- LOGIN
- MemberShip
- 2026-06-11 00:25:05
- Big pharma companies report strong financial results
- by Chon, Seung-Hyun | translator Hong, Ji Yeon | 2026-02-12 06:38:47
Last year, major South Korean pharmaceutical and biotech companies reported robust earnings, driven by differentiated R&D capabilities in innovative drugs, biosimilars, and contract development and manufacturing (CDMO). Samsung Biologics and Celltrion set all-time highs, while traditional pharmaceutical firms maintained record-breaking performances based on their proprietary research.
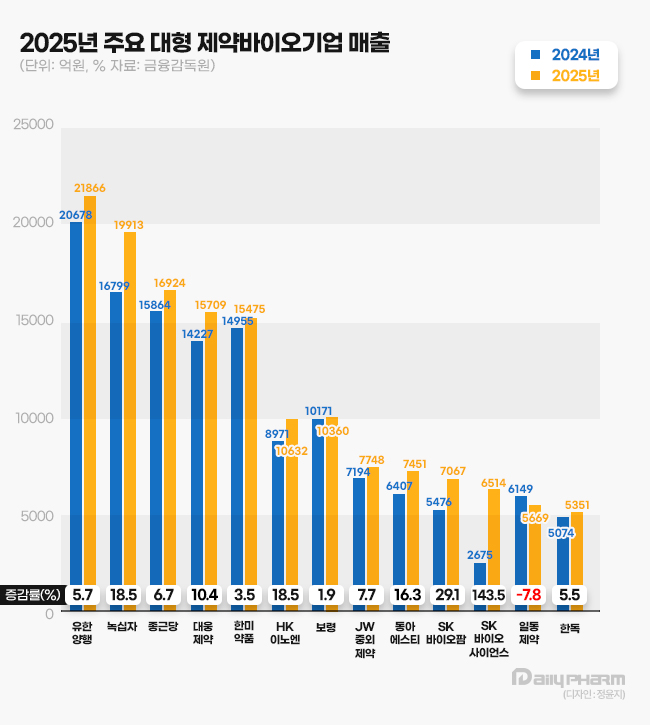
According to the Financial Supervisory Service on the 12th, 14 out of 15 leading domestic firms with annual revenues exceeding KRW 500 billion, including Yuhan Corporation, GC Biopharma, Daewoong Pharmaceutical, and Hanmi Pharmaceutical, showed sales growth compared with the previous year. 13 of these 15 firms reported increased operating profits, with the exceptions of Chong Kun Dang and Dong-A ST.
Samsung Biologics·Celltrion Continue Record Performance…Operating Profit Hike
Samsung Biologics and Celltrion have significantly accelerated their growth, widening the distance from traditional pharmaceutical companies.
Samsung Biologics and Celltrion have significantly accelerated their growth, widening the distance from traditional pharmaceutical companies through aggressive expansion and high-margin business models. Samsung Biologics recorded an unprecedented operating profit of KRW 2.07 trillion, a 56.6% increase, on revenue of KRW 4.56 trillion. Its operating profit margin reached 45.4%.
Samsung Biologics primarily focuses on Biopharmaceutical Contract Manufacturing (CMO) and Contract Development (CDO). The company’s growth was increased by the stable, full-capacity operation of Plants 1-3, alongside the successful launch of Plant 4.
Since its inception, Samsung Biologics has steadily increased its capacity from Plant 1 (30,000L), Plant 2 (155,000L), and Plant 3 (180,000L) to Plant 4, which stands as the world’s largest single facility at 240,000L. With the 180,000-liter Plant 5 commencing operations in April last year, Samsung Biologics’ total production capacity has expanded to 785,000 liters.
The company's performance exceeded the previous year's consolidated figures, even after excluding its biosimilar subsidiary, Samsung Bioepis.
Following a corporate spin-off in November, Samsung Biologics now focuses strictly on the CDMO business, while the newly formed Samsung Epis Holdings oversees biosimilars and new drug development.
