- LOGIN
- MemberShip
- 2026-06-10 19:09:25
- Company
- Korean firms circumvent 1 patent on neuropathic pain drug Taleaje
- by Kim, Jin-Gu May 29, 2026 09:14am
- Generic drug makers have succeeded in circumventing the salt and composition patent protecting the neuropathic pain treatment Taleaje Tab (mirogabalin). If they also succeed in bypassing the formulation patent they are separately challenging, they may be able to move the generic launch timing forward to June 2031.According to the pharmaceutical industry on the 28th, the Korean Intellectual Property Trial and Appeal Board ruled in favor of Huons, Dong-A ST, and JW Pharmaceutical in their passive scope confirmation trial against Daiichi Sankyo regarding Taleaje’s salt and composition patent (10-2142257).Taleaje is protected by 3 patents: a substance patent (10-1335784) expiring in June 2031, a salt and composition patent expiring in April 2034, and a formulation patent (10-2425821) expiring in 2036.Generic companies filed passive scope confirmation trials against the salt/composition patent in May last year. In addition to the 3 companies that won the first ruling, Kyongbo Pharmaceutical, Samjin Pharmaceutical, BC World Pharm, Dongwha Pharm, Daewoong Pharmaceutical, and HK inno.N also filed similar challenges. These companies additionally challenged the formulation patent as well.Among the challengers, BC World Pharm, Dongwha Pharm, and HK inno.N voluntarily withdrew their trials and exited the patent challenge race. The remaining 6 companies continue their challenges. Following the recent favorable ruling, the industry expects KyungDong Pharmaceutical, Samjin Pharmaceutical, and Daewoong Pharmaceutical to succeed in bypassing the salt/composition patent.If they additionally succeed in circumventing the formulation patent following their success with the salt and composition patents, the generic launch timing could be advanced to June 2031 when the substance patent expires.Taleaje is used for neuropathic pain treatment. Daiichi Sankyo received approval for four dosage strengths (2.5mg, 5mg, 10mg, and 15mg) in January 2020.However, the product failed to secure coverage under the National Health Insurance. It is currently being sold as a non-reimbursed medication. As of 2024, its sales revenue stood at approximately KRW 3.7 billion. Given that the product generates significant sales even without insurance coverage, analysts believe it is well worth the challenge for generic drug manufacturers.If companies challenging the patent successfully obtain generic approval and secure insurance coverage, they are expected to compete in the neuropathic pain treatment market against pregabalin (brand name Lyrica) and gabapentin (brand name Neurontin).Pregabalin and gabapentin are already covered by national health insurance and are reported to form a market worth KRW 200 billion annually. Additionally, potential competition exists with COX-2 inhibitor analgesics such as celecoxib (brand name Celebrex) and SNRI antidepressants such as duloxetine (brand name Cymbalta).
- Company
- Amvuttra listed for reimbursement in Korea
- by Son, Hyung Min May 28, 2026 10:28am
- The treatment landscape for hereditary transthyretin amyloid polyneuropathy (hATTR-PN), a rare inherited disease, is beginning to show signs of change.Given the nature of the disease, which takes several years from symptom onset to diagnosis and leads to rapid functional decline, the importance of early diagnosis and intervention is growing. Expectations are also rising for improved treatment access following reimbursement coverage for the RNA interference (RNAi)-based therapy ‘Amvuttra (vutrisiran).’On the 27th, Medison Pharmaceuticals held a press conference at the Plaza Hotel in Jung-gu, Seoul, to commemorate the domestic launch of Amvuttra for the treatment of hATTR-PN.(From left: Professor Jeeyoung Oh (Department of Neurology, Konkuk University Hospital), and Professor Gyeongmo Sohn (Department of Neurology, Haeundae Paik Hospital)Hereditary transthyretin amyloid polyneuropathy (hATTR-PN) is a rare disease in which structurally abnormal transthyretin (TTR) proteins transform into amyloid forms and accumulate in various organs, including peripheral nerves, autonomic nerves, and the heart, causing nerve damage and organ dysfunction.This disease is characterized by a complex combination of systemic symptoms, including not only peripheral sensory and motor neuropathy but also autonomic dysfunction, cardiovascular abnormalities, and gastrointestinal symptoms. A substantial number of Korean patients reportedly present with mixed phenotypes involving both neurologic and cardiac symptoms.Delayed diagnosis is considered one of the biggest challenges. Early symptoms such as numbness in the hands and feet, sensory abnormalities, and gastrointestinal symptoms are relatively nonspecific and are often mistaken for other diseases. In Korea, the average time from symptom onset to diagnosis is reported to be 3.7 years.Professor Gyeongmo Sohn of Haeundae Paik Hospital said, “hATTR-PN often begins with early sensory abnormalities, gastrointestinal problems, or weakness of finger muscles. It is important to recognize so-called ‘red flags’ such as family history, vitreous opacity, carpal tunnel syndrome (CTS), and autonomic symptoms at an early stage.”He continued, “Autonomic symptoms such as dizziness caused by orthostatic hypotension, gastrointestinal motility disorders, and sexual dysfunction often appear even before motor nerve damage occurs. Early suspicion and diagnosis have a significant impact on patient prognosis.”The disease also progresses rapidly. According to Professor Sohn, the median survival after diagnosis for hATTR-PN patients is approximately 4.7 years, shortening to about 3.4 years when cardiomyopathy is present.RNAi-based Amvuttra added to reimbursement list… “Goal is to suppress disease progression”Treatment of hATTR-PN in Korea broadly involves gene silencers and protein stabilizers. Currently approved treatments include the RNAi-based gene silencer Amvuttra and Pfizer’s TTR stabilizer Vyndamax (tafamidis).Amvuttra is a therapy that suppresses the production of abnormal TTR proteins via RNA interference. It is considered a disease-modifying treatment because it fundamentally lowers production of the disease-causing protein and slows disease progression. Another key feature is that it can be administered as a subcutaneous injection once every three months.After receiving Korean approval in November 2024, Amvuttra additionally gained approval in March this year for reducing cardiovascular death, hospitalization, and urgent heart failure visits in adult patients with wild-type or hereditary transthyretin amyloid cardiomyopathy (ATTR-CM).Furthermore, as of April 1, health insurance coverage was extended to include Stage 1 patients with hATTR-PN for whom Vyndamax was insufficiently effective or difficult to administer, as well as all Stage 2 patients regardless of prior treatment status.Amvuttra’s clinical efficacy was confirmed through the global Phase III HELIOS-A study. This study was conducted on 164 hATTR-PN patients across 22 countries.Trial results showed that at 18 months, the Amvuttra treatment group exhibited a 0.46-point reduction in the Modified Neuropathy Impairment Score +7 (mNIS+7) compared to baseline, whereas the external placebo control group showed a 28.1-point increase.Improvements were also observed in the quality of life and physical function. The treatment group showed statistically significant improvements in the Norfolk Quality of Life-Diabetic Neuropathy (Norfolk QoL-DN) score compared to the external placebo group, and preservation of function was also seen in the 10-meter walk test assessing ambulatory ability.Professor Jeeyoung Oh, Department of Neurology at Konkuk University Hospital, said, “Amvuttra has demonstrated data showing improvements in neurologic function and quality of life in clinical studies. Because both physicians and patients have long faced significant unmet needs due to limitations in treatment accessibility, we hope reimbursement coverage will allow patients to receive treatment benefits at more appropriate stages.”
