- LOGIN
- MemberShip
- 2026-05-01 23:44:14
- Company
- Alteogen signs 2 licenses out agreements with AZ subsidiary
- by Cha, Jihyun Mar 18, 2025 05:56am
- Alteogen has signed two licensing-out agreements with AstraZeneca’s subsidiaries. According to the Financial Supervisory Service on the 17th, Alteogen has signed 2 exclusive license agreements with MedImmune, a subsidiary of AstraZeneca's bio R&D, for its subcutaneous (SC) formulation modification platform 'ALT-B4' based on recombinant human hyaluronidase enzyme. The company signed agreements with the UK subsidiary of MedImmune and the US subsidiary of MedImmune, respectively. The non-refundable upfront payment with the UK entity was KRW 36.4 billion. The technology fee (milestone payments) for development and commercialization is KRW 1.547 trillion, with separate sales royalties upon successful commercialization. Alteogen will receive sales royalties equivalent to a certain percentage of the net sales generated after the first commercial sale of products using ALT-B4. The upfront payment for the contract signed with the US corporation is KRW 29.1 billion. The milestone payment is 843.8 billion won, with separate sales royalties. The total upfront payment for the two technology export contracts signed by Alteogen is KRW 65.5 billion. The total milestone payment for the 2 contracts is KRW 1.8985 trillion. Alteogen's ALT-B4 technology can change an intravenous (IV) formulation to a subcutaneous (SC) formulation by hydrolyzing hyaluronic acid under the skin. Unlike the IV formulation that patients have to receive for 4-5 hours in the hospital, the SC formulation allows patients to self-inject the treatments at home in 5 minutes. Since 2019, Alteogen has signed a series of technology transfer agreements with global pharmaceutical companies including Merck (MSD) in the United States, Intas Pharmaceuticals in India, and Sandoz in Switzerland. Alteogen has licensed out its ALT-B4 technology to the global pharmaceutical company GPC for USD 1.373 billion in 2019, MSD for USD 3.865 billion in 2020, Intas for USD 109 million in 2021, and Sandoz for USD 145 million in 2022. Among these, the contract with MSD was extended by 4 months in February last year, increasing the contract amount by USD 432 million to USD 4.317 billion. The contract with Sandoz, signed in 2022, was replaced by a contract to develop an SC formulation biosimilar through the joint development of a new hyaluronidase in July. The specific terms and conditions of the contract and development strategy were not disclosed.
- Company
- Can the myelofibrosis drug Ojjara pass CDDC review in KOR?
- by Eo, Yun-Ho Mar 18, 2025 05:56am
- Industry attention is rising on whether GSK’s new drug for myelofibrosis, Ojjara, will be able to make progress in its reimbursement journey and be listed in Korea. According to industry sources, GSK's myelofibrosis treatment Ojjara (momelotinib) will be presented for review to the Health Insurance Review and Assessment Service's Cancer Disease Deliberation Committee tomorrow (19th). Specifically, the indication being reviewed is its use as a ‘treatment for adults with anemia at intermediate or high risk of myelofibrosis.’ The drug is currently indicated for primary myelofibrosis, post-polycythemia vera myelofibrosis, or post-essential thrombocythemia myelofibrosis. GSK recently launched Ojjara as a non-reimbursed drug. It remains to be seen whether the application will lead to the introduction of a covered treatment option that will significantly improve anemia, which has remained an unmet need in the treatment of myelofibrosis in Korea. Ojjara has a unique triple-inhibition mechanism that blocks JAK1, JAK2, and ACVR1 (Activin A Receptor Type 1). In the treatment of myelofibrosis, inhibition of JAK1 and JAK2 can contribute to the improvement of systemic symptoms and reduction of splenomegaly in patients, while inhibition of ACVR1 can help alleviate anemia by inducing a reduction in hepcidin expression. Managing anemia is one of the unmet needs in the treatment of existing patients with myelofibrosis. Anemia, which increases the need for blood transfusions, causes more than just dizziness, and depending on the severity, it can lead to a serious condition that can be life-threatening. The Phase III SIMPLIFY-1 and MOMENTUM studies have shown that, regardless of prior treatment with JAK inhibitors, Ojjara can significantly improve the main symptoms of splenomegaly and transfusion dependence in the treatment of patients with myelofibrosis with anemia. In the SIMPLIFY-1 study, which confirmed the clinical efficacy and safety of Ojjara compared to that of JAK inhibitors in the first-line treatment of patients with myelofibrosis who had no prior experience with JAK inhibitors, Ojjara demonstrated non-inferiority to JAK inhibitors in the primary endpoint of spleen volume response at Week 24 of treatment. The proportion of transfusion independence in each patient group was 66.5% for the Ojjara arm and 49.3% for the ruxolitinib arm, indicating that Ojjara showed significantly less transfusion dependency. “While JAK inhibitors, which were used in the treatment of myelofibrosis, showed effects in alleviating splenomegaly and systemic symptoms, they worsened anemia or increased the need for blood transfusions, which left an unmet need,” said Seo-Yeon Ahn, Professor of Hematology & Oncology at Chonnam National University Hwasun Hospital, “Ojjara has confirmed its significant clinical value in managing anemia, which is closely related to the prognosis of patients with myelofibrosis, and we expect that its launch in Korea will contribute to improving the treatment outcomes and quality of life of more patients.”
- Company
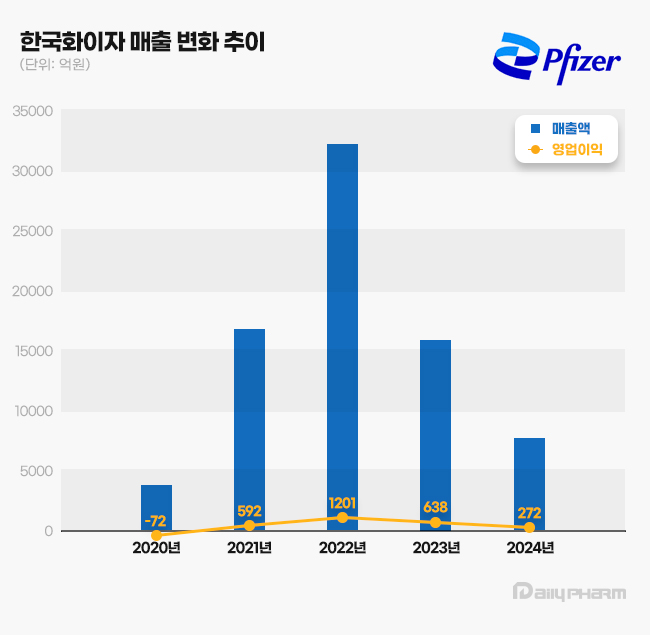
- Pfizer distributed KRW 160 billion dividend over 2 years
- by Chon, Seung-Hyun Mar 18, 2025 05:56am
- Pfizer Korea has decided on a large-scale dividend payout for the second consecutive year. While the company maintained an annual dividend policy of KRW 12.48 million, it has distributed KRW 160 billion to its parent company over the past two years. Large-scale profits during the COVID-19 pandemic enabled this high dividend. According to the Financial Supervisory Service (FSS) on the March 15, Pfizer Korea’s total dividend payout last year amounted to KRW 60.01 billion. It included an interim dividend of KRW 60 billion and an additional annual dividend of KRW 12.48 million. Pfizer Korea’s 2024 interim dividend was applied a 6,501% face value dividend rate, meaning shareholders received over 60 times the face value of KRW 5,000 per share. The interim dividend was distributed to 172,104 common shares, totaling KRW 55.9 billion, and 12,480 preferred shares, amounting to KRW 4.1 billion. Pfizer Korea made the second