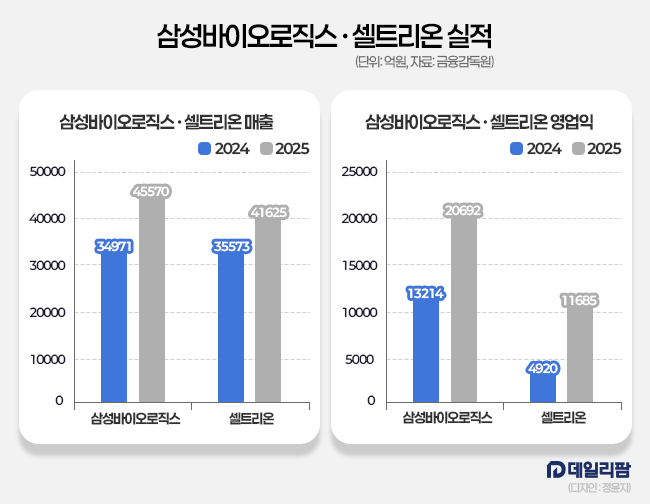
Celltrion also recorded an annual operating profit of 1.17 trillion KRW, a 137.5% year-on-year increase. Revenue grew by 17.0% to exceed 4.16 trillion KRW for the first time in the company’s history, yielding an operating profit margin of 28.1%.
Celltrion has obtained marketing authorizations in Europe and the United States for a robust portfolio, including Remsima, Herzuma, Truxima, Remsima SC, Zymfentra, Yuflyma, Vegzelma, Steqeyma, Stoboclo·Osenvelt, Omlyclo, AVTOZMA, and Eydenzelt.
While existing products such as Remsima, Truxima, and Herzuma maintained stable growth, Celltrion's recently launched biologics, including Remsima SC, Yuflyma, Vegzelma, Steqeyma, Stoboclo·Osenvelt, Omlyclo, AVTOZMA, and Eydenzelt, were classified as new revenue drivers. All of these products reached record-high annual sales.
Celltrion has secured 25 approvals across Europe and the United States. Specifically, Remsima, Herzuma, Truxima, Remsima SC, Zymfentra, Yuflyma, Vegzelma, Steqeyma, Stoboclo·Osenvelt, Omlyclo, AVTOZMA, and Eydenzelt have all received regulatory green lights in these regions.
Remsima recorded sales of KRW 1.0495 trillion last year. Additionally, Remsima SC, Truxima, Yuflyma, Vegzelma, Herzuma, Steqeyma, and Zymfentra each surpassed KRW 100 billion in annual revenue.
Traditional Pharmaceutical Companies Show Record Sales...In-House Developed Drugs Drive Performance
Major traditional pharmaceutical companies also posted record-breaking financial results, led by the success of their proprietary new drugs.
Companies such as GC Biopharma, Daewoong Pharmaceutical, and HK inno.N saw both revenue and operating profit rise by more than 10%, driven by the strong performance of medicines developed through their accumulated R&D expertise.
GC Biopharma reported an operating profit of KRW 69.1 billion last year, a 115.4% increase year-on-year, while revenue grew 18.5% to KRW 1.9913 trillion. This represents the company's largest annual revenue to date.
Strong U.S. sales of the blood product Alyglo significantly bolstered performance. Alyglo's revenue in the U.S. market reached $106 million (KRW 151.1 billion) last year, a 211% increase from the previous year.
Approved by the U.S. Food and Drug Administration (FDA) in December 2023, Alyglo is a liquid immunoglobulin G (IVIG-SN 10%) purified from human plasma. It is indicated for the treatment of primary humoral immunodeficiency (PI), such as congenital immunodeficiency and immune thrombocytopenia. Alyglo is the first blood product developed by a South Korean company to enter the U.S. market.
GC Biopharma commenced full-scale sales in July 2024, following the initial shipment of Alyglo.
Alyglo's U.S. sales reached $106 million (KRW 151.1 billion) last year, growing 211% year-on-year. GC Biopharma initiated full-scale commercialization after shipping the first batch in July 2024 and surpassed $100 million in just its third year of entering the U.S. market.
Daewoong Pharmaceutical's operating profit rose 33.0% to KRW 196.8 billion last year, with revenue increasing 10.4% to KRW 1.5709 trillion. This marks the fifth consecutive year since 2021 that the company has broken its own records for both revenue and operating profit.
According to the pharmaceutical market research firm UBIST, prescription sales for Fexuclue reached KRW 90 billion last year, a 10.6% increase from the previous year. Fexuclue is a potassium-competitive acid blocker (P-CAB) indicated for gastroesophageal reflux disease (GERD). It received marketing authorization in December 2021 and began full-scale sales in July 2022 following its addition to the National Health Insurance drug reimbursement list.
