- LOGIN
- MemberShip
- 2026-06-10 20:05:12
- Industry calls for more proactive drug pricing incentives
- by Lee, Jeong-Hwan | translator Alice Kang | 2026-03-27 08:32:25

When asked what should be the top priority administrative action after the government fully implements the drug pricing reform plan in the second half of this year, the pharmaceutical industry immediately responded, “Establishing proper drug pricing incentive policies.”
Given that the reform plan places significant emphasis on lowering generic drug prices, the industry is demanding proactive measures to design policies that either clearly favor generics, which have contributed to savings in the National Health Insurance budget, or ensure that no further price cuts are imposed.
At the same time, the industry proposed conducting a proper post-evaluation of whether past policies, such as blanket price cuts for generics and differential pricing based on criteria, have actually achieved their intended goals of reducing NHI expenditure and addressing the excessive proliferation of generic products.
Following the across-the-board price cuts in 2012, virtually all pharmaceutical companies decided to produce multiple generic products, triggering the first generic drug boom. The proposal calls for a thorough analysis of why a second boom, marked by a further increase in the number of generic drug items, occurred following the 2020 announcement of a tiered pricing system based on requirements for in-house bioequivalence testing and Drug Master File (DMF) registration, in order to set the direction for a new drug pricing system.
On the 26th, the pharmaceutical industry criticized that government efforts to establish drug pricing incentives, which are closely tied to domestic companies’ investment in innovative drug R&D, remain significantly insufficient.
It also criticized the government for repeatedly introducing price cuts without conducting follow-up administrative measures to evaluate policies in collaboration with the industry, despite the fact that a boom in the number of generics has occurred every time drug prices were revised.
Domestic new drugs and generics lack sufficient incentives…“requires proactive administration”
Domestic pharmaceutical companies argue that a multidimensional approach to pricing incentives is necessary to achieve the government’s stated goal of shifting toward a new-drug-centered innovative pharmaceutical industry structure.
They criticize that the MOHW’s efforts are severely lacking, noting that corresponding preferential regulations are necessary to encourage pharmaceutical companies to develop globally competitive, first-in-class blockbuster drugs with no substitutes.
Suggestions followed that it is urgent to design a drug pricing system that comprehensively recognizes the value of domestic pharmaceutical companies, including establishing exclusive preferential regulations for domestic new drugs, creating an exception track for post-listing price reductions for domestic new drugs, and reducing the price reduction rate for generics that have significantly replaced originals and contributed to savings in the national health insurance budget.
In essence, they argue that the government must address the criticism that “there are plenty of policies that cut prices, but none that add value.”

Criticism that the MOHW focuses solely on price reductions and neglects efforts to establish preferential pricing regulations for domestically developed new drugs persists even in this latest drug pricing reform plan.
In the latest reform, the MOHW included provisions to delay or defer price cuts for already-listed generics for innovative and quasi-innovative pharmaceutical companies, and to apply a premium to their reduction rates. It also included preferential provisions for innovative and quasi-innovative companies regarding newly listed drugs
Nevertheless, the pharmaceutical industry remains steadfast in its position that there are no preferential policies that adequately compensate for the fair value of domestically developed new drugs.
This is because, despite the MOHW’s stated intention to restructure the domestic pharmaceutical industry around innovative new drugs through the drug price reform, there are no preferential policies in place for new drugs first developed by Korean pharmaceutical companies.