- Company
- Access to later-line therapy for kidney cancer remain unchanged
- by Son, Hyung Min May 28, 2026 10:27am
- Although treatment for advanced renal cell carcinoma (kidney cancer) is rapidly shifting toward immuno-oncology-based combination therapies, Korea’s treatment environment still lags behind global guidelines.In particular, even after immunotherapy combinations became established as first-line standard treatments, access to second-line therapies following disease progression remains limited. As a result, advances in treatment outcomes are not being fully translated into continuity of care for actual patients.On the 26th, Ipsen Korea held a press conference at the JW Marriott Hotel in Seocho-gu, Seoul, in celebration of World Kidney Cancer Day in June, highlighting the current treatment environment and patient access challenges in renal cell carcinoma. At the event, quality-of-life issues faced by kidney cancer patients and their families, along with the limitations of the domestic second-line treatment environment following immunotherapy, were presented as key topics.In Ho Kim, a professor of Medical Oncology at Seoul St. Mary’s Hospital, explained, “Renal cell carcinoma is one of the cancer types that has seen the most rapid advancements in treatment over the past decade. Thanks to advances in immunotherapy and targeted therapy, treatment environments now allow not only long-term disease control but also long-term survival in some patients.”Renal cell carcinoma is the most common type of kidney cancer, accounting for approximately 90% of all cases. While it often presents with no specific symptoms in the early stages, as the disease progresses, symptoms such as hematuria, flank pain, and fatigue may appear. In advanced stages, a cure through surgery alone is difficult, making drug therapy the cornerstone of treatment.Shift toward immunotherapy combination strategies…changes in first-line treatment strategiesProfessor In Ho Kim, Department of Medical Oncology, Seoul St. Mary’s HospitalCurrent treatment strategies for advanced renal cell carcinoma are selected based on patient risk categories. In clinical practice, physicians use the International Metastatic Renal Cell Carcinoma Database Consortium (IMDC) risk classification to divide patients into favorable-, intermediate-, and poor-risk groups. In intermediate- and poor-risk groups, immunotherapy-based combinations have effectively become the global standard.Professor Kim stated, “In the past, treatment focused on targeted therapy monotherapy, but immunotherapy combinations have changed the treatment paradigm. Particularly in the intermediate and poor-risk groups, the combination of Opdivo (nivolumab) and Yervoy (ipilimumab) has demonstrated survival benefits and established itself as a major first-line treatment strategy.”Currently, the first-line immunotherapy combinations approved in Korea for renal cell carcinoma include: ▲Opdivo + Yervoy ▲Opdivo + Cabometyx (cabozantinib) ▲Keytruda (pembrolizumab) + Lenvima (lenvatinib) ▲Keytruda + Inlyta (axitinib). Combinations involving immunotherapy plus immunotherapy or immunotherapy plus targeted therapy have improved survival rates and disease control compared to conventional targeted therapy monotherapy approaches, driving major changes in renal cell carcinoma treatment strategies.However, Korea’s reimbursement environment is considered insufficient to keep pace with treatment advances. Currently, the only reimbursed immunotherapy combination is Opdivo + Yervoy for IMDC intermediate- and poor-risk patients. Insurance coverage is also limited to a maximum of 2 years. In contrast, immunotherapy plus targeted therapy combinations widely recommended in global treatment guidelines are not reimbursed.Limitations in access to follow-up therapy highlightedThe problem lies in the period following first-line immunotherapy. While many patients in Korea currently use the Opdivo plus Yervoy combination therapy, which is covered by insurance, the options available for subsequent treatment after disease progression are limited.In Korea, reimbursable drugs for second-line renal cell carcinoma treatment are limited to certain VEGF-targeted anticancer agents, such as Sutent (sunitinib), Votrient (pazopanib), and Inlyta. Meanwhile, Cabometyx, which is recommended as a key subsequent therapy option in international guidelines, is not only non-reimbursed but also limited in actual use.International guidelines from the NCCN, ESMO, and EAU recommend cabozantinib as a major follow-up treatment option after first-line immunotherapy-based therapy. However, Korean approval is limited to ‘advanced renal cell carcinoma patients previously treated with VEGF-targeted therapy.’ As a result, patients who received immunotherapy combinations in the first line face structural barriers to using Cabometyx under the current label.Cabometyx demonstrated improvements over everolimus in progression-free survival (PFS), overall survival (OS), and objective response rate (ORR) in the METEOR study. Median PFS reached 7.4 months, OS was 21.4 months, and ORR was 24%, confirming clinical efficacy across all three major endpoints in second-line renal cell carcinoma treatment.Kim stated, “While patients in Korea frequently use the Opdivo plus Yervoy combination therapy as first-line treatment, there remains a structural gap that makes it difficult to naturally transition to evidence-based follow-up treatment afterward.”He further emphasized, “To translate the treatment outcomes of renal cell carcinoma into real-world patient care, we need to discuss improving access not only to first-line treatment but also to subsequent lines of therapy to ensure continuity in treatment.”