Envlo, the 36th domestically developed new drug, saw its prescription sales rise 11.7% to KRW 11.8 billion last year. Envlo is an SGLT-2 inhibitor for diabetes, the first of its kind developed by a domestic pharmaceutical company. It received domestic approval in late 2022 and launched in May 2023.
The botulinum toxin Nabota recorded KRW 228.9 billion in sales last year, up 19.0% from the prior year. Nabota's export performance grew 23% year-on-year, driven by strengthened partnerships in North America and expanded supply to emerging markets, including South America and the Middle East. Nabota received FDA approval in 2019 through a partnering company, Evolus.
HK inno.N surpassed the KRW 1 trillion milestone for the first time, recording revenue of KRW 1.0631 trillion, an 18.5% increase. Operating profit rose 25.7% to KRW 110.9 billion.
K-CAB, a new drug for GERD, saw its annual prescription sales reach KRW 217.9 billion, up 10.6% year-on-year. Authorized in 2018 as South Korea's 30th new drug, K-CAB is a P-CAB class treatment. After surpassing KRW 100 billion in prescriptions in 2021, just three years post-launch, it has maintained the 100-billion-won level for four consecutive years, setting a new record by exceeding KRW 200 billion last year.
HK inno.N's performance was also supported by co-promotion agreements for Pfizer's COVID-19 vaccine and Roche's oncology drug Avastin.
Profitability Gains for Hanmi, Yuhan, JW Pharm, and Boryung... SK Biopharm and SK Bioscience Benefit from New Drugs and M&A
Hanmi Pharmaceutical, Yuhan Corp, JW Pharmaceutical, and Boryung significantly improved profitability through their in-house new drugs.
Hanmi Pharmaceutical's operating profit rose 19.3% to KRW 257.8 billion last year, while revenue increased 3.5%. Both figures represent all-time highs. Its operating profit margin stood at 16.7%, the highest among traditional pharmaceutical firms.
The new combination drug Rosuzet recorded KRW 227.9 billion in outpatient prescription sales, an 8.4% increase from the previous year. Rosuzet is a combination therapy of rosuvastatin and ezetimibe. In 2024, Rosuzet became the first domestically developed drug to lead the overall market with KRW 210.3 billion in sales and has maintained the top position for two consecutive years.
Last year, Hanmi Pharmaceutical's total outpatient prescription sales reached KRW 1.0151 trillion, a 2.0% increase, securing the top market position. Hanmi has held the lead in prescription performance for eight consecutive years since 2018 and is the first pharmaceutical company (domestic or foreign) to exceed KRW 1 trillion in annual prescription sales.
Beijing Hanmi Pharmaceutical, the company's Chinese subsidiary, recorded revenue of KRW 402.4 billion and an operating profit of KRW 77.7 billion, surpassing the KRW 400 billion mark for the first time since its founding. This was driven by the normalization of local distribution inventory and increased sales of respiratory disease treatments.
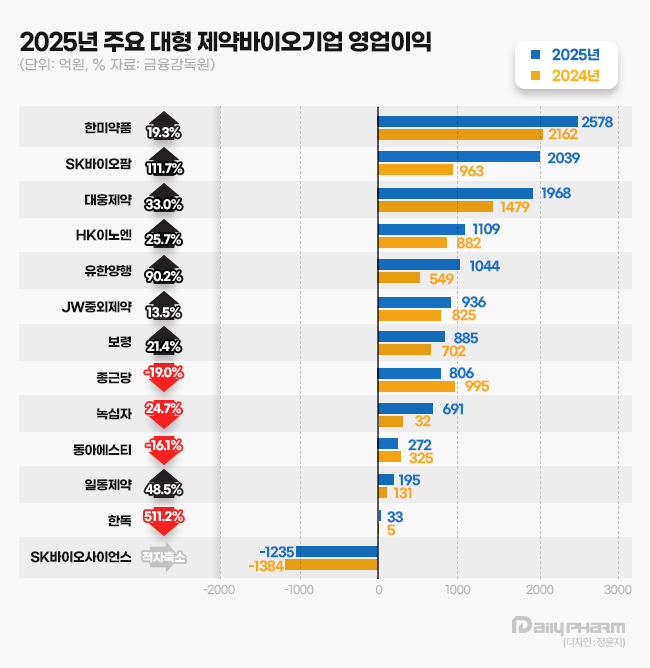
Yuhan's operating profit surged 90.2% to KRW 104.4 billion last year, while revenue rose 5.7% to KRW 2.1866 trillion. This marks the first time the company’s operating profit has exceeded KRW 100 billion, surpassing the previous high of KRW 97.8 billion set in 2016.
