- LOGIN
- MemberShip
- 2026-06-10 20:04:31
- Prices of top-priced generics could drop 32% upon pricing reform
- by Chon, Seung-Hyun | translator Alice Kang | 2026-03-18 09:11:33
The government’s announced tiered pricing system for generics is expected to function as a powerful mechanism that significantly lowers generic drug prices. Concerns have been raised that the faster exposure to the tiered pricing system compared to the current system could dampen the momentum for later-entering generics. It is estimated that the price of the 13th generic to enter the market will drop to half the current level, as the discount rate increases for products that fail to meet the highest-price criteria, such as those failing bioequivalence tests.
Analysis suggests that even if a product meets the highest-price criteria, if more than 13 products are launched simultaneously, a new price reduction mechanism, which applies an additional 15% price cut one year later, will be triggered, causing the price of the first generic to fall by 32% compared to current levels.
Tiered pricing system with 15% reduction applied from the 13th generic onward... Drug prices plummet due to stricter eligibility requirements
According to industry sources on the 16th, the Ministry of Health and Welfare presented a principle at the HIPDC subcommittee meeting held on the 11th that, under the price reform, the 13th generic would be subject to pricing. The price reduction rate applied to each tier would be 15%.
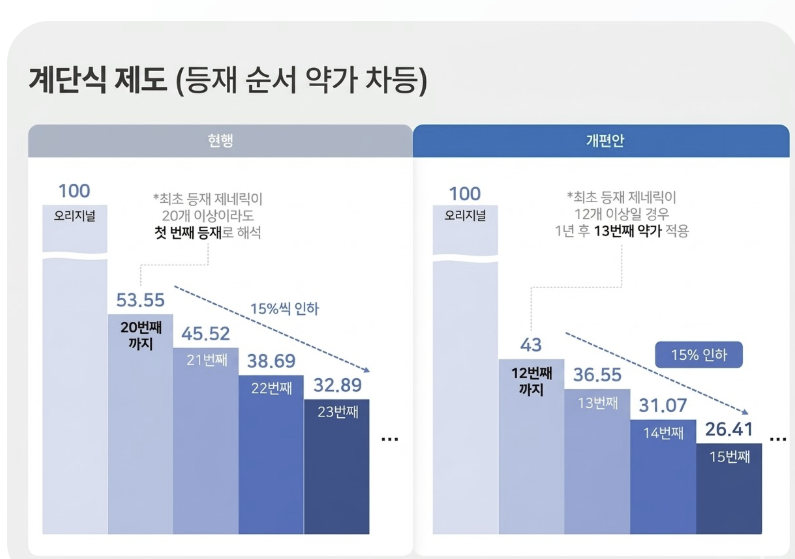
The tiered pricing system is structured such that the insurance ceiling price decreases on a monthly basis, the later a generic enters the market. Although it was abolished in 2012, the system was reinstated with the 2020 drug pricing reform. Under the current system, if there are more than 20 pre-listed products of the same formulation, the price of generics entering the market as latecomers is reduced by 15% at each step.
When reporting the drug pricing system reform to the Health Insurance Policy Deliberation Committee on November 11 of last year, the Ministry of Health and Welfare proposed a policy to grant the first generic a price reduced by 5 percentage points (p) from the calculated price starting from the 11th listing of the same formulation. This effectively means the Ministry presented a relaxed version of the stepped pricing system just four months after its initial report.
However, because the tiered system would now begin applying from the 13th generic instead of the 21st under the current system, products would be exposed much earlier to this additional price-cutting mechanism.
The impact of the tiered pricing system becomes even greater as the generic pricing benchmark itself is lowered. The government has proposed lowering the benchmark for calculating generic drug prices from the current 53.55% to the low to mid-40% range, a reduction of about 10 percentage points. If the generic drug pricing benchmark drops from 53.55% to 43%, the maximum generic price would be reduced by 19.7% in absolute terms.
For example, under the current drug pricing system, when the maximum generic price is KRW 53.55, the 21st generic’s price cannot exceed KRW 45.52, which is a 15% reduction. The price of 22nd and 23rd generics then drops to KRW 38.69 and KRW 32.89, respectively. The 24th generic would be KRW 27.95, and the 25th would be KRW 23.76, meaning drug prices decrease as generics enter the market later.