- Company
- Immunotherapy Tevimbra's five indications to be reviewed
- by Eo, Yun-Ho May 28, 2026 10:27am
- Product photo of 'Tevimbra'Five additional indications for 'Tevimbra' are anticipated to enter the final review stage of the Health Insurance Review and Assessment Service (HIRA) for expanded reimbursement.According to industry sources, Tevimbra (tislelizumab), a PD-1 inhibitor cancer immunotherapy from BeOne Medicines Korea, is expected to be considered by the HIRA Pharmaceutical Reimbursement Evaluation Committee in June.After obtaining successful reimbursement for esophageal cancer in April last year, which was a first as an immunotherapy, Tevimbra has added five indications for solid tumors, including esophageal cancer, gastric cancer, and non-small cell lung cancer (NSCLC). While expanding Tevimbra's indications, BeOne Medicines applied for expanded reimbursement, which subsequently passed the final Cancer Disease Review Committee (CDRC) meeting of 2025.The specific indications submitted for review include ▲first-line combination therapy for patients with unresectable, locally advanced, or metastatic esophageal cancer ▲first-line combination therapy for patients with unresectable or metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma ▲two types for first-line combination therapy and one type for second-line monotherapy in non-small cell lung cancer (NSCLC).If Tevimbra secures expanded reimbursement this time, its role is expected to expand across various cancer types in South Korea in the future.Since BeOne Medicines has a record of finalizing negotiations with the government by advocating for a 'reasonable drug price' at its initial listing, anticipation is growing for the reimbursement review on the additional indications.It is to be watched whether BeOne Medicines can once again maintain its corporate philosophy of 'providing innovative new drugs at reasonable prices to ensure no patient is left behind.'Meanwhile, Tevimbra has confirmed efficacy and safety across diverse indications through the RATIONALE clinical trial series (RATIONALE-303, 304, 305, 306, 307).Notably, clinical benefits were confirmed in overall patient populations in study groups of esophageal squamous cell carcinoma (ESCC) and gastric or gastroesophageal junction (GEJ) adenocarcinoma. In addition, consistent results were also observed in prespecified subgroups based on PD-L1 expression.
- Company
- Potential shift to IgA nephropathy treatment…'Nefecon'
- by Son, Hyung Min May 27, 2026 04:10pm
- The treatment landscape for IgA nephropathy (IgAN) is likely to shift from a focus on conservative management toward targeted therapies that address the disease's underlying cases.While treatments centered on renin-angiotensin system (RAS) inhibitors, aimed at blood pressure control and reduction of proteinuria, have shown limitations, a local immunomodulatory therapy targeting the Peyer’s patches (immune tissue in the ileum) has demonstrated efficacy in reducing proteinuria and protecting renal function, drawing attention to a potential paradigm shift in treatment strategies.On the 26th, Everest Medicines hosted a media session at the Plaza Hotel in Jung-gu, Seoul, to highlight the clinical value of 'Nefecon (micronized budesonide),' a targeted treatment for IgA nephropathy.IgA nephropathy is a primary glomerular disease characterized by the deposition of immunoglobulin A (IgA) within the glomeruli, which induces inflammation and a decline in kidney function. It accounts for approximately 40% of glomerulonephritis cases in South Korea and is known to commonly manifest in individuals in their 20s to 40s who are socially and economically active. Unlike conventional chronic kidney disease (CKD), which frequently occurs in the elderly population in association with diabetes and hypertension, IgAN is distinct in its onset among a relatively younger ages.Although disease progression varies across patients, persistent proteinuria and renal decline carry a high risk of progression to end-stage renal disease (ESRD). The recurrence rate remains at 20% to 60% even after kidney transplantation, and some studies report that the average life expectancy is shortened by approximately 10 years, with a mortality rate roughly twice as high as that of the general population.Jung Pyo Lee, Department of Nephrology, Boramae Medical CenterIn particular, proteinuria and a depressed estimated glomerular filtration rate (eGFR) are recognized as key indicators of disease progression and elevated cardiovascular risk. A British cohort study reported that a considerable number of cases progressed to kidney failure within 10 to 15 years post-diagnosis, demonstrating that patients with higher baseline proteinuria faced a substantially increased risk of ESRD or mortality.Previously, the management of IgA nephropathy has centered on supportive care aimed at slowing the rate of renal deterioration rather than modifying the disease itself. Renin-angiotensin system (RAS) inhibitors designed for blood pressure and proteinuria control. While RAS inhibitors help reduce proteinuria by lowering intraglomerular pressure, they limitations because they do not directly target the underlying pathogenesis.Professor Jung Pyo Lee explained, "While a renal biopsy is vital for a definitive diagnosis of IgA nephropathy, it cannot be practically performed on every patient. Global guidelines recommend a biopsy when proteinuria exceeds 0.5g. However, in real-world clinical practice, RAS inhibitors or SGLT-2 inhibitors are prioritized when proteinuria is under 0.5g. Immunotherapy is considered when proteinuria