- LOGIN
- MemberShip
- 2026-04-25 07:01:15
- Product
- Who will succeed in COVID-19 treatment?
- by choi-sun Mar 25, 2020 06:01am
- As pharmaceutical companies around the world are plunging into the development of vaccines and treatments for COVID-19 treatment, attention is focused on who will succeed in commercialization. Among the treatments, Gilead Sciences entered Phase III of clinical trials, and in the field of vaccines, it entered Phase I by Moderna Therapeutics. However, it is not the first to release it because it may fail clinically. Among the other candidates that follow, the mechanism is slightly different, and it is also a concern that they may be able to get the title of a COVID-19 treatment or COVID-19 vaccine. ▲Treatment = Pre-Clinical ~ Phase III There are two ways to respond to the COVID-19. There is a 'therapeutic agent' that suppresses the proliferation of the virus after infection, and there is a 'vaccine' that focuses on preventing infection. COVID-19 is a type of RNA virus that shares its roots with SARS and MERS. A similar principle is that previously used Ebola treatments are used to treat COVID-19. #Remdesivir, an Ebola treatment that Gilead Science is developing, has a mechanism to inhibit RNA polymerase necessary for virus replication and proliferation. It is for this reason that it quickly entered phase III clinical trials. On the 26th of last month, Gilead started phase III of Remdesivir in the United States and China, followed by investigational new drug application on the 2nd of this month in Korea and completed clinical preparation. In fact, Seoul National University Hospital on the 9th concluded a clinical research agreement with the National Institutes of Health and started enrolling patients immediately from this day, so it is expected that the results will be confirmed in the first half of this year. The main goal is whether to lower the boby temperature and get cured within two weeks at the same time. However, it is still unknown whether Ebola virus clinical trials will lead to commercialized products in that it failed with a high mortality rate compared to competing drugs. The rest of the treatment remains in the Pre-Clinical, except that Remdesivir is in Phase III clinical trials. This means that it takes at least one or two years to confirm the reality. Johnson & Johnson is approaching COVID-19 as a treatment and vaccine development based on its past experience in developing Ebola and Zika viruses. Johnson & Johnson is developing a virus-inactivating vaccine at the preclinical stage, and is also working with the Biomedical Advanced Research and Development Authority (BARDA) to develop therapeutics for patients who are already infected. Through this, it is examined whether existing medicines, such as Remdesivir, an Ebola treatment, Kaletra, an AIDS treatment, and Avigan, a flu drug developed in Japan, are effective in suppressing COVID-19. US bio-company Regeneron Pharmaceuticals is focusing on the development of antibody therapeutics through genetic engineering. Although it is still in the preclinical stage, Regeneron plans to develop an antibody therapy against COVID-19 through mouse genetic manipulation technology capable of producing human antibodies. The candidate is expected to go into animal testing soon, and is preparing for human clinical trials by the third quarter of this year. In fact, during the Ebola incident in 2015, Regeneron had experience in developing a mixed-antibody drug that doubled the patient's survival rate. Vir Biotechnology is also developing antibody-based therapeutics. The difference from Regeneron is that instead of generating antibodies by genetic manipulation, antibodies that are naturally produced after infection with similar corona-like viruses such as SARS in the past are used. In fact, this method has been applied to MERS and SARS in the past. A method of collecting serum from a patient who formed a natural antibody and administering it to a serious patient was achieved. Vir Biotechnology is investigating whether the SARS virus is similar to COVID-19, so that antibodies from patients recovering from SARS can be isolated and applied to COVID-19. #Vir is in the early stages of development in cooperation with Chinese company WuXi Biologics, and it is not clear when the clinical trial is expected. ▲Vaccine = Pre-Clinical ~ Phase I Currently, phase I is at the forefront from vaccine development. Modena therafutics developed the vaccine candidate mRNA-1273 only 42 days after the nucleotide sequence of the COVID-19 was confirmed and jumped to the promising group. mRNA has a mechanism in the body that allows cells to form antibodies on their own. Modena is expected to begin research next month with healthy volunteers in cooperation with the National Institutes