consecutive year of large-scale dividend payments after 2023. In 2023, the company distributed an interim dividend of KRW 100 billion and an annual dividend of KRW 12.48 million. The 2023 interim dividend was applied at a 10,835% face value dividend rate. Pfizer Korea The largest shareholder of Pfizer Korea is Pfizer’s subsidiary in the Netherlands, 'PF OFG South Korea 1 B.V.,' which holds a 99.99% stake in the company. Pfizer Korea significantly increased its dividend payouts as its sales surged during the COVID-19 pandemic. In 2020, Pfizer Korea recorded KRW 391.9 billion in sales. The sales more than quadrupled to KRW 1.694 trillion in 2021 and further skyrocketed to KRW 3.2254 trillion in 2022, an eightfold increase in sales over two years. Although its 2023 sales fell to KRW 1.6018 trillion, roughly half of the previous year's, it still was a fourfold increase compared to 2020. In 2020, Pfizer Korea reported an operating loss of KRW 7.2 billion and a net loss of KRW 21.2 billion. However, in 2022, its operating profit and net profit surged to KRW 120.1 billion and KRW 119.5 billion, respectively. In 2023, the company reported KRW 59.2 billion in operating profit and KRW 84.9 billion in net profit. Pfizer began its messenger RNA (mRNA) vaccine development in March 2020 in collaboration with BioNTech, using BioNTech's mRNA technology and Pfizer's extensive experience in global clinical trials. Within less than a year of COVID-19 spread, Pfizer successfully developed Comirnaty, a vaccine with 95% preventive effect. The U.S. Food and Drug Administration (FDA) granted it Emergency Use Authorization (EUA) in December 2020, and it received EUA approval in Korea in March 2021, leading to its full-scale supply through Pfizer's Korean subsidiary. Pfizer later developed the COVID-19 antiviral treatment Paxlovid. The Korean government used Paxlovid as a COVID-19 treatment. Pfizer Korea continued implementing a unique dividend policy, consistently paying KRW 12.48 million in annual dividends. The company calculates its dividend payout based on a 20% dividend rate applied to preferred share capital, which totals KRW 62.4 million. Pfizer Korea's total capital stands at KRW 922.92 million, with KRW 860.52 million in common shares (172,104 shares) and KRW 62.4 million in preferred shares (12,480 shares). Since 2005, Pfizer Korea has maintained a consistent dividend payout of KRW 12.48 million annually, based on a 20% preferred share dividend rate, except for four instances in the past 20 years. However, in 2017, the dividend payout was set at KRW 79.794 billion, exceeding net profit. At that time, the dividend rate was set at 660% of the face value (KRW 5,000 per share) for both common (2,455,520 shares) and preferred shares (12,480 shares), resulting in a significantly expanded payout. In 2008, the company allocated KRW 190 billion in dividends despite posting a KRW 600 million deficit that year, setting a 3045% dividend rate based on the face value. However, whether Pfizer Korea's high dividends will continue remains uncertain. In 2023, revenue fell 51.1% year-over-year to KRW 783.7 billion. Operating profit declined 57.4%, from KRW 63.8 billion to KRW 27.2 billion. The company's 2023 revenue and operating profit decreased 75.7% and 77.4%, respectively, compared to 2022, mainly due to the impact of entering endemic and declining COVID-19-related sales.