reaches 1g or more, and there are approximately 10,000 severe patients who would be eligible candidates for Nefecon therapy."Nefecon is a targeted therapeutic designed to modulate the mucosal immune response implicated in the pathogenesis of IgA nephropathy. It is engineered to locally deliver the active drug to the Peyer's patches in the terminal ileum, a recognized primary anatomical site where IgAN originates.Through this mechanism, it downregulates the production of galactose-deficient IgA1 (Gd-IgA1), a key pathogenic biomarker, thereby suppressing abnormal immune cascades. It was explained that while conventional supportive therapies focus mainly on reducing proteinuria and controlling blood pressure, Nefecon distinguishes itself as a 'disease-modifying' approach that modulates the core progression of the disease.The drug-delivery mechanism is also key. It delivers the drug to the target tissue utilizing a dual formulation technology that integrates delayed-release and sustained-release profiles. The budesonide component is specifically designed to minimize systemic exposure, as the vast majority of the active pharmaceutical ingredient is cleared by extensive first-pass hepatic metabolism. According to the presented data, its impact on the HPA axis (hypothalamic-pituitary-adrenal axis) was also relatively limited.Nefecon was designated as the second product under the Ministry of Food and Drug Safety (MFDS)'s Global Innovative Product on Fast Track (GIFT) program in November last year, and it secured approval in the United States in 2021 under the brand name 'Tarpeyo.'Professor Lee explained, "IgA nephropathy features a high proportion of young patients, and the risk of progressing to ESRD fluctuates depending on the severity of proteinuria and renal function decline. It is critical to consider early immunological interventions in high-risk patients."Confirmed effectiveness in proteinuria·renal function protection Professor Jung-ho Shin, Division of Nephrology, Chung-Aug University HospitalThe clinical value of Nefecon was validated through the global Phase 3 NefIgArd study.NefIgArd was a global, randomized, double-blind, placebo-controlled study conducted in patients with primary IgA nephropathy who exhibited persistent proteinuria despite optimized RAS inhibitor therapy. Patients were randomized to receive either Nefecon or a placebo for 9 months, followed by a 15-month observational follow-up period.The clinical results showed that Nefecon significantly reduced levels of the core pathogenic biomarker Gd-IgA1 by 34% compared to the placebo group.Proteinuria was reduced by up to 51.3% at 3 months post-treatment completion, and the preservation effect on eGFR was robustly maintained throughout the 15-month post-treatment follow-up period.Notably, a modeling analysis based on the eGFR slope from the NefIgArd study generated predictive data suggesting that Nefecon treatment could delay the onset of kidney failure and the initiation of dialysis by up to 12.8 years.From a safety perspective, the most frequently reported adverse events (AEs) included peripheral edema, hypertension, muscle cramps, and acne development.Professor Shin stated, "The key feature of IgA nephropathy management lies in long-term preservation of renal function and delaying progression to ESRD, rather than short-term reduction of proteinuria alone. Nefecon is different from conventional conservative therapies as it directly addresses the disease pathogenesis by targeting mucosal immune responses."Professor Shin added, "The clinical data confirmed that the eGFR preservation benefit was sustained even after treatment cessation, and the modeling analysis supported its potential to delay progression to renal failure. It is now time to consider the necessity of earlier therapeutic intervention in high-risk patients."
- Company
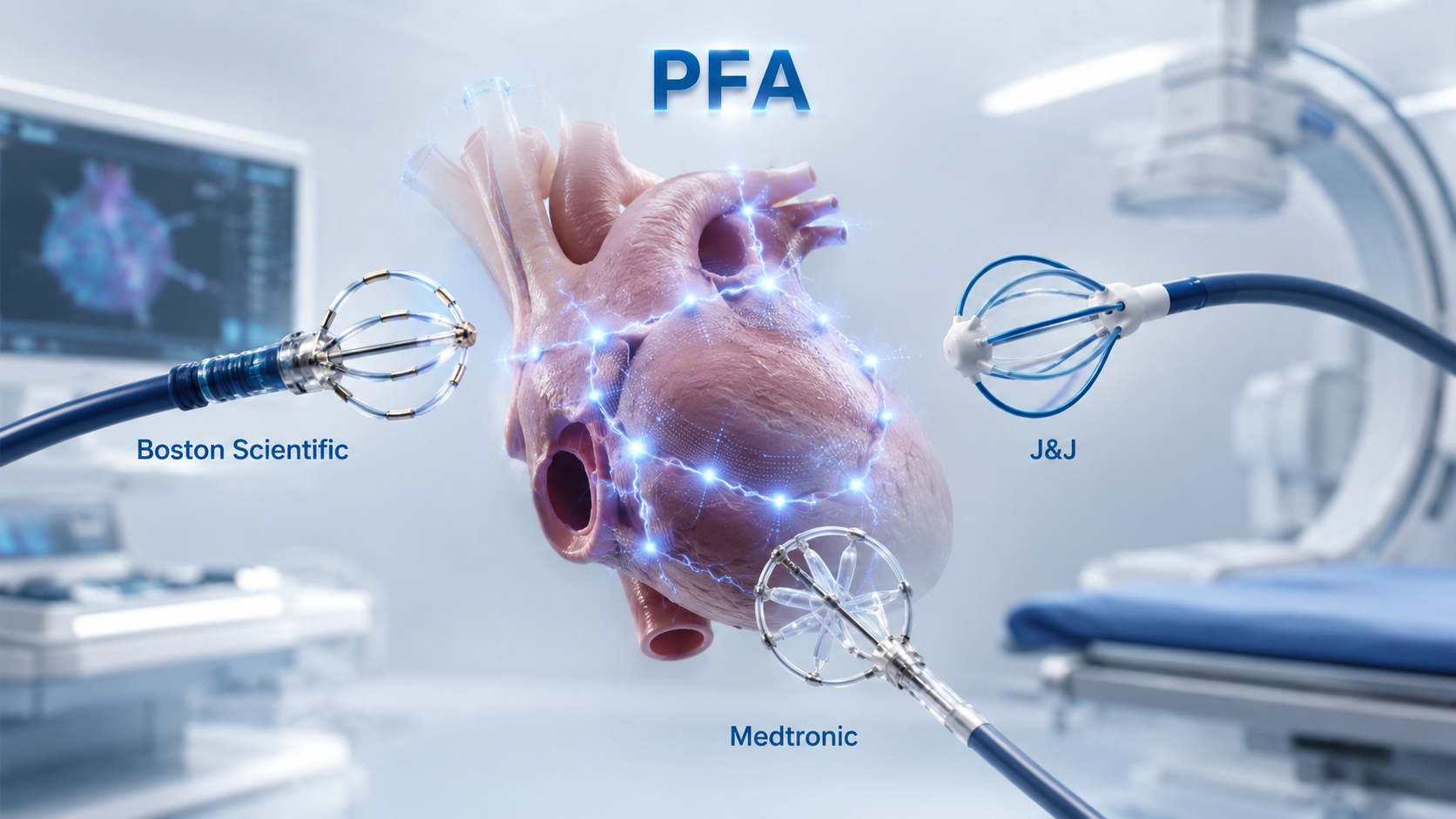
- Pulsed field ablation gains reimbursement in Korea
- by Hwang, byoung woo May 26, 2026 03:21pm
- Reference Material (AI-generated image)The atrial fibrillation ablation market has entered a full-scale competition following reimbursement coverage for pulsed field ablation (PFA).PFA has been garnering attention as a next-generation technology capable of replacing existing radiofrequency and cryoablation procedures, but its adoption was limited due to non-reimbursement barriers. However, with national health insurance coverage beginning in May, patient access has improved, and market conditions are changing.In particular, because the reimbursement applies not to a specific company’s product but to the ‘PFA procedure’ itself, Boston Scientific, Johnson & Johnson (J&J), and Medtronic are all expected to compete under the same regulatory framework.Reimbursement marks a turning point in the market... Will PFA adoption accelerate?PFA is an ablation procedure that uses high-voltage electric fields to selectively damage myocardial cells. Unlike conventional radiofrequency ablation or cryoablation, which rely on heat, PFA is considered a next-generation atrial fibrillation treatment because it can reduce damage to surrounding tissues.In particular, its ability to reduce the risk of complications, such as esophageal injury, phrenic nerve injury, and pulmonary vein stenosis, which were concerns with traditional thermal ablation, is cited as a key strength. Combined with the potential for shorter procedure times, the shift to PFA is already progressing rapidly in the global market.According to a report by the Korea Health Industry Development Institute, Korea’s PFA market is expected to grow from approximately USD 31 