- LOGIN
- MemberShip
- 2026-05-09 22:18:36
- Montelukast sales 22%↓ in 2 years and adds impurity issue
- by Kim, Jin-Gu | translator Alice Kang | 2022-01-25 05:55:51
The market for the montelukast ingredient used to treat allergic rhinitis and asthma has contracted for two consecutive years.
The reduction is analyzed to be due to the combined effect of the US FDA’s side effect warning request for the ingredient in 2020 and the prolonged COVID-19 crisis.
Industry eyes are on how the impurity issue that recently emerged would additionally affect the market.
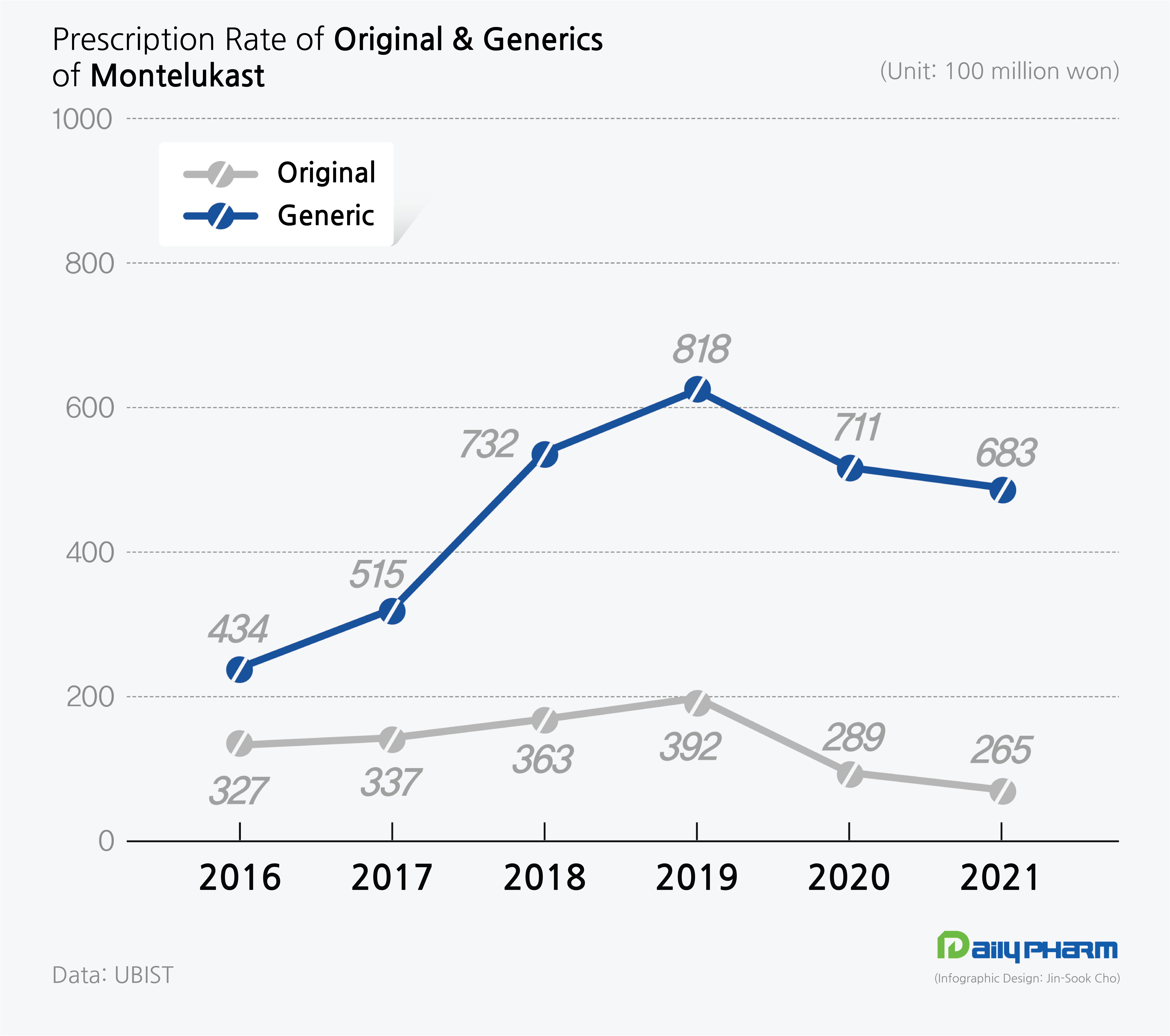
◆Conflicting results by product… Singulair ↓ 32% vs Monterizine ↑ 15% According to the industry research institution UBIST on the 25th, outpatient prescription sales of the allergic rhinitis and asthma treatment market for montelukast was ₩94.8 billion last year.