- Company
- Sanofi 'Dupixent' adds COPD indication
- by Whang, byung-woo Mar 17, 2025 06:00am
- 'Dupixent,' dominating the market for atopic dermatitis·asthma drugs, has expanded treatment areas to chronic obstructive pulmonary disease (COPD), thus gaining attention. Since Dupixent was approved for expanded reimbursement last year, the latest news on COPD indication will likely improve its prescription competitiveness. Product photo of DupixentAccording to pharmaceutical industry sources, Dupixent (dupilumab) was approved by the Ministry of Food and Drug Safety (MFDS) on the 13th for expanded indication to include add-on maintenance therapy to treat adult COPD who have an elevated blood eosinophil count and are not adequately controlled by standard inhaled therapy. Patients with COPD experience decreased quality of life due to trouble breathing, fatigue, and acute exacerbation, and the disease may lead to death if worsened. However, even if triple combination therapy, consisting of conventional inhalation therapy, is used, about 50% of patients still experience severe worsening. Therefore, patients have unmet needs for treatment. Dupixent works by targeting signaling pathway of interleukin (IL)-4 and IL-13, which are primary cause of type 2 inflammation. Dupixent became the first and only domestically approved targeted biologic medication for COPD. The drug is expected to provide new clinical benefits to patients with COPD who do not respond to conventional treatments. The current approval was based on two Phase 3 clinical studies, demonstrating a reduction in COPD annual rate of exacerbations and a significant improvement in lung function and patient quality of life. According to the Phase3 BOREAS and NOTUS, serving basis of expanded indication, at 52 weeks of Dupixent administration, the annual rates of moderate or severe exacerbations were 0.78 and 0.86, respectively, which were 30% and 34% lower than the 1.1 and 1.3 of the placebo group. The results met the primary endpoints for efficacy. Additionally, improvement in lung function started to show from week 2 of administration and maintained up to week 52. In the BOREAS and NOTUS clinical studies, the prebronchodilator FEV1 at week 12 increased from 77 mL and 57 mL of the placebo group to 160 mL and 139 mL with Dupixent, and FEV1 at week 52 increased from 70 mL and 54 mL to 153 mL and 115 mL, demonstrating significant improvement in effects. The St George's Respiratory Questionnaire (SGRQ) score of 4 or above had improved from 43% and 47% of the placebo group to 51.5% and 51.4% of the Duxient group. Both clinical studies showed a consistent safety profile of Dupixent, similar to previous studies. Kay Bae, Sanofi-Aventis Korea Country Lead, said, "COPD has a high disease burden due to acute exacerbations and lung function deterioration. However, it has high unmet needs because there are patients whose symptoms are not regulated with conventional treatments," adding, "The diagnosis rate is low. Thus, many patients who require treatment are not properly being treated." "As Dupixent is the first-and-only approved targeted biologic medication, it is expected to provide a new treatment paradigm for COPD. We hope that current approval will allow more patients with COPD to receive treatments, thereby improving their symptoms and quality of life," Bae said. According to Sanofi's reporting on performance, Dupixent's global sales for last year amounted to EUR 13.11 billion (approximately KRW 19.7 trillion), which was an increase of 23.1% from EUR 10.715 billion in 2023. The fourth quarter sales amounted to EUR 3.5 billion (approximately KRW 5.3 trillion), showing an increasing trend. In South Korea, Dupixent's sales surpassed KRW 100 billion, with KRW 105.2 billion in 2022, based on IQVIA. After that, it recorded KRW 143.2 billion in 2023, showing an increasing trend. Additionally, the impact of Dupixent is anticipated to grow due to the approval of expanded indication to include severe atopic dermatitis and drug switching between JAK inhibitors. Dupixient is effective in diseases that occur due to type 2 inflammation, including atopic dermatitis, asthma, chronic rhinosinusitis with nasal polyps, and eosinophilic esophagitis. It is expected to add indications in diseases with similar mechanisms. Sanofi is conducting Phase 3 clinical trials on Dupixent, aiming to secure indications in chronic spontaneous urticaria (CSU), chronic pruritus of unknown origin (CPUO), and bullous pemphigoid (BP).