million in 2024 to around USD 53 million by 2029, representing a compound annual growth rate of 11.1%.The global PFA market is projected to expand from approximately USD 1.53 billion in 2024 to about USD 9.25 billion by 2029.In practice, even during the non-reimbursed stage, some major tertiary hospitals had already accumulated experience using PFA. According to the report, Severance Hospital introduced PFA at the end of 2024 and has been actively using it since 2025.Out of roughly 1,000 total atrial fibrillation ablation procedures currently performed, about 350 are now conducted using PFA, and the hospital expects more than 70% of all procedures to eventually transition to PFA.Ultimately, while PFA was previously used only on a limited basis depending on whether patients had out-of-pocket insurance coverage, its reimbursement coverage is expected to reduce cost barriers and position PFA as a major treatment option for atrial fibrillation ablation.Market Size and Growth of Korea's PFA Market (Reconstructed AI image of Korea Health Industry Development Institute's report)Boston Scientific’s early approval advantage…J&J focuses on integration, Medtronic on expandabilityAccording to the medical device industry, Boston Scientific currently holds the leading position in the early competitive landscape.Boston Scientific’s Farapulse PFA platform became the first medical device approved in Korea for PFA when it received Ministry of Food and Drug Safety approval in September 2024.Boston Scientific emphasizes that, in connection with the new reimbursement coverage, it possesses both the “Farawave PFA Catheter,” capable of two-dimensional visualization, and the “Farawave NAV PFA Catheter,” which enables three-dimensional visualization of treatment sites.Farapulse’s strengths lie in its early approval and accumulated clinical evidence. According to the company, Farapulse has built up safety and efficacy data through studies such as the ADVENT trial, which directly compared it with thermal ablation, and the MANIFEST-17K registry, which analyzed real-world clinical outcomes in more than 17,000 patients.Its competitor, Johnson & Johnson, is promoting a different type of competitiveness through Varipulse. The Varipulse system is a three-dimensional PFA treatment solution consisting of the Varipulse catheter, TruPulse generator, Carto3 3D mapping system, and Vizigo Steerable Sheath.Its key advantage is integration. Varipulse is designed to integrate with J&J’s electroanatomical mapping system, Carto 3. Because confirming catheter position, visualizing cardiac structures, and identifying lesion locations are increasingly important during PFA procedures, integration with mapping systems may become an important factor in hospital purchasing decisions.Being somewhat of a late entrant in Korea, Medtronic is establishing a competitive presence in terms of expandability.Medtronic’s PulseSelect PFA system received U.S. FDA approval in December 2023 for the treatment of paroxysmal and persistent atrial fibrillation. The company is expected to leverage the fact that PulseSelect was the first PFA system approved by the FDA, along with its global clinical usage experience.Another differentiating point for Medtronic is its future product lineup. Through products such as the dual-energy Affera Mapping and Ablation System, which combines pulsed field and radiofrequency energy, and the Sphere-9 Catheter, the company is pursuing a strategy of expanding treatment areas beyond atrial fibrillation into atrial flutter and ventricular tachycardia.KHIDI Report (AI image reconstruction)Competition lies in hospital workflows rather than productsFollowing reimbursement listing for PFA, market competition is likely to unfold not as a one-sided dominance by a specific company, but rather as a diversification of selection criteria among individual hospitals.Atrial fibrillation ablation procedures are not determined by a catheter alone. Mapping systems, imaging equipment, physician expertise, existing hospital equipment infrastructure, training support, long-term clinical data, and cost structures all play important roles. As a result, preferred platforms may vary from hospital to hospital.For example, hospitals prioritizing early adoption experience and accumulated clinical evidence may favor Boston Scientific’s Farapulse, while hospitals already operating electrophysiology workflows centered on the Carto3 system may choose J&J’s integrated Varipulse solution.Hospitals focusing on future technologies and expandability of indications may instead see Medtronic’s long-term product lineup as more competitive.Nevertheless, the general consensus is that as PFA becomes mainstream, the atrial fibrillation ablation market has entered a full-fledged transition phase.Whereas the technology was previously used mainly by select patients and leading hospitals, patient accessibility and hospital adoption potential are now expanding simultaneously.As a result, while domestic approval and early adoption were previously the key competitive factors, post-reimbursement competition is expected to depend increasingly on real-world experience, interoperability with existing hospital equipment, procedural training, data accumulation, and the speed of follow-up product introductions.A medical device industry official said, “In overseas markets such as the United States, PFA is already rapidly establishing itself as the standard of care. With reimbursement now improving patient access in Korea as well, the transition toward PFA driven by procedural efficiency and safety is expected to accelerate further.”