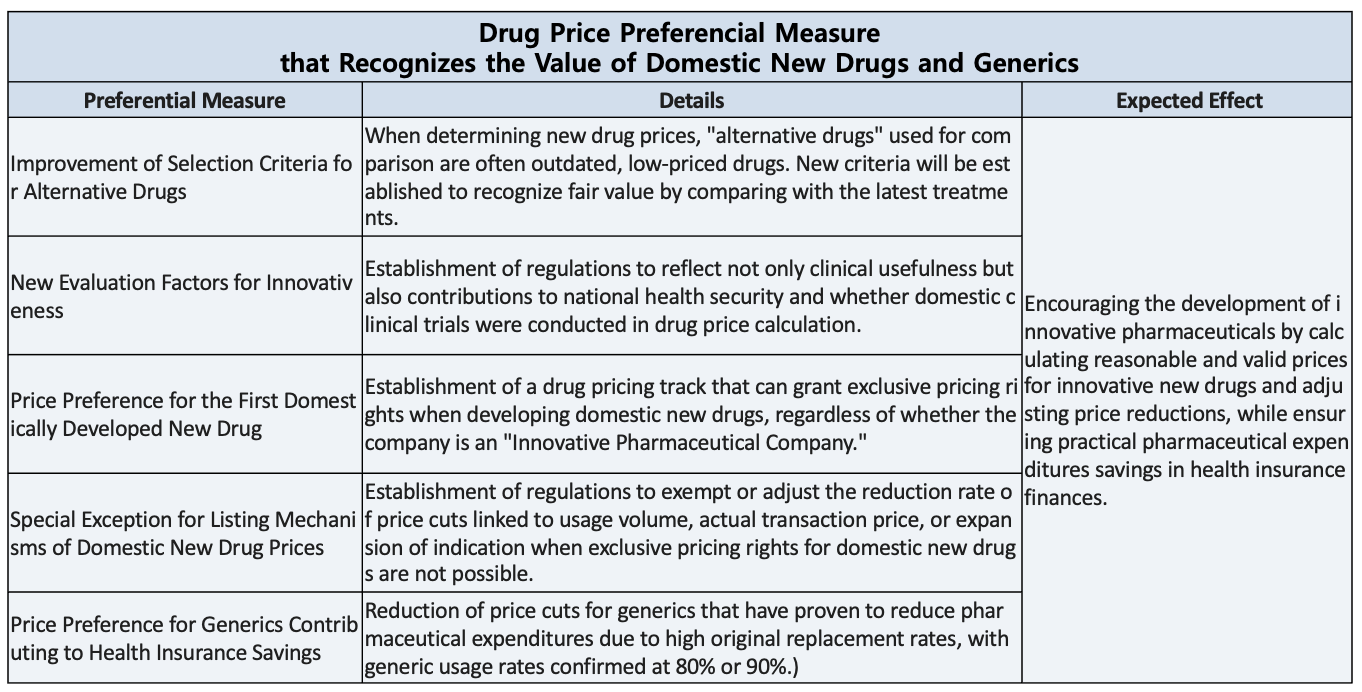
In fact, in response to requests from the National Assembly and the industry to introduce exclusive incentives for domestic new drugs, the MOHW has repeatedly stated that such measures must be carefully reviewed due to potential trade conflicts with countries such as the United States.
In contrast, Japan applies innovation and utility premiums to domestically developed drugs with proven clinical value, and also provides a priority introduction premium for new drugs approved for the first time in Japan.
The pharmaceutical industry is demanding that if preferential drug pricing (premiums) at the time of listing is realistically impossible, a track be created to grant them exceptional benefits when the post-listing price reduction mechanism is applied. In other words, they are asking that domestic new drugs be excluded from price-volume agreement negotiations, price reductions based on actual transaction prices, and price reductions due to expanded indications, or that the reduction rates be adjusted so that these drugs can derive tangible benefits.
Furthermore, they argued that the domestic pharmaceutical industry’s innovation could be supported by recognizing the value of generics, which have a high substitution rate for expensive original drugs, and lowering their price reduction rates, in line with the goal of saving national health insurance finances, which is the very reason for the existence of generics.
For generics with usage rates exceeding 80–90% in prescriptions, confirming clear contributions to NHI savings, the industry requested the government to explore proactive measures to lessen price reductions.
The National Assembly is also demanding the establishment of a clear differential compensation mechanism for pharmaceutical companies that have demonstrated contributions to the development of the pharmaceutical industry and investments in the value of new drugs, in order to foster the pharmaceutical and biotech industries.
Rep. In-soon Nam of the Democratic Party of Korea stated, “The current drug pricing system provides insufficient returns relative to R&D investment. A differentiated pricing system based on factors such as R&D investment ratio, successful new drug development, and global exports is needed, including expanded incentives at listing or exemptions and reductions in price cuts.
Lack of post-evaluation in pricing reform also an issue
The pharmaceutical industry also urged the MOHW to reflect on the historical trajectory of Korea’s drug pricing system and jointly set the future direction.
Korea established the framework of its pricing system with the introduction of the Positive List System (PLS) in 2006 and the price-volume agreement (PVA), and has since operated the system with incremental adjustments for nearly 20 years.
In November 2011, the government reinstated the joint bioequivalence testing system and implemented a blanket price cut in April 2012, applying a generic pricing rate of 53.55%. In 2020, it introduced differential pricing based on direct bioequivalence testing and DMF requirements, along with the 1+3 joint bioequivalence rule.
Then, in November last year, the MOHW presented a new reform plan, stating that distortions caused by the proliferation of generics under past policies had reached a point where they could no longer be ignored.
The MOHW’s rationale is that the problem has become entrenched, with pharmaceutical companies uniformly launching dozens to hundreds of generics containing the same active ingredient and focusing their efforts on sales and rebate competition for generics rather than innovative drugs, leaving the MOHW with no choice but to intervene.
While the domestic industry partially agrees with this assessment, it points out that the root cause of the current multi-product generic structure lies in past government policies on approvals and pricing.
In fact, when joint bioequivalence testing and across-the-board drug price cuts were implemented simultaneously in 2012, pharmaceutical companies sought to overcome declining profit margins by rushing into multi-product generic production. This triggered the first generic drug boom, resulting in a significant increase in the number of products despite short-term cost-saving effects.
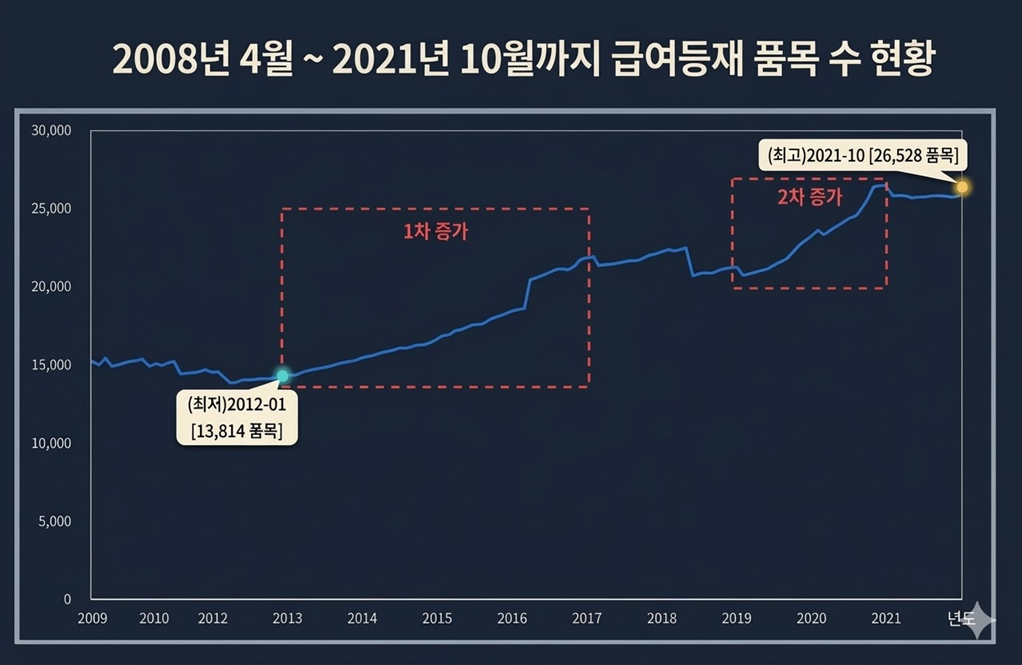
According to the MFDS, newly approved generics nearly doubled from 727 in 2012 to 1,283 in 2013, and further increased to 1,684 in 2014 and 1,914 in 2015. The number of reimbursed generics also doubled from 1,094 in 2012 to 1,717 in 2013 and 2,359 in 2015.
The 2020 reforms, including differential pricing based on criteria and restrictions on joint bioequivalence (1+3), triggered a second generic boom.
At the time of the blanket price cut, the MOHW promoted the policy as a way to elevate Korea’s pharmaceutical industry to a global level. However, in reality, it resulted in the unintended consequence of an explosive increase in the number of generic products.

ccordingly, academia and the pharmaceutical industry are calling for post-evaluation and reassessment of the MOHW’s past pricing policies aimed at reducing drug expenditure, curbing generic proliferation, fostering industry development, and promoting innovation.
Professor Jong-hyuk Lee of Chung-Ang University College of Pharmacy stated, “It has been 20 years since the introduction of the positive list system. The government’s direction on managing new drug reimbursement and generic pricing is not wrong, but while the goal of addressing excessive multi-product listings is valid, we need to analyze whether the current reform will actually lead to increased R&D investment or reduced NHI expenditure.”
Jae-hyun Lee, head of the Pharmaceutical Research Center at Sungkyunkwan University, added, “Both the 2012 blanket price cut and the current reform plan link generics and new drugs. While it is true that the government mobilizes various systems to achieve policy goals, it is necessary to evaluate and reflect on whether these systems were designed and implemented in accordance with their original intentions, and to make revisions accordingly.”
Director Lee added, “It is undesirable for policymakers to design a system in a way that expects secondary effects on new drugs through generic drug pricing or aims to achieve other outcomes, such as eliminating rebates as a side effect. If generic drug prices are to be adjusted, the system must remain faithful to its original intent by limiting reductions and surcharges to generics alone. It is contradictory to adopt a half-hearted approach that allows innovative pharmaceutical companies to have their generic drug prices cut by a smaller margin.”
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.