of Health (NIH). Once the safety of mRNA-1273 has been demonstrated, Phase II will be undertaken to confirm the effectiveness of the vaccine in hundreds. However, Modena, which is developing mRNA or mRNA designed for cells in the body to produce antibodies against viruses, has not been approved by the FDA as a related drug. Except for Modena, vaccines of various mechanisms are still in the Pre-Clinical. CureVac, like Modena, develops a vaccine that promotes the production of antibody proteins using synthetic mRNA. It is expected to be ready for clinical trials within a few months. China's Clover Biopharmaceuticals, was transferred its technology to a proprietary adjuvant compound that improves the effectiveness of the vaccine from GSK in February and started developing vaccines. The company is developing a vaccine against infection by injecting a protein that stimulates the immune response and activating body immunity, but the clinical trial plan has not yet been detailed. Inovio, the United States, has been developing DNA medicines for the past 40 years, and has received subsidies for the development of DNA-based vaccines from the Coalition for Epidemic Preparedness Innovations (CEPI). In partnership with Chinese manufacturer Beijing Advaccine Biotechnology, INO-4800 candidate vaccine is in Pre-Clinical, and clinical trials are expected in the second half of this year. Sanofi, which has successfully developed a yellow fever and diphtheria vaccine, is working with the BARDA to develop COVID-19 vaccine. Sanofi aims to create a chimeric DNA vaccine that activates the patient's immunity by mixing a portion of the DNA of COVID-19 with the genetic material of the harmless virus. The vaccine candidate is expected to be tested in the laboratory within 6 months, and the clinical trial is expected to be available within 1 to 18 months. Sanofi said the technology has been applied to SARS. However, Sanofi expects to take at least three years to get approval. Experts say it is too early to make a positive outlook, as various therapeutics and vaccines are still in the Pre-Clinical of exploring candidates and evaluating safety. Professor Ki-seok Jeong of the department of Pulmonology at Hallym University said, "I think it is difficult to cure the drug in the near future. However, in the case of Remdesivir, an ebola drug, it would be better if indications were added as much as it promotes clinical trials in Korea". "An RNA virus has many mutations, so it is not easy to develop a therapeutic drug and it is more difficult to develop a vaccine. Considering that multinational companies spend ₩1 trillion on new drug development, a government-level investment in development is necessary" he advised.
- Product
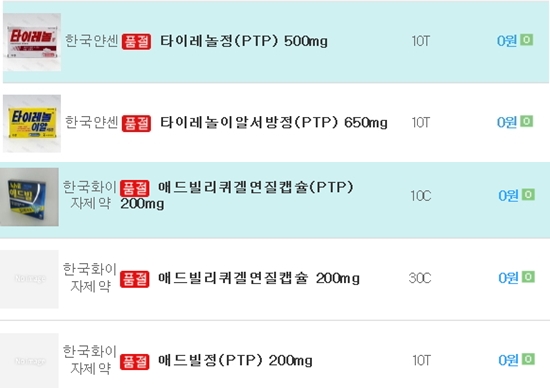
- Panic buying Tylenol-Advil in fear of COVID-19 fake news
- by Jung, Heung-Jun Mar 04, 2020 06:27am
- Related drugs are experiencing shortage in online pharmacy shops Tylenol and Advil are out of stock as their demands have skyrocketed due to online fake news encouraging panic buying of OTC drugs and claiming the Korean government’s containment of COVID-19 has failed. On Mar. 2, a multiple online pharmacies have posted notice on shortage of Tylenol, Advil and Aspirin. Apparently, the circulating ‘story’ is supposedly a chat between graduates of Seoul National University College of Medicine, which claims the outbreak has entered the ‘community infection’ phase that cannot be contained even with utmost care, and the prognosis of individual would heavily depend on their immune system. The made-up story also talked about concerning stories of how visiting hospital has gotten impossible once infected, and contracting the virus at selected testing sites. The story advised to hoard non-steroidal anti-inflammatory drug, antibiotics and antitussive, expectorant and mucoactive agents, such as aspirin, Advil and Tylenol. It also stressed about acquiring emergency food supply as self-quarantined patients would have to stay at home for over two weeks. The participants in the fake chat also argued seniors and individuals with cardiovascular or respiratory conditions are considered high-risk group. The people in the story assumed and hyped the fear of the public institutes and transportations shutting down. A pharmacist from Songpa-gu, Seoul, wondered “There has been a surge of customers buying specifically Tylenol and aspirins. It seems like inaccurate information has been circulating on Youtube and blogs.” Some pharmacies started securing stocks of the said products with abruptly heightened demands, and pharmacists are conflicted among themselves. Another pharmacist in Gyeonggi province commented, “Some pharmacists were worried about stocking up the drugs in the group chat. Basically they are encouraging panic