- Company
- Varying performance levels of medical imaging AI companies
- by Hwang, byoung woo May 26, 2026 03:21pm
- AI-generated imageDomestic medical imaging artificial intelligence (AI) companies reported varying performance in their first-quarter results, even as they posted losses.While attention was previously focused on whether these companies could generate any sales during the initial stages of commercialization, the market is now asking "How do these companies make money?"Consequently, the attention is on the Q1 2026 performance is not top-line sales rankings, but rather the specific channels through which sales is generated. Performance divergence among medical imaging AI companies is accelerating, depending on factors such as overseas commercialization channels, integration into domestic institutional healthcare frameworks, and volume-based recurring revenue streams.Positive indicators in overseas·recurring revenue…Lunit·Coreline Soft differentiating factorsLunit recorded revenue of KRW 24.0 billion for the first quarter of 2026, a 25% increase compared to the KRW 19.2 billion recorded in the same period last year. Concurrently, its operating loss narrowed from KRW 20.8 billion to KRW 13.6 billion, demonstrating increased revenue alongside a reduction in losses.The most critical indicator in Lunit’s performance is its overseas revenue. According to the company's Q1 financial data, international sales reached KRW 23.2 billion, a 29% increase from KRW 17.9 billion in the prior-year period, accounting for a 97% of total revenue.Domestic medical AI markets are highly susceptible to institutional and regulatory factors, such as New Medical Technology Assessments, non-reimbursement status, and reimbursement rates. Conversely, Lunit has successfully established a structure in which the vast majority of its quarterly revenue is secured through global distribution channels.By business segment, cancer screening remains the primary growth driver. Software revenue from Lunit's cancer screening segment totaled KRW 21.3 billion in Q1, accounting for approximately 89% of total revenue.Although its cancer treatment decision-support segment, which includes Lunit SCOPE, is emerging as a new growth vector, the core revenue generator as of Q1 2026 remains the cancer screening business, which includes diagnostic imaging assistance and breast cancer screening software.However, challenges persist on the bottom line. While its operating loss narrowed, the company still posted a KRW 13.6 billion deficit in the first quarter. Although an improving trend in profitability has been confirmed, further revenue expansion and disciplined cost management are required to achieve a turnaround to operating profitability.Comparison of 2025-2026 first quarter revenue of major medical imaging artificial intelligence (AI) companies (unit: KRW 100 million/source: quarterly reports). Lunit, Vuno, Deepnoid, Coreline Soft, and JLK.Coreline Soft, while not yet achieving Lunit's massive exterior performance, showed other positive indicators in its revenue structure.Coreline Soft recorded consolidated revenue of KRW 1.3 billion in the first quarter of this year, representing an approximate 44% increase year-over-year.Out of this total, overseas revenue amounted to approximately KRW 800 million, representing 62.4% of its overall business. This was the first time the company's international revenue share has exceeded 50%.The expansion of its recurring revenue stream is also noteworthy. The company announced that the share of recurring revenue derived from volume-based utilization, term licenses, and software maintenance agreements reached 49.1% in Q1, climbing roughly 10 percentage points from 38.9% in the prior-year period.Specifically, volume-based pay-per-use revenue surged by 319.7% year-over-year. This indicates that the business model of medical AI companies is successfully pivoting from one-off, on-premise deployments and perpetual license sales toward a framework dynamically tied to actual utilization rates and diagnostic screening traffic.This structural shift is closely aligned with international national screening initiatives. According to Coreline Soft's documents, the company secured 11 new hospital contracts in Germany during Q1 alone, surpassing its entire annual total of 10 new contracts in Germany for the previous year within a single quarter.Coinciding with Germany’s statutory health insurance reimbursement coverage for low-dose CT (LDCT) lung cancer screening, demand for AI-driven diagnostic reading, quality control, and longitudinal tracking systems is poised for further expansion.Ultimately, Lunit leveraged international revenue to establish a quarterly baseline of over KRW 20 billion, while Coreline Soft signaled a clear structural pivot toward recurring revenue streams despite its smaller absolute volume. The former differentiated itself through scale, and the latter through its business model structure.Vuno shows high reliance on DeepCARS…performance staggers due to regulatory variablesIn the case of Vuno, the company had previously achieved record-high consolidated annual revenue of KRW 34.8 billion last year, growing 35% year-over-year, and successfully narrowed its operating loss by 60% down to KRW 4.9 billion through aggressive cost-optimization initiatives.Performance was led by its flagship product, DeepCARS, an AI-driven cardiac arrest prediction medical device, which generated KRW 25.7 billion in revenue, representing an approximate 18% year-over-year increase.However, its momentum slowed in the first quarter of 2026, with revenue dipping 16% quarter over quarter to KRW 6.0 billion.The company explained that this temporary revenue fluctuation occurred as DeepCARS navigated the New Medical Technology Assessment process, coinciding with the expiration of its assessment deferral window.Vuno’s challenge lies in the high concentration of its core revenue stream in domestic DeepCARS sales.Although Vuno has designated the US commercial launch of DeepCARS as its top strategic priority, a fully realized revenue stream backed by US regulatory clearance and insurance reimbursement has not yet materialized. Consequently, Vuno's Q1 financial performance remained dependent on domestic DeepCARS sales and the progress of the domestic New Medical Technology Assessment procedures.While market expansion is anticipated once the regulatory assessment process is finalized, establishing and diversifying commercial pipelines in global markets remains an urgent, ultimate objective.AI-generated imageDeepnoid·JLK, In a transitional phase prior to scaling revenue volumeDeepnoid generated only KRW 7.5 billion in annual revenue, falling significantly short of its previously guided revenue forecasts.Deepnoid attributed this discrepancy to slower-than-expected expansion in medical AI adoption, delays in securing health insurance reimbursement pricing and international regulatory clearances, and intensifying competition from incumbent PACS (Picture Archiving and Communication System) vendors.Furthermore, Deepnoid disclosed that it halted the