- LOGIN
- MemberShip
- 2026-06-10 20:04:32
- Complete revision to the "Innovative Pharma Company" certification
- by Lee, Jeong-Hwan | translator Hong, Ji Yeon | 2026-03-27 08:32:58
The Ministry of Health and Welfare (MOHW) will make administrative move to encourage drug investment by raising the R&D expenditure requirement for "Innovative Pharmaceutical Company" certification.
The MOHW will improve administrative fairness by revising the disqualification criteria related to illegal rebates. Specifically, "rebate violations that concluded more than five years before the point of certification or renewal evaluation" will now be excluded from the review process. The new policy aims to address the irrationality of revoking certifications based on violations that occurred in the distant past. However, the proposed "points-based scoring system" for rebate-related cancellations will not be introduced.
Additionally, the certification track for foreign pharmaceutical companies will be newly established and revised. To encourage foreign firms to host research and production facilities in Korea, attract overseas capital, and engage in joint research and open innovation, the score weightings for these categories will be increased.
Transparency in the certification process will also be enhanced. The minimum passing score for certification (65 points) will be explicitly stated in the official notice, and companies that fail to gain certification will be formally notified of the specific reasons for their disqualification.
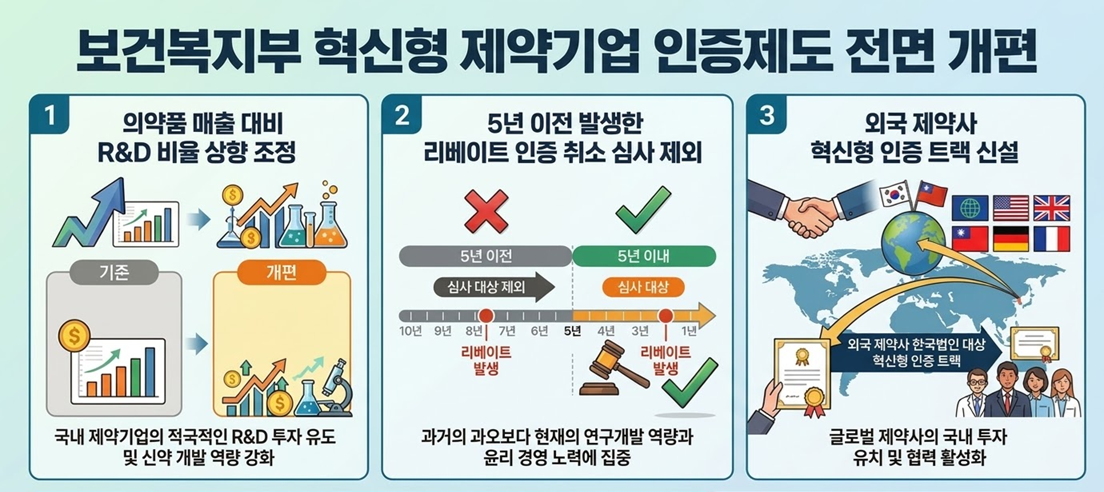
On the 26th, the MOHW issued a legislative and administrative notice regarding amendments to the Enforcement Decree, Enforcement Rules, and related notices of the "Special Act on Promotion and Support of Pharmaceutical Industry" (the Pharmaceutical Industry Act). The public opinion engagement remain open until May 6.
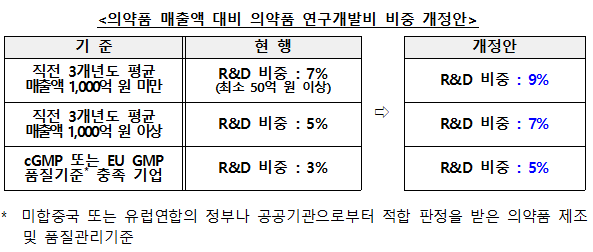
R&D Expenditure Ratio to Rise by 2%p
First, the R&D expenditure ratios required for the certification and renewal of Innovative Pharmaceutical Companies will be increased. While the ratio relative to pharmaceutical sales will rise by 2 percentage points across the board, the application of this new standard will be 'deferred for three years from the date of enforcement' to allow companies sufficient time to prepare.