Significant licensing income (milestone payments) contributed to this growth. Yuhan Corp recognized KRW 104.1 billion in licensing revenue last year, marking the second consecutive year of exceeding KRW 100 billion in technology-related inflows, following KRW 105.3 billion in 2024.
In the fourth quarter of last year, KRW 70.3 billion in licensing revenue was generated. The milestones are from the Chinese market entry of the oncology drug Leclaza.
In August last year, China's National Medical Products Administration (NMPA) approved Leclaza, in combination with Johnson & Johnson's Rybrevant, as a first-line treatment for adults with locally advanced or metastatic non-small cell lung cancer (NSCLC) harboring EGFR Exon 19 deletions or Exon 21 L858R substitution mutations. Yuhan Corp received a $45 million (KRW 69 billion) milestone payment from Janssen Biotech in Q4 for achieving this stage.
JW Pharmaceutical's operating profit grew 13.5% to KRW 93.6 billion last year, with revenue increasing 7.7% to KRW 774.8 billion.
The Livalo family, based on pitavastatin for dyslipidemia, has shown remarkable growth. Livalo (monotherapy) recorded KRW 84.8 billion, Livalozet recorded KRW 101.0 billion, and Livalo V recorded KRW 3.5 billion. Combined sales of the three Livalo products reached KRW 189.3 billion, a 16.9% increase year-on-year.
Livalozet, a combination of pitavastatin and ezetimibe, has maintained a high growth trajectory since its launch in October 2021. Livalozet posted sales of KRW 64.4 billion in 2023 and KRW 76.2 billion in 2024; last year, it continued its strong performance, exceeding the KRW 100 billion mark just 4 years after launch.
Hemlibra, a hemophilia treatment, saw its revenue expand 48.5% to KRW 72.6 billion last year. Hemlibra is a routine prophylactic treatment for Hemophilia A caused by Factor VIII deficiency. Sales skyrocketed after the drug was covered by health insurance for 'Hemophilia A patients aged 1 year or older without Factor VIII inhibitors' starting in May 2023.
Boryung's revenue grew modestly by 1.9% to KRW 1.0360 trillion, while its operating profit jumped 21.4% to KRW 85.5 billion.
Boryung improved its profitability by "maximizing self-produced product capabilities." As the proportion of in-house manufactured products—which offer better cost-of-goods margins, increased, operating profit improved. Product revenue refers to sales derived from items a company manufactures itself. Last year, Boryung's self-produced product revenue rose 11.5% to KRW 550.3 billion. In the fourth quarter, product revenue hit an all-time high of KRW 148.4 billion, up 16.8% year-on-year.
Profitability was further bolstered as Boryung transitioned and began producing original drugs, such as Gemzar, Zyprexa, and Alimta, in-house. The steady growth of core businesses, including the Kanarb family and oncology treatments, drove the company's overall expansion.
SK Biopharmaceuticals and SK Bioscience saw significant performance improvements driven by new drug success and M&A activity.
SK Biopharmaceuticals' operating profit expanded 111.7% to KRW 203.9 billion, while revenue grew 29.1% to KRW 706.7 billion.
U.S. sales of the epilepsy drug Xcopri rose 43.7% to KRW 630.3 billion. Xcopri (cenobamate) is prescribed for adults with partial-onset seizures. SK Biopharmaceuticals managed the entire process from initial development to FDA approval independently, receiving authorization in November 2019. Since May 2020, it has been sold directly through SK Life Science, the company's U.S. subsidiary. Xcopri surpassed KRW 100 billion in 2022, with sales of KRW 169.2 billion, and has continued its steep annual growth.
SK Bioscience's revenue surged 143.5% to KRW 651.4 billion last year. Revenue jumped significantly as the financial results of IDT Biologika, a German CDMO acquired in 2024, began to be reflected in the consolidated statements.
SK Bioscience acquired IDT Biologika in October 2024. Through a wholly owned German subsidiary, it purchased a 60% stake in IDT Biologika from the Klocke Group.
Last year, IDT Biologika recorded revenue of KRW 465.7 billion, a 17% increase year-on-year. IDT Biologika accounted for more than 70% of SK Bioscience's total revenue. While sales had dropped sharply after the end of the COVID-19 pandemic, the M&A strategy successfully offset the revenue gap.
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.