standalone commercialization of DEEP:PHI, citing diminished differentiation for platform-type business models following the advent of generative AI.Its revenue for the first quarter of 2026 amounted to approximately KRW 400 million. Even within this figure, the company relied heavily on its industrial AI division (roughly KRW 300 million) rather than its core medical AI segment (which brought in just KRW 57.75 million).As operating deficits widened due to increased fixed costs associated with R&D scaling and expert talent acquisition, the company’s top priority has shifted to proving the self-sustainability of its medical division. This hinges on successfully anchoring its cerebral aneurysm solution, "DEEP:NEURO," within the domestic non-reimbursement market.JLK is actively driving market entry into the non-reimbursement sector for its pipeline expansion, including its large vessel occlusion (LVO) detection solution, "JLK-LVO", starting with the current non-reimbursement prescriptions of its ischemic stroke solution, "JLK-DWI."However, to overcome the limitations of a constrained domestic market, the company is committing substantial capital to establishing international commercial networks via its wholly owned subsidiaries in the United States (JLK USA INC.) and Japan (JLK Japan, Inc.).Both JLK USA and JLK Japan reported zero revenue in the first quarter, posting net losses of approximately KRW 100 million each. Because these international subsidiaries have yet to generate meaningful top-line contributions, the company is currently in a wait-and-see window to determine whether these infrastructure investments will successfully translate into global financial performance.A medical device industry employee stated, "With digital health companies that have secured insurance reimbursement pricing recently reporting robust financial metrics, pressure to perform is intensifying for medical imaging AI firms as well. We have entered a stage where regulatory clearance or initial hospital adoption alone no longer satisfies market evaluation. The core mandate now is establishing a commercial architecture that translates into sustainable, predictable revenue."
- Company
- Will reimbursement of CDK4/6 inhibitors finally be expanded?
- by Eo, Yun-Ho May 26, 2026 03:21pm
- The decisive moment for expanding reimbursement coverage of CDK4/6 inhibitors in early breast cancer is approaching.According to industry sources, Eli Lilly Korea’s Verzenio (abemaciclib) and Novartis Korea’s Kisqali (ribociclib) are expected to be submitted this week to the Health Insurance Review and Assessment Service’s Cancer Disease Deliberation Committee.The specific indication under consideration for reimbursement expansion is “adjuvant therapy for patients with HR-positive (hormone receptor-positive)/HER2-negative stage II and III early breast cancer at high risk of recurrence.”Although the two drugs share the same mechanism of action, their situations differ somewhat. Verzenio is making its fourth attempt before the reimbursement committee, while Kisqali is being reviewed for the first time. The reimbursement expansion application for Kisqali was filed by the pharmaceutical company itself, whereas the application for Verzenio was submitted by the Breast Cancer Subcommittee of the Korean Society of Medical Oncology.Verzenio faced difficulties in being reviewed by CDDC from its first attempt for early breast cancer. After a long wait of 6 months after submitting the reimbursement application, it was finally reviewed by CDDC in May 2023, but the result was “reimbursement criteria not set.” Five months later, in October, Lilly resubmitted the reimbursement application to HIRA, and in March and July last year, it was submitted to CDDC for review, resulting in the same outcomes.The overall survival (OS) data was cited as the primary reason for Verzenio’s failure to secure expanded coverage. However, with Verzenio now securing OS data, expectations are rising for this CDDC review.In contrast, Kisqali, which is making its first attempt, has not yet secured OS data. While an improvement in OS is foreseeable, there is no direct supporting data available yet.Invasive disease-free survival (iDFS) is regarded as a clinically meaningful surrogate endpoint strongly correlated with OS in early breast cancer due to the disease characteristics, and Kisqali demonstrated encouraging results in the NATALEE study.Although the two drugs share the same mechanism, their differing circumstances make it noteworthy to see what outcomes the two drugs will yield at the CDDC review.
- Company
- 10th year of Boksan-Suzuken partnership…"Establishing a platform for mutual growth"
- by Kim, Jin-Gu May 22, 2026 10:25am
- Commemorating the 10th anniversary of their strategic partnership, Boksan Nice and Japan's Suzuken presented the 'redefinition of roles between pharmaceutical companies-wholesalers' and the 'transition to digital distribution platforms' as key strategies to overcome the crisis facing the pharmaceutical industry, which is currently pressured by drug price cuts and market restructuring.On the 21st, Boksan Nice and Suzuken hosted the 'Boksan Nice-Suzuken Partnership 10th Anniversary Seminar' at the Orchid Room of the Westin Josun Seoul. The event brought together executives from both companies and around 80 stakeholders from the pharmaceutical distribution industry.The event was organized to reflect on the decade-long partnership between the two companies and to outline specific future collaboration pathways for pharmaceutical companies and the wholesale sector to achieve mutual growth amid ongoing large-scale drug price reductions and structural inefficiencies in distribution in South Korea. The two companies established their relationship in 2016 when Suzuken made a KRW 52 billion investment in Boksan Nice.In his opening remarks, Boksan Nice Chairman Eom Tae-ung emphasized, "This seminar goes beyond a simple 10th-anniversary celebration; it is a venue to collectively address the rapid changes and future direction facing the pharmaceutical industry and distribution sector today." and added, "As the global weight of marketing and distribution continues to grow, Boksan Nice’s clear direction in this era of selection and concentration is to become a healthy and competitive partner for domestic pharmaceutical companies."Suzuken Chairman Hiromi Miyata introduced the background behind the long-standing trust between the two companies over the past decade. Chairman Miyata stated, "When we entered the Korean market 10 years ago, we conducted interviews and surveys targeting pharmaceutical companies, healthcare institutions, and pharmacies, and Boksan Nice's reputation and credibility were overwhelmingly high," and added, "This deep trust became the decisive factor for our collaboration. Over the last 10 years, we have built a foundation for growth through continuous joint projects and close talent exchange. We will continue to support the growth of Boksan Nice."Boksan Nice Chairman Eom Tae-ung (left) and Suzuken Chairman Hiromi Miyata.Boksan Nice and Suzuken jointly