buying, but as others reprimanded them the group chat was split.” The pharmacist emphasized, “It seems like fake news is kept on regenerated and disseminated. Professional pharmacists should not align themselves with it.” Another pharmacist in Gyeonggi province complained of the fake news spreading through social media as many of senior patients suddenly started looking for aspirin. Pharmacists from other regions have agreed on the problem of spreading inaccurate information as many of their customers were asking for aspirin, Tylenol and antitussive, expectorant and mucoactive agents. A few of pharmacy chains have sent out text messages to member pharmacies to inform purchase limit of Tylenol, and suggested purchasing other options with the same agent.
- Product
- What drug will be replaced by Belviq’s withdrawal?
- by Lee, in-bok Feb 18, 2020 10:49am
- As Ildong’s Belviq, which dominated the obese drug market for a long time, is finally exiting the market, there is a growing interest in which drug will fill this gap. Obesity experts are predicting that safety-oriented prescriptions may be available for the time being, while Saxenda and Qsymia are expected to benefit directly. The Ministry of Food and Drug Safety recently decided to stop selling and recall Belviq(lorcacerin) because of the risk of cancer. The U.S. Food and Drug Administration (FDA) has recommended its recovery, saying the risk is greater than the benefit. As a result, Eisai in charge of the U.S. sales, immediately took over its own collection, and Ildong, a domestic sales company, is also in the process of collecting it. According to IQVIA, a global market research firm, Belviq has been overwhelmingly occupying the No.1 ranking in the obese market until 2018 since it landed in Korea in 2015. Indeed, in 2018, Belviq's outpatient prescriptions amounted to ₩9.07 billion, the highest in the obese drug market, beating Dietamin(₩8.48 billion) and Hutermin (₩7.86 billion). Literally the market dominated by the number one leader suddenly withdrawn from the market is bound to be a big ripple Obesity experts point out that safety will be an issue for obesity prescriptions for the time being. It is believed that Belviq could also create the same situation, reflecting on the safety controversy that has been taking place for the time being after the 2010 Reductil(Sibutramine) eviction Professor A, a member of the Korean society for the study of obesity, said, "The safety issue for obesity drugs persisted for a while even when Reductil was evicted as a cardiovascular safety issue, and Belvik is also likely to create such a situation". What kind of drugs are obese experts considering as a substitute for Belviq? First of all, Saxenda(Liraglutide) is expected to be nearing peak item. Prescriptions have already increased since last year, conquering the obese drug market, and safety issues cannot be ignored. Professor A said, "I think that the Saxenda, which was developed with GLP-1 diabetes, will benefit as much as it has been tested for safety, also, it's already been imprinted on obese patients and for now it's almost the only drug that is free from safety issues". In fact, Saxenda has already led the reorganization of the overweight drug market, surpassing Dietamin(₩9.3 billion) and Belviq(₩8.8 billion), recording outpatient prescription amounts of ₩15.1 billion in the third quarter of 2019. With the disappearance of the biggest competitor among them, there is a prospect that Saxenda’s prescription may be accelerated. However, some are expecting the limited competitiveness of the Saxenda. Rather, Oral medications will be more popular because the characteristics of the drugs are completely different An executive of the DAOR said, “Saxenda has long-term safety data, but it just quotes past clinical trials. Furthermore, there may be a difference in compliance when switching to injection in that Belviq is oral”. He said, "While Qsymia is likely to benefit from the new drug effect, some people are considering Phentamines, which have been prescribed for a long time". Some experts predict that Belviq's withdrawal will not have a big impact on the market right away. Obesity drugs are not a long-term use treatment itself, and it is not urgent enough to change the prescription to another drug right now. Professor B, a member of the Korean society for the study of obesity, said, "Stopping a medication used to treat obesity right away does not cause a problem immediately. Belviq's withdrawal will not have a big impact on the market". In addition, the professor said, "Because it is a drug that has been prescribed a lot, Pharmaceuticals will go for the potion and aggressively market it, but I think that the potion won't be moved to other drugs right now, although the potion may come down".