The market had risen from ₩76.1 billion in 2016 to 121 billion in 2019 but had seen a decline for two consecutive years since.
Compared to 2019 when the market expanded to its greatest, the market had shrunk 22% over the recent 2 years.

The original drug is Organon Korea’s Singulair.
In Korea, MSD Korea received approval for the original drug in 2000, and around a hundred domestic pharmaceutical companies are selling generic versions with the same ingredient.
Most of the key products were unable to avoid the reduction in their prescription amount.
Sales of the market lead Singulair shrunk 32% from ₩39.2 billion in 2019 to ₩26.5 billion last year.
Other montelukast drugs also saw a double-digit reduction in sales.
HK Inno.N’s ‘Lukio’ dropped 31% (₩12.8 billion→₩8.8 billion), Hanmi Pharmaceutical’s ‘Montezal’ dropped 35% (₩7.3 billion→₩4.7 billion), Hutec Korea Pharmaceutical’s ‘Singuldown’ dropped 26% (₩5.3 billion→₩3.9 billion), Daewon Pharmaceutical’s ‘Singuluka’ dropped 42%(₩3.1 billion→1.5 billion).
On the other hand, prescription of some products has increased greatly.
Sales of Hanmi Pharmaceutical’s ‘Monterizine’ increased 15% from ₩8 billion to ₩9.3 billion in the same period.
Monterizine is a combination of montelukast and a third-generation antihistaminic agent ‘levocetirizine.’ It is the only combination drug sold in Korea.
The FDA required a ‘Black box warning’ on montelukast products in March 2020.
The black box warning is the FDA's most stringent warning for drugs and medical devices in the market.
In particular, the FDA strongly advised health care providers to avoid prescribing montelukast to patients with mild allergic rhinitis, as it is the FDA’s judgment that the risk outweighs the benefits for mild patients.
According to the FDA, serious adverse reactions, including suicide, have been reported with the use of Singulair since 2008.
Some of these side effects occurred while the patients were treated with montelukast and disappeared after discontinuing the drug.
The prolonged COVID-19 incidence had also reduced the prescriptions.
The pediatric clinics have been one of the areas most affected by the COVID-19 crisis.
Since most patients with allergic rhinitis and asthma are children and adolescents, the prolonged COVID-19 crisis has aggravated the reduction in prescriptions of montelukast.
◆MFDS orders NDPA impurity tests…rises as a ‘new variable’ in market The industry has also been paying attention to the impurity issue that had arisen recently.
The Ministry of Food and Drug Safety had recently ordered pharmaceutical companies to conduct an investigation into their montelukast ingredient and finished products for impurities and submit the reports by April 25th.
This precautionary measure was issued after the ministry received safety information that N- nitrosodipropylamine (NDPA) was detected in the API montelukast.
The NDPA is a new nitrosamine impurity that has been identified.
Since 2018, two types of nitrosamine impurities, - ‘N-nitrosodimethylamine (NDMA)’ and ‘N-nitrosodiethylamine (NDEA)’ were detected in valsartan, ranitidine, and nizatidine.
The MFDS predicts that NDPA occurred in the API manufacturing process of montelukast.

Those who believe that impurity will not affect the market are focusing on the fact that such impurity issues have risen every year.
After the valsartan incident in 2018, impurities were consecutively detected in ranitidine, nizatidine, metformin, losartan, varenicline, etc., building resistance among frontline clinics and hospitals regarding the issue.
The government had also initially suspended the sale of all items, but recently ordered only specific lot numbers to be recalled.
Others who have opposing views argue that it is too soon to predict that the impurity concern will not be significant, given that the montelukast are mainly prescribed to children and adolescents, unlike other items.
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.
- [Reporter's View] Contradiction of "K-passing" and a new drug powerhouse
- Reporter's view | Lee, Jeong-Hwan
- [Reporter’s View] Medical Devices Act binds payment within 6 months
- Reporter's view | Hwang, byoung woo