assessed that the pharma-biotech markets and pharmaceutical distribution environments in both Korea and Japan are undergoing profound shifts. In Korea's case, a heightened sense of crisis is escalating due to large-scale drug pricing restructuring.In response to this crisis, Japan's Suzuken proposed a "digital platform" strategy as an alternative solution. Shigeru Asano, Director and Chairman of Suzuken, shared collaboration case studies from the Japanese market, noting, "Amid changing market environments, wholesalers as well as pharmaceutical companies must move beyond simple logistics fulfillment to deliver new value-added functionalities."Chairman Asano identified "Visualization, Connection, and Change" as the core strategies of the Suzuken Group, explaining, "We are currently building a digital-based 'healthcare data platform' that organically links data from medical institutions, healthcare professionals, and the supply chain," and added, "Through this, we will perform an advanced role by supporting the digital marketing efforts of pharmaceutical companies while, in turn, providing a stable infrastructure for pharmaceutical supply."Chairman Eom Seung-wook of Boksan Nice identified fundamental inefficiencies in the domestic pharmaceutical distribution structure and proposed streamlining distribution channels as a solution.Eom diagnosed, "Currently, there are around 4,000 wholesalers in Korea, and the collective market share of the top 10 companies does not exceed 40%," and added, "Consequently, Korean wholesalers have remained in the role of simple delivery vendors until now, and under the pressure of large-scale drug price cuts, the legacy approach is unsustainable for both pharmaceutical companies and wholesalers."Eom proposed a structural division of business as a concrete model for collaboration. He emphasized, "Simplifying distribution channels is no longer an option but an absolute necessity. Roles must be redefined so that pharmaceutical companies can focus entirely on their core operations, such as R&D and manufacturing. At the same time, wholesalers solely handle distribution and operational efficiency," and concluded, "Boksan Nice will become a core partner that substantially contributes to improving the profitability of pharmaceutical companies hit by drug price cuts by eliminating unnecessary costs, expanding market accessibility, and simplifying workflows."
- Company
- HPV prevention expanded to male adolescents
- by Son, Hyung Min May 22, 2026 10:25am
- With the National Immunization Program (NIP) expanding to include male adolescents, Korea’s HPV (human papillomavirus) prevention strategy is shifting from a male-centered approach to a system that prevents HPV in both male and female individuals.Analysts emphasize the significance of this move, noting that while major countries abroad have been implementing simultaneous vaccination for both genders for several years as part of their strategies to reduce HPV-related cancers, Korea has finally established a foundation for vaccinating male adolescents, albeit belatedly.On the 20th, MSD Korea held a media session at Seongam Art Hall in Seoul under the theme “A New Standard for HPV National Immunization: Vaccination for Both Genders.”Starting this month, the Korea Disease Control and Prevention Agency (KDCA) has included 12-year-old male adolescents (born in 2014) in the National HPV Immunization Program. Previously, the program only covered female adolescents aged 12 to 17 and low-income women.This expansion is considered significant because it extends free HPV vaccination, which had been limited to females for the past decade, to include male adolescents. According to the presented materials, the first-dose HPV vaccination rate among male adolescents in Korea stood at just 0.2%. In contrast, rates were 77.7% in Australia, 71.2% in the UK, and 59.0% in the US.HPV is known to be a virus that can infect both men and women. According to the International Papillomavirus Society (IPVS), approximately 5% of all cancer cases worldwide are reported to be related to HPV. While most infections clear up naturally, persistent infection can lead to cervical cancer, vaginal cancer, anal cancer, vulvar cancer, and genital warts.Dong-hyun Kim, Professor of Pediatrics at Inha University HospitalDong-hyun Kim, a professor of pediatrics at Inha University Hospital, stated, “HPV is not just a cervical cancer issue. It is a virus that can also cause anal cancer and genital warts in men. Since it is an infectious disease that affects and spreads among both men and women, it is difficult to approach it with prevention strategies targeting only a specific gender.”According to data from the Korea Disease Control and Prevention Agency, reported HPV cases in Korea increased from 10,945 in 2020 to 14,534 in 2024. In particular, reported cases among men rose 82.9% during the same period, from 117 to 214 cases. A domestic study of men found that approximately 59% tested positive for HPV DNA.Professor Kim cited high reinfection risk and low natural clearance rates as characteristics of HPV infection in men.“HPV is a virus that can infect both men and women, and it is not an issue limited to a specific gender. We need to approach prevention strategies from the perspective of infectious disease, rather than gender.”He continued, “Men often carry the virus asymptomatically, and the rate of natural clearance is known to be slower than in women. This means transmission may continue repeatedly without the person even being aware of it.“Experts recommend 11–12 years of age as the optimal age for HPV vaccination. This is because vaccination before sexual debut ensures high immunogenicity, and the 9–14 age group can expect a sufficient immune response with fewer doses than adults.Professor Kim emphasized that HPV vaccination is not a gender-specific issue, but an infectious disease prevention strategy for both men and women.The quadrivalent HPV vaccine available through the HPV national immunization program is administered in two doses for those aged 9 to 13 (6–12 months apart)In particular, study results showed that two doses administered at ages 9 to 13, when immune responses are active, demonstrated non-inferior immunogenicity (geometric mean antibody titers, GMT) against HPV types 16 and 18 compared with three doses in women aged 16 to 26. This highlights the need to secure preventive effects through early vaccination.Professor Kim said, “When vaccinated before age 15, two doses alone show high immunogenicity compared with three doses in older age groups. It is also meaningful in terms of vaccination convenience because it can be administered at the same time with other vaccines included in the national immunization schedule, such as Tdap and Japanese encephalitis vaccines.”He continued, “Overseas, countries have already set the eradication of cervical cancer as a national goal based on simultaneous vaccination of both men and women. Korea also needs to begin serious discussions on shifting its HPV prevention strategy, starting with this expansion of the NIP to male adolescents.”