- LOGIN
- MemberShip
- 2026-05-09 22:18:31
- Record number of new homegrown drugs released this year
- by Chon, Seung-Hyun | translator Alice Kang | 2021-12-31 05:51:47
Domestic pharmaceutical and biopharmaceutical companies this year had received approval for four new drugs.
This is the most amount of new drugs released in a single year since the first homegrown drug was introduced in 1999.
Major companies with an annual sales record of 1 trillion won such as Yuhan Corp, Celltrion, Hanmi Pharamcetucial, and Daewoong Pharmaceutical have made this performance.
According to the Ministry of Food and Drug Safety on the 31st, Daewoong Pharmaceutical’s gastroesophageal reflux disease (GERD) treatment ‘Fexclu,’ which contains fexuprazan, was approved as a new drug on the 30th.
Daewoong Pharmaceutical made this achievement in 14 years since it first started the development of Fexclu in 2007.
Fexclu is a potassium-competitive acid blocker (P-CAB) GERD treatment.
It has a faster onset of action than proton pump inhibitors (PPIs) and has the advantage of being able to be taken regardless of meal ingestion and the longer-lasting effect.
These types of drugs had quickly settled in the market since HK inno.N introduced its first P-CAB type of new drug ‘K-CAB’ in 2018.
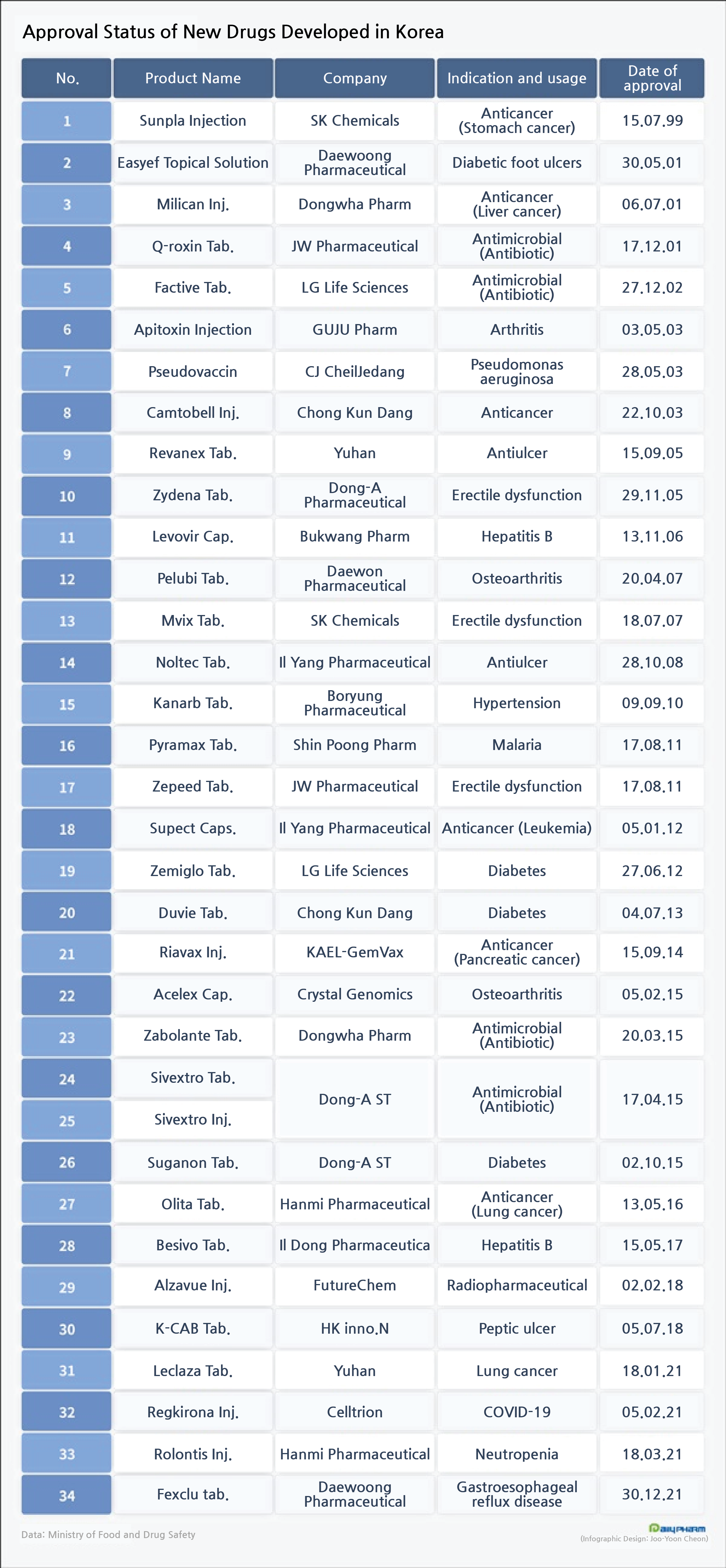
First was Yuhan Corp’s anticancer drug ‘Leclaza’ in January, followed by Celltrion’s COVID-19 treatment ‘Regkirona’ in February and March, and Hanmi Pharmaceutical’s neutropenia treatment ‘Rolontis.’ In January, Leclaza received conditional approval for the treatment of patients with advanced non-small-cell lung cancer who have a mutation in a specific gene (EGFR) who have previously received lung cancer treatment.
Leclaza is a targeted anticancer therapy that inhibits the signal transduction that is involved in lung cancer cell proliferation and growth.
It is an oral tyrosine kinase inhibitor (TKI) that has high selectivity against the EGFR T790M resistant mutation.
It showed superior efficacy and tolerability against lung cancer patients with brain metastasis as it can pass through the blood-brain-barrier (BBB).
Celltrion’s COVID-19 antibody treatment ‘Regkirona’ received conditional approval in February.
Regkirona Inj.
is a recombinant neutralizing antibody produced by selecting a neutralizing antibody gene in a COVID-19 convalescent patient's blood and inserting the selected gene into a host cell that is cultured for large-scale production.
Regkirona is the first locally developed COVID-19 treatment and the third COVID-19 antibody treatment in the world to be verified by the regulatory authority.
It received marketing approval based on its Phase III trial results in August and its indication was extended to the treatment of high-risk adult patients with mild and moderate symptoms who have been confirmed with COVID-19 according to the provisional pathway.
In March, Hanmi Pharmaceutical’s Rolontis Pre-filled Syringe inj.
was approved as a new locally developed new drug.
Rolontis uses recombinant technology to connect a specific protein to a human’s granulocyte-colony stimulating factor (G-CSF) analog to increase the sustainability of its effect.
It is used to treat chemotherapy-induced neutropenia.
It binds to the G-CSF receptor to stimulate neutrophil production.
This year, domestic pharmaceutical and biopharmaceutical companies have broken the record and received approval for the largest number of homegrown new drugs in a single year, and in three years since HK inno.N received approval for K-CAB in 2018.
That these new drugs were released after a long period of time by major companies that recorded annual sales exceeding 1 trillion won - Yuhan Corp, Celltrion, Hanmi Pharmaceutical, and Daewoong Pharmaceutical – is also being considered significant.
Yuhan Corp released its second novel drug this year in 16 years since ‘Revancex’ in 2005.
Regkirona was Celltrion’s first new drug.
Celltrion had focused on biosimilars, but with the spread of COVID-19 in earnest, the company had entered into the treatment development business and succeeded in the commercialization of the treatment within a year.
Rolontis is the first new biologic and the only new drug Hanmi Pharmaceutical succeeded in commercializing and is in sale.
Hanmi Pharmaceutical had received approval for its NSCLC treatment ‘Olita’ in 2016 but announced the discontinuation of its development after three years, based on the strategic judgment that stopping the development is more beneficial than bearing the cost of Phase 3 clinical trial.
Daewoong Pharmaceutical succeeded in developing a new drug in 20 years since Easyef in 2011.
It is evaluated that the potential of success of Fexclu is high as the same class drug K-CAB’s marketability was recognized in the industry.
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.
- [Reporter's View] Contradiction of "K-passing" and a new drug powerhouse
- Reporter's view | Lee, Jeong-Hwan
- [Reporter’s View] Medical Devices Act binds payment within 6 months
- Reporter's view | Hwang, byoung woo