- Product
- Ebola drug Remdesivir is used to treat COVID-19
- by Lee, In-Bok Feb 13, 2020 06:28am
- Remdesivir by Gilead, which is being developed as an Ebola treatment, is expected to benefit significantly from the spread of COVID-19. The explosive demand for therapeutics around the world has led to unintended recruitment of clinical trials, which has resulted in significant time and cost savings. In some cases, the possibility of quick permits is also increasing. Clinical trials in the US, China, etc, Skip Prior Authorization The National Institute of Allergy and Infectious Diseases (NIAID) announced on the 12th that it will begin a clinical trial of Remdesivir with COVID-19. Remdesivir is a drug that Gilead Science is developing for the Ebola virus, and it is preparing for phase III full-scale after completing phase II clinical trials, However, the analysis predominates that the recruitment of the Ebola virus patient population will be limited and it will take some time due to the budget and time constraints for multinational clinical trials. It is a fact that some aspects of the permit has been placed in a somewhat opaque situation. However, with the rapid rise of Remdesivir in the treatment of new COVID-19, the demand has risen sharply and the environment is ready for immediate clinical trials without prior review and approval. This has been decisive in the case of the first COVID-19 patient from the United States. Indeed, researchers in the Centers for Disease Control and Prevention (CDC) have received a lot of attention from the New England Journal of Medicine (NEJM), with a case report describing the first patient prescribed Remdesivir. At the time, the patient was given other antiviral agents and antibiotics, but the symptoms got worse. Eventually, the administration of Remdesivir improved significantly. Since the Ebola virus is an RNA virus such as COVID-19, Remdesivir, which has a mechanism that prevents RNA replication, also inhibits COVID-19. NEJM Case Report Critical Leads to a quick permit As these effects become known, patients in China and Thailand are already taking Remdesivir in patients. The Chinese government has also urged Gilead to cooperate urgently and to expand the supply of Remdesivir. The same is true for large-scale clinical trials in China ahead of the United States. As soon as the potential for treatment was recognized, the Chinese health authorities immediately approved the medication in the form of clinical trials without prior review or approval. As a result, China has already entered into a controlled trial of 761 patients and is planning to proceed with the trial until April, and then review the rapid approval. This is not the only case in China. The U.S. Food and Drug Administration (FDA) and the CDC also mention rapid approval. Since the drug is already in the form of phase III clinical trials, it is in a position to grant a new drug based on this result. This fact has already been communicated to foreign countries through the New York Times and international press, and for this reason, China and Thailand are reportedly preparing for clinical trials and rapid approval. At the end of last year, the drug, which had even entered phase III clinical trials, was opaque, and in a few months, the phase III clinical trial could be approved for rapid approval. For Gilead, this is a great opportunity to save a great deal of time and budget in collecting patients, selecting institutions, and systematically analyzing them to prepare for permits. However, it is confirmed that clinical trials and rapid approvals are not being reviewed in Korea yet. As China's demand is so large that it attracts the world's quantity, it is not easy to consider prescription even in Korea. "Remdesivir is expected to be effective, but it is not easy to even prescribe it due to the lack of stock in our country, and for this reason, the policy recommendation was to exclude from primary treatment", said Jihwan Bang, the team leader of the central clinical task force (TF). An official from the Ministry of Food and Drug Safety said, "No immediate approval has been reviewed regarding COVID-19 treatment".
- Product
- It costs ₩867 million to become a physician
- by Kang, Shin-Kook Feb 11, 2020 06:29am
- It is estimated that it costs ₩867 million from entering medical school to obtaining professional qualification. The Korean Medical Association(KMA)'s research institute for healthcare policy (Director Deok-sun Ahn) announced on the 7th that it has published a research report on the estimation of doctor training costs and public support. According to the research report, the average cost of education per student in the basic medical education stage (1 year of premedical course and 1 year of regular course) was ₩64.97 million (minimum ₩54.12 million, maximum ₩77.62 million). Divided into premedical courses and medical courses, the average annual education cost per premedical student was ₩25.3 million (at least ₩14.93 million, up to ₩38.81 million). The average annual education cost per medical student was ₩39.95 million (minimum ₩3257 million, maximum ₩48.51 million). As a result of estimating the cost of internships, the average annual training cost per intern was ₩73 million (min. ₩55.5 million, max. ₩93.95 million). As a result of estimating the training costs of majors (5 departments: internal medicine, surgery, obstetrics, pediatrics, family medicine, etc.), the average annual training cost for each major is ₩146.04 million(minimum ₩111.18 million, max. ₩187 million). According to the results of estimating the cost of each doctor's training, the total cost of training one doctor (to become a specialist) is ₩887 million, and the total annual cost for training doctors is ₩2.7175 billion. On the other hand, in the US, UK, Germany, Japan, and Canada, the state and society share a large part of the cost of physician training. In the United States, for example, the share of medical costs needed to train doctors is shared by various organizations, including 23% in the state, 8% in federal research funds, 18% in medical schools, 28% in clinical care, and donations. According to the U.S. medical schools, 70% of the per capita training costs are shared by Medicare (20% direct, 50% indirect) and 30% by Medicaid and other private health insurance companies. Korea's budget is ₩72 trillion (₩60 trillion in social welfare and ₩12 trillion in health care) in 2019, and in the budget, the portion for training and aptitude and supply of medical personnel is ₩24.9 billion, of which, the budget for support for nurturing majors and qualification examinations for specialists is about ₩1.3 billion. The Research institute for healthcare policy said, "As the social interest in patient safety increases, the education and training environment for excellent doctor training is increasing, the study was meaningful in estimating the cost of training and total cost per doctor and suggesting the justification for public support". The research institute said, “Based on the results of the study on the estimation of the cost of doctor training and public support measures, it is necessary to form a consultative body for social discussion of the cost sharing of doctor training, and social councils should involve the important stakeholders in health care, the nation, the Korean Medical Association, medical schools, intern & training Centers, local governments, and the general public”.
- Product
- Lyxumia by Sanofi stops supply in the second half
- by Kim, Min-Gun Feb 10, 2020 06:31am
- Sanofi-Aventis KoreaSanofi-Aventis Korea's supply of GLP-1 analog diabetes treatment ‘Lyxumia (Lixisenatide)’ will be discontinued from June this year. According to the distribution industry on the 7th, Sanofi recently announced that it has stopped supplying two formulations, such as 10μg and 20μg of Lyxumia pen, due to company circumstances. Sanofi said in an announcement that it is scheduled to withdraw its product license (from the Ministry of Food and Drug Safety) for the convenience of the company. If Lyxumia is withdrawn in March, the supply is expected to cease in June this year, when inventory held by wholesalers is exhausted. The remaining benefit period is expected to be up to six months from the date of withdrawal. As a result, benefits are expected to cease from October this year. The reason for the withdrawal of Lyxumia is unknown. However, the rapid decline in revenue after the advent of competitive products is believed to have had some effect on supply disruption. Licensed as a GLP-1 analogue in March 2013 by the Food and Drug Administration, the product was approved for glycemic control, diet and exercise therapy supplements in adult patients with type 2 diabetes. At the time of the market, once-daily administration showed attention for lowering blood sugar regardless of pre and post meal. However, Lixumia's sales declined sharply in 2016 because of Trulicity (Dulaglutide), which is administered once a week with the same GLP-1 analog family. According to drug market research institutes such as IQVIA, The sales fell to ₩500 million in 2018 and ₩300 million in 2019. The poor performance of the family of GLP-1 analogs administered once a day is not the only Lixumia. Victoza (Liraglutide) by Novonordisk and Byetta(Exenatide) by Astrazeneca also fell below ₩100 million. At present, the market size of the GLP-1 analogue is known to be about ₩30 billion. The industry believes that it is due to the convenience of once-weekly administration and Trulicity, which is recognized for its combination with basal insulin at the end of 2017. The long-lasting product has reduced the rejection of injections. This is a long-lasting product that has increased convenience. The market grew, but as Trulicity encroached, once-daily dosing products gradually disappeared.
- Product
- Taltz for interleukin Psoriasis expands Rx Area in hospitals
- by Eo, Yun-Ho Jan 06, 2020 06:22am
- Lilly's interleukin IL-17A antagonist, Taltz, is settling on its prescription area at general hospitals. According to the related industry on the 6th, psoriasis treatment Taltz (Ixekizumab) passed the drug commitee (DC) of Seoul National Hospital recently after Asan Hospital and Severance Hospital among the Big Five. In addition, prescription codes have been made into major hospitals such as Seoul National University Bundang Hospital and Pusan National University Hospital. Taltz, which was released in late 2018, was slower to be priscribed in general hospitals compared to Janssen's IL-23 antagonist Tremfya (Guselkumab), which had entered the same period. Novartis' Cosentyx (Secukinumab) which has the same mechanism as Taltz, entered the market earlier. For this reason, Taltz had difficulty, but it is gradually expanding being prescribed. Accordingly, the issue is how the competition between interleukin drugs in the psoriasis treatment field will change. Lilly has also published the latest IXORA-R study in patients with moderate-to-severe plaque psoriasis , which directly compares the efficacy of Tremfya and Taltz. As a result, the Taltz-administered group achieved a statistically significant level of skin improvement compared to the Tremfya-treated group, which achieved 41.3% of patients with complete clean skin (PASI 100), the primary endpoint at week 12. All major secondary endpoints were also met. The reaction rate were higher than Tremfya at the 2nd PASI 75 ratio, the 4th and 8th PASI 90 ratio, the 4th and 8th and 24th PASI 100 ratio, the 12th week's static Physician's Global Assessment (sPGA) 0 ratio, and the 1st PASI 50 ratio. Andrew Blauvelt, Ph.D., working at the University of Oregon Research Center, USA, said, “The results of IXORA-R showed that Taltz is an effective treatment that helps more patients reach complete clean skin at week 12 of treatment, as early as the first week of treatment, skin plaque lesions improved by 50%”. In the IXORA-R clinical trials, the safety profiles of Taltz and Tremfya were similar to those reported previously. Lilly plans to announce in 2020 the results of the 24th week PASI 100 attainment rate, one of the key secondary endpoints for the IXORA-R trial.
- Product
- Samsung completes Phase III of Lucentis Biosimilar
- by Lee, Seok-Jun Jan 02, 2020 06:08am
- Samsung Bioepis’ Lucentis Biosimilar (SB11)' phase III completed. Lucentis' global sales last year were about ₩4.2 trillion. Samsung Bioepis recently announced its last patient visit for the SB11 phase III trial was done. Phase III examined compared non-inferiority with the original Lucentis in a total of 705 patients with Wet Age-related Macular Degeneration from Mar 2018. Lucentis is a treatment for eye diseases such as macular degeneration and diabetic macular edema, jointly developed by Roche’s subsidiary Genentech and Novartis, multinational pharmaceuticals. Samsung Bioepis plans to announce the results of phase III next year and proceed with the marketing authorization application process in Europe and the United States. SB11 is Samsung Bioepis' first eye disease treatment project. Meanwhile, Samsung Bioepis recently signed marketing partnerships about two types of eye disease treatment pipelines (SB11: Lucentis Biosimilar, SB15: Ilia Biosimilar) with Biogen, including the US and Europe. SB15 is a case that recognized market value from partners even though it is a candidate for clinical preparation.
- Product
- Oral treatment by Sanofi has the highest number of defects
- by Kim JiEun Dec 26, 2019 06:30am
- Ten defects were found in the same product of the same company a year The Seoul Pharmaceutical Society (Chairman Dong-ju Han), the pharmacy committee (Vice chairman Yong-seok Choi, Head Commissioner Woo-young Chang, Soo-hyun Byun, and Tae-seok Kang), and the Pharmacist Guidance Committee (Chairman Kyung-Jin Jeon and Soo-Yul Lim) announced the results of the submission of bad drugs this year. The Seoul Pharmaceutical Society has received reports from member pharmacies since this year by promoting the operation of the bad drugs report center. According to the Seoul Pharmaceutical Society, a total of 174 cases were received and processed at 70 member pharmacies in 24 Seoul branches. The most common types of defective drugs received were simple damages (33 cases), including ▲shortage of 17 cases, ▲poor PTP packaging, ▲12 poor tablets wear, ▲8 poor inspections, ▲8 poor tablet and poor coating, ▲ Seven PTP and bad inspection ▲4 bad inspection, ▲4 bad sealing, ▲4 bad containers, ▲Three poor granulation and bad tablets. In addition, there were discoloration and change of appearance, mixing of other products, mixing of foreign materials, poor filling, and excessive quantities. The Seoul Pharmaceutical Society received the most reports with 10 cases of oral infectious drugs by Sanofi. It recently announced that a recovery action was requested for the serial number and expiration date. In addition, a total of 136 items were reported. The Seoul Pharmaceutical Society receives a reception for defective drugs through text messages (Tel: 010-3568-5811), and the member pharmacy receives the name of the department, the name of the pharmacy, the name of the pharmaceutical company, the name of the drug, the manufacturing number/expiration date, and the bad drug picture. The pharmacy society asked for information from member pharmacies as it could be used as a basis for data accumulation even when pharmacies and pharmaceutical companies (wholesalers) provided their own drugs. They also pointed out that even though there are many cases of drug shortages in pharmacies, it is not easy to pass on because of lack of proof. In this case, they also asked for active reporting. Chairman Dong-ju Han said, “Defective drug notification serves as an important stepping stone to improve the quality and to ensure the safety of patients by identifying the cause and preventing recurrence and awaits the participation of member pharmacies”.
- Product
- “Lipitor not sold out”, Pfizer clarifies rumors
- by Kim JiEun Dec 18, 2019 06:24am
- Rumors of cholesterol lowering treatment Lipitor tablet going out of stock have been spreading throughout major pharmacies, but its company clarified the rumor is false. Pfizer Upjohn Korea on Tuesday told Daily Pharm that the manufacturing of Lipitor 10 mg tablet (28BLP/ 90BLT) has no issue at the moment. The company’s statement was an answer to the recent Lipitor stock-out rumor spreading among pharmacies. Apparently, the talk of Lipitor going out of stock until next year was looming from pharmacists on social media channels and triggered some pharmacies to start hoarding the product. With a temporary spike in order volume, a certain distributor had to notify their salespeople to explain about the situation of pharmaceutical distributors and online shops having scarce stock of the drug to their designated pharmacies. The distributor informed their salespeople that “Lipitor has been sold out due to a baseless false rumor. It has affected all distributors in Korea. As temporary demand in other products has been increasing as well, please visit designated pharmacies to explain the situation”. The whole situation has caught the global company by surprise. The company has been ordinarily manufacturing and supplying the item, when the order has exponentially hiked because of the rumor. Pfizer Upjohn Korea official told, “As of afternoon of Dec. 16, Lipitor 10 mg supply is in a normal state. The company has confirmed Lipitor’s stock in some pharmacies have been sold out. To minimize inconvenience of doctor, pharmacist and patient, the company is in process of studying the origin of the rumor and resolving it”. The company also explained the drug is supplied based on market demand projection and it notifies the public when it predicts drug stock-out from plausible cause. “The company provides products considering a projection based on multiple years of collected market supply and demand data. Accordingly, supply amount is regularly monitored. When the company expects a setback in supply process, the company informs organizations of distributor and pharmacist to minimize turmoil in the market”, the official elaborated. “Pfizer Upjohn Korea would continue to strive for stable supply of drug”, the company official added.