Eliquis composition patent expired last September.
Since re-entering the market, Eliquis generics have been gradually increasing their market share. The combined prescription value for Eliquis generics in the first quarter was KRW 3.2 billion, representing a market share of approximately 25% in the apixaban-based DOAC market. This is higher than the market share just before withdrawal (24% in Q1 2021). As of Q1, Chong Kun Dang’s ‘Liquisia’ and Samjin Pharmaceutical’s ‘Elxaban’ recorded KRW 1.4 billion and KRW 1.2 billion, respectively, taking the top two spots among generics. Prescription sales for other products were less than KRW 300 million.
In contrast, sales of the original Eliquis fell by 27% from KRW 13.5 billion to KRW 9.8 billion in just one year. Compared to the KRW 20.5 billion recorded in Q3 2024, just before the re-entry of generics, prescription sales have dropped to less than half in just a year and a half.
Lixiana, the current market leader, faces patent expiration by the end of this year… DOAC market to be reshaped with the entry of generics
Industry attention is now focused on the generics of Lixiana, the market leader. Lixiana’s composition patent expires this November. Generic companies have successfully circumvented the remaining patents.
Currently, 15 pharmaceutical companies, including Nexpharm Korea, DongKwang Pharm, Dongkook Pharmaceutical, Samsung Pharm, Shinil Pharma, Shinpoong Pharm, Ahngook Pharmaceutical, HK Inno.N, Ildong Pharmaceutical, Genuone Science, Union Korea Pharm, Korea Prime Pharm, Handok, and Hanmi Pharmaceutical, are awaiting the patent expiration with 35 generic product approvals already secured. With a KRW 120 billion market at stake, expectations among generic drug manufacturers are reportedly high.
Last year also saw a series of challenges to patents by generic companies. In 2023 alone, about 10 companies, including HLB Pharmaceutical, Daehwa Pharmaceuticals, Austin Pharma, CMG Pharmaceutical, Chong Kun Dang, Huons, Korea United Pharm, NBK Bio Korea Pharm, Myungmoon Pharm, and Boryung, filed patent invalidation suits and won their cases.
Challenges to the Lixiana formulation patent were conducted on a large scale in 2018 and concluded with a victory for the generic drug companies. Analysts suggest that a significant number of companies that were unable to avoid the patent at that time subsequently filed patent challenges in order to launch Lixiana generics. The pharmaceutical industry forecasts that about 30 companies, including those that have already received approval, will join the competition.
The original Lixiana further expanded its prescription volume ahead of the launch of generics. Lixiana’s prescription sales in Q1 reached KRW 31.8 billion, a 5% increase YoY. This marks the highest quarterly sales figure on record.
Lixiana was the last of the DOAC class of drugs to be launched. In South Korea, Xarelto (rivaroxaban) was approved in 2009, followed by Pradaxa (dabigatran) and Eliquis (apixaban) in 2011, then Lixiana in 2015. In the early stages of its launch, Lixiana ranked third in the market, trailing behind Xarelto and Eliquis. However, after partnering with Daewoong Pharmaceutical as its domestic co-promotion partner, it rose to the top of the market in 2019 and has been steadily increasing its prescription volume ever since.
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.
- "Vabysmo PFS shifts the retinal disease treatment persistence·efficiency"
- Reporter's view | Son, Hyung Min