Under the revised pricing system, where the generic drug pricing benchmark is set at 43%, if the maximum price is KRW 43, the price of the 13th and 14th generics drops to KRW 36.55 and KRW 31.07, respectively, following a 15% step-down reduction. The price of the 15th generic drops to KRW 26.4. Under the current drug pricing system, the 15th generic drug is not subject to the tiered pricing structure and can maintain a price of 53.55%, but under the revised system, its price drops to less than half that level.
As the stricter maximum price requirements under the revised system will also be applied, the magnitude of price reductions under the tiered pricing system will become even greater.
Since July 2020, under the revised generic pricing rules, a generic product must both conduct its own bioequivalence study and use a registered active pharmaceutical ingredient to qualify for the top price. For each unmet requirement, the ceiling price is reduced by 15%. If both requirements are not met, the price falls by 27.75%. Applying that 15% reduction, the 53.55% premium benchmark falls to 45.52% if one requirement is unmet and to 38.69% if both are unmet.
According to the criteria introduced by the ministry after the 2020 pricing reform, “Even if a product meets both premium-price requirements, if there are already 20 or more listed identical products, starting from the 21st product, the price will be listed at 85% of the lower of the lowest price of the identical product or 38.69%.” The 38.69% rate is the result of two 15% reductions applied when a product failed to meet both of the highest-price criteria. (53.55% × 0.85 × 0.85)

Currently, the 21st generic drug,the first to be subject to the tiered drug pricing system, is priced at 32.86%, which is a 15% reduction from the 38.69% baseline. Compared to the highest price of 53.33%, this means the first generic drug subject to the tiered pricing system will see a 38.6% reduction. The prices of the 22nd and 23rd generic drugs will be reduced even further.
The Ministry of Health and Welfare plans to increase the reduction rate applied when the highest price requirement is not met under the revised drug pricing system from 15% to 20%. If the benchmark for the highest generic drug price is set at 43%, the calculation standard will be further lowered to 34.40% for generics that fail to meet one requirement, and to 27.52% for generics that fail to meet both requirements.
Under the revised drug pricing system’s tiered pricing structure, which applies the highest-price requirement, the ceiling price drops significantly starting with the 13th generic.
The price of the 13th generic is calculated to drop from 27.52%, the rate applied to generics failing to meet two highest-price requirements, to 23.39%, a reduction of 15%. Comparing the same 13th generic drug, the price under the current system is KRW 53.55, whereas under the revised system, it drops to roughly half that amount. The 13th and 14th generic drugs each decrease by 38.6% from the lowest price, falling to KRW 14.36 and KRW 8.8, respectively.
Under the revised drug pricing reform, the combined effects of shortening the sequence for applying the tiered pricing system, a 15% reduction upon tiered application, and a 20% price reduction for drugs that do not meet the highest-price requirement create a structure that effectively prevents late-entrant generics from entering the market.
If 13 or more highest-priced generics are listed, prices will be reduced by 15% after one year.
Even top-priced generics could see their prices cut a year later, depending on the number of products listed at the same time.
To prevent excessive competition when the first generics enter, the ministry plans to apply pricing standards equivalent to tiered cuts to generics whose listing causes the total number of identical products to exceed 13.
Even if a generic is listed within the first 12 products and initially receives the top price of 43%, if that product is among a group whose simultaneous entry causes the total to exceed 13, its price would be cut by 15% one year later.

For example, if 8 generic products are listed in January and receive prices in the low-to-mid 40% range, and another 8 generics are listed in February, those products would all be treated as being listed simultaneously starting from the 9th position and could receive the top price. However, because the additional eight February listings push the total number of identical products beyond 13, those products would, one year later, fall to the 85% tiered level.
Even the earliest listed generic could have its price cut by 15% one year later if 13 or more products enter simultaneously. In other words, even if it initially secures the top-price benchmark of 43%, it could later fall to 36.55% after one year. In that case, the price would be 31.75% lower than the current ceiling price.
Within the industry, this is being interpreted as the government effectively intending to allow only up to 12 generics. Under restrictions on joint development, only three products may participate in one bioequivalence study. Generics beyond three such groups would face such sharp price declines that their incentive to enter the market would effectively collapse.
One industry official noted, “Once the revised drug pricing system is implemented, late-entering generics will effectively be unable to turn a profit, so competition to secure the highest price by capturing the market first will inevitably intensify. Even if a company secures a leading position in the generic market, if there are many competing products, the tiered pricing mechanism will cause drug prices to fall, which could even lead to companies abandoning their plans to enter the generic market altogether.”
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.