- Company
- K-Bios busy developing new CAR-NK cell therapies
- by Son, Hyung Min Mar 17, 2025 06:00am
- The domestic pharmaceutical industry is initiating clinical trials for cell therapies and confirming new possibilities. GC Cell, Isu Abxis, Vaxcell Bio Therapeutics, GI Cell, and HK Inno.N are developing CAR-NK (chimeric antigen receptor) cell therapies. CAR-NK, which is derived from allogeneic cells, has the advantage of being able to compensate for side effects caused by the use of CAR-T cell therapies. CAR-T induces cytokines such as interleukin (IL) associated with neurotoxicity, but activated NK cells generally produce interferon-gamma (IFN-γ) and granulocyte/macrophage colony-stimulating factor (GM-CSF). As such, CAR-NK cell therapies are less likely to cause cytokine release syndrome (CRS) and neurotoxicity. CAR-T cell therapy is an immune cell therapy drug created by combining genetic information that expresses CAR into the patient's T cells. CAR-T has been proven effective in hematologic cancers and has been successfully commercialized, but its disadvantages include the lack of solid tumor indications, complex production process with CRS side effects, and high cost. GC Cell drives the development of CAR-NK cell therapy#EB According to industry sources on the 15th, GC Cell has recently started the first patient administration of GCC2005 (AB-205), a CAR-NK cell therapy candidate, in a Phase I trial. GCC2005 is a CAR-NK cell therapy that has improved the short duration of existing NK cells and enhanced efficacy by co-expressing CAR and IL-15. GC Cell has developed a method for culturing large quantities of highly active, high-purity NK cells from a small amount of cord blood. The Phase I trial aims to evaluate the safety and tolerability of GCC2005 in up to approximately 48 patients with relapsed/refractory NK and T-cell malignancies and to determine the maximum tolerated dose (MTD) and recommended Phase II dose (RP2D). Artiva Biotherapeutics Last November, GC Cell signed a third-party license agreement with its US affiliate, Artiva Biotherapeutics, and MSD to develop and commercialize two CAR-NK candidate substances, GC2005 and AB-201, using the affiliate’s intellectual property rights. The three companies terminated their joint research agreement in June last year, but will now work together again with this agreement. The candidate substances included in this contract utilize GC Cell’s CAR-NK platform technology and are new anticancer drug candidates that were previously developed through joint research between Artiva and MSD. According to GC Cell, the difference between this contract and the contract terminated in June is the main entity for research and development. CAR-NK development will be carried out by GC Cell, whereas Artiva was the main entity in the previous agreement. GC Cell has secured global exclusive rights to the CAR-NK candidate substances and will lead the research and development (R&D) in the future. In preclinical studies, GCC2005 showed anticancer effects in vivo in various CD5+ T-ALL models (RPMI-8402, CCRF-CEM). GCC2005 showed higher survival rates and tumor suppression efficacy compared to the control (vehicle). GC Cell is also developing the CAR-NK therapy AB-201. AB-201 targets solid tumors such as HER2-overexpressing breast cancer and gastric cancer. Artiva received approval from the US Food and Drug Administration (FDA) in 2022 for a Phase I/II clinical trial plan (IND) for AB-201. Since then, GC Cell has been conducting clinical trials after receiving approval for the Phase 1 IND for AB-201 from the Ministry of Food and Drug Safety and the Human Research Ethics Committee (HREC) of Australia in December last year. Through the trial, GC Cell plans to evaluate the safety and tolerability of AB-201 in patients with HER2-positive solid tumors and determine the recommended Phase II dose. Domestic companies also confirm the possibility of developing CAR-NK cell therapies Isu Abxis also recently released preclinical trial results and began full-scale development of CAR-NK cell therapies. The company's ISU104, which is under development, targets the HER3 (ErbB3) protein, which is mainly expressed in breast cancer. Isu Abxisand Seok-Ho Kim, a professor at Dong-A University's Department of Biomedical Engineering, has developed CAR-NK cells that have been engineered to express CAR cells targeting ErbB3 from cord blood-derived NK cells. The results of the research showed that when treating breast cancer cell lines with CAR-NK cells, the cancer cells were killed. Furthermore, in a mouse model implanted with breast cancer cells, ISU104-CAR-NK showed the effect of reducing the size of the tumor without any particular side effects. To date, no HER3-targeted anticancer drugs have been developed yet. Patritumab deruxtecan, an antibody-drug conjugate (ADC) that is in Phase II clinical trials by Daiichi Sankyo and MSD, is currently developed the furthest. Last year, Vaxcell Bio signed a business agreement with Bio Design Lab, a virus vector design company, to jointly develop CAR-NK cell therapies for the treatment of autoimmune diseases. Previously, Vaxcell Bio has signed MOUs with Bio Design Lab, Samsung Medical Center, and others to conduct research and development of new CAR-NK cell therapies. Under this agreement, Vaxcell Bio will provide third-generation NK cells and oversee the entire process of CAR-NK development, including research and clinical trials that meet GMP standards. Bio Design Lab will independently design and produce lentivirus vectors, one of the core technologies in CAR-NK cell therapy development. GI Innovation's affiliates GI Cell and Y Biologics signed a memorandum of understanding last year and began developing CAR-NK anticancer drugs. The two companies plan to develop anticancer drugs through GI Cell's CAR-NK cell therapy development and mass culture technology and Y Biologics' antibody discovery platform technology, Nanobody. GI Cell has completed a Phase I clinical trial in Korea for its NK cell therapy, T.O.P. NK, for patients with solid and hematological cancers. After proving the tolerability and safety of T.O.P. NK, GI Cell has been conducting a Phase IIa clinical trial since last month. GI Cell is embarking on joint research with HK Inno.N for a CAR-NK cell therapy. HK Inno.N and GI Cell are conducting basic research on 7 targets together. HK Inno.N is conducting two basic research projects on its own in addition to the joint research.
- Company
- BESREMi lands in Big 5 Hospitals in Korea
- by Eo, Yun-Ho Mar 17, 2025 05:59am
- BESREMi, a new drug for polycythemia vera, has been approved for prescription at tertiary hospitals in Korea. According to the industry sources, PharmaEssentia Korea's BESREMi (ropeginterferon alfa-2b, a treatment for polycythemia vera, has recently passed the Drug Committees (DCs) of the Big 5 medical institutions in Korea, including Samsung Medical Center, Seoul National University Hospital, Seoul St. Mary's Hospital, Asan Medical Center, and Severance Hospital. As the drug’s reimbursement review is underway, the drug’s reimbursement listing is expected to quickly lead to actual prescriptions. Polycythemia vera is a rare blood disorder where a somatic cell mutation in the bone marrow abnormally activates bone marrow function and produces excessive red blood cells. It has a short survival period and is so fatal that 10~15% of patients with polycythemia vera develop myelofibrosis or leukemia within 10 years. Although hydroxyurea had been used as the standard of care, it was difficult to fundamentally cure the disease with hydroxyurea, and patients who could not be treated with hydroxyurea had limitations as there were practically no drugs available for them in Korea’s domestic reimbursement environment. BESREMi, an interferon treatment that selectively removes JAK2 mutations that cause polycythemia vera. In Korea, the drug received approval in October 2020 to treat low-risk and high-risk patients with polycythemia vera without symptomatic splenomegaly. The drug demonstrated its potential as a radical treatment for polycythemia vera in patients who had not received cytoreduction therapy or received less than 3 years of treatment with hydroxyurea. Therefore, whether the only interferon treatment option approved for polycythemia vera will be born in Korea is receiving attention. BESREMi demonstrated its efficacy and safety in the Phase III PROUD/CONTINUATION-PV trial that was conducted on polycythemia vera patients. Trial results showed that 53% of the patients in the Besremi arm achieved a complete hematological response, an improvement compared with the hydroxyurea patient arm (38%). The hematologic and molecular response rates at 72 months were also high, at 80.4% and 65.3% in low-risk and high-risk patients, respectively. Regardless of their risk, patients treated with Besremi did not require phlebotomy even 6 years after administration. Besremi is recommended as a first-line or second-line treatment for polycythemia vera in the National Comprehensive Cancer Network (NCCN) and European Leukemia Network (ELN) guidelines, regardless of previous treatment experience.