Through this administrative move, the MOHW aims to encourage a continuous expansion of R&D investment among certified firms.
Furthermore, the system will now categorize firms into "Innovative Pharmaceutical Companies" and "Foreign Innovative Pharmaceutical Companies" to provide a regulatory system that accounts for the unique characteristics of multinational corporations. The track for foreign firms will be implemented immediately upon announcement.
For companies holding cGMP (current Good Manufacturing Practice) or EU GMP certification seeking renewal under the relaxed "R&D expenditure-to-sales" ratio, they must now submit evidentiary materials prepared within 3 years of the certification's expiration date.
This administrative move resolves the current lack of a specific timeframe for GMP-related documentation.
The amendments to Enforcement Rules will take effect on the date of announcement and are expected to apply to renewal applications starting in the second half of this year.
Revising 'Rebate Checking Policies' and 'Detailed Evaluation Criteria'
Regarding illegal rebates, the MOHW has decided to exclude violations that occurred more than 5 years before the certification or renewal review.
In cases where administrative appeals or lawsuits are filed, certification can be revoked within one year of the date of dismissal of the appeal or a final ruling against the company.
By maintaining the current revocation structure rather than switching to a points-based system, and excluding violations older than five years, the MOHW aims to eliminate the irrationality of penalizing firms for old actions. This addresses repeated parliamentary criticism that legal stability and predictability for pharmaceutical companies were undermined when certifications were revoked for ancient violations.
The current standards state that administrative actions for rebates under the Pharmaceutical Affairs Act or the Fair Trade Act that occurred five years before the certification review are excluded. However, if a lawsuit is filed challenging the administrative action, the date the judgment becomes final is treated as the date of the administrative action.
Consequently, cases have occurred in which "Innovative Pharmaceutical Company" certifications were revoked due to rebate violations that occurred a long time ago. Criticisms rose from the National Assembly that the predictability and legal stability for pharmaceutical companies are being undermined.
Furthermore, the detailed evaluation criteria for certification will also be improved and will be noted in the appendix.
The total score will be adjusted from 120 to 100 points, and the number of evaluation items will be reduced from 25 to 17.
Objectivity in certification standards will be enhanced by converting evaluation items, such as R&D investment, the number of clinical trials, and export volume, into quantitative indicators (4 out of 17 items).
Notably, a new category will be established to recognize excellence in social responsibility activities, such as the production and supply of medicines that help stabilize the supply chain.
For the Domestic Innovative Pharmaceutical Company certification standards, the score weightings for items such as partnership·collaboration activities, non-clinical·clinical trials, candidate development, and transparency in corporate management will be increased. Conversely, items such as research personnel, research/production facilities, and R&D strategies will be adjusted downward.
Establishment of Certification Standards for Foreign Innovative Pharmaceutical Companies
Furthermore, based on the types of Innovative Pharmaceutical Companies classified under the Enforcement Decree of the Pharmaceutical Industry Act, detailed certification review criteria for Domestic Innovative Pharmaceutical Companies and Foreign Innovative Pharmaceutical Companies will be separately regulated.
Foreign pharmaceutical companies will be permitted to choose and apply under either the Domestic Innovative Pharmaceutical Company certification standards or the Foreign Innovative Pharmaceutical Company certification standards.
The certification standards for Foreign Innovative Pharmaceutical Companies will have increased weightings for specific items to encourage foreign pharmaceutical firms to establish domestic research and production facilities, as well as to attract foreign capital, facilitate joint research, and promote open innovation.
Given the characteristics of foreign pharmaceutical companies, in which the global headquarters typically holds the technology and patents, the score weightings for items related to the development of non-clinical and clinical trial candidates and to pharmaceutical patent technology transfer performance will be reduced.
Finally, the MOHW will state the minimum passing score of 65 points and mandate that the reasons for failure be specified in notifications to companies.
These changes will be reflected starting with new and renewal applications in the second half of this year.
Meanwhile, the MOHW plans to systematically analyze the types and capabilities of Korean pharmaceutical and biotech companies and establish a "Strategy for Fostering National Pharmaceutical & Biotech-Industry" within this year.
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.