- Company
- Radioligand 'Pluvicto' available at tertiary gen hospitals
- by Eo, Yun-Ho Mar 17, 2025 05:59am
- Product photo of Pluvicto 'Pluvicto,' a new drug for prostate cancer, is now available for prescription at tertiary general hospitals. According to industry sources, Novartis Korea's targeted radioligand therapy, Pluvicto (Lutetium vipivotide tetraxetan), has passed the drug committees (DC) of the 'Big 5' hospitals, including Samsung Medical Center, Asan Medical Center in Seoul, Seoul St. Mary's Hospital, and Sinchon Severance Hospital, as well as the 17 medical institutes nationwide. A radioligand combines a therapeutic radioactive isotope with a ligand (targeted substance). A therapeutic radioactive isotope is released when radioligand binds to a targeted cell. This mechanism suppresses the proliferation of cancer cells. The MFDS designated Pluvicto, a treatment for metastatic castration-resistant prostate cancer (mCRPC), as the 6th medicine to be added to the 'Global Innovative products on Fast Track (GIFT)' following acknowledgment of the drug's innovativeness in June 2023. It was officially approved in May of last year. However, Pluvicto is still a non-reimbursed drug. Novartis is now preparing to apply for an insurance reimbursement listing. Pluvicto is a radioligand used to treat patients with prostate-specific membrane antigen (PSMA)-positive mCRPC who have been treated with androgen receptor (AR) pathway inhibition and 'taxane'-based chemotherapy. Pluvicto has been regarded as the next-generation innovative therapy that delivers a therapeutic radioisotope to prostate cancer cells by binding the radioisotope 177Lu to PSMA, ultimately killing cancer cells. Pluvicto was designated as an innovative drug and priority review drug by the U.S. Food and Drug Administration (FDA) and obtained FDA approval (March 2022). It was acknowledged for its innovativeness overseas. Meanwhile, the efficacy of Pluvicto was demonstrated through the Phase 3 VISION study. The combination therapy containing Pluvicto and the optimal standard therapy in study participants who have been previously treated with androgen receptor (AR) pathway inhibition and 'taxane'-based chemotherapy reduced mortality risk by 38% compared to the standard therapy alone. It has also statistically reduced the radiographic disease progression or death by 60%. Furthermore, approximately 1/3 (30%) of patients in the combination therapy group, treated with Pluvicto+standard therapy, who can be evaluated showed an objective response, compared to 2% of the monotherapy group.
- Company
- Lotte Biologics signs MOU with Asimov for CDMO business
- by Whang, byung-woo Mar 14, 2025 05:57am
- (Fromt the left) James Park, CEO of Lotte Biologics, Alec Nielsen, co-founder and CEO of Asimov Lotte Biologics (CEO: James Park) announced on the 13th that it has signed a memorandum of understanding (MOU) with Asimov, based in Boston, Massachusetts, for collaboration in the contract development and manufacturing organization (CDMO) business. Through the agreement, Lotte Biologics will provide services covering the entire process from cell line development to GMP (Good Manufacturing Practice) production by utilizing Asimov's next-generation cell line development platform technology. This is expected to further the companies’ CDMO capabilities and track record. Earlier, Lotte Biologics successfully confirmed its scale-up and mass production capabilities through a test applying Asimov's proprietary cell line development platform, CHO Edge system, at the Syracuse Biocampus. This agreement is part of a collaboration to provide full-scale end-to-end services to clients based on these achievements. Asimov's CHO Edge system is a synthetic biology-based cell line development platform that accelerates the production of antibody and protein therapeutics through the genetic design and optimization of Chinese hamster ovary (CHO) cells. In particular, it supports the rapid development of customized high-productivity and uniform cell lines by applying automated work processes and AI-based analysis. In addition, the CHO Edge system combined with Lotte Biologics' GMP manufacturing capabilities enables a smooth transition from cell line development to commercial production. The company believes that this will be an important turning point in expanding the scope of Asimov's business along with the expansion of its CDMO business. In addition, the signing of the MOU is expected to maximize the benefits of Lotte Biologics' US production base. James Park, CEO of Lotte Biologics, said, “I hope this agreement will serve as an opportunity to maximize the strengths of both companies. We will continue to provide differentiated services to our customers by bringing synergy with Asimov's technology and Lotte Biologics' production capacity, while striving to become a company that contributes to the development of the bio-industry and, ultimately, to the improvement of the quality of life of patients.”
- Company
- "More BTK inhibitor options for MCL, Brukinsa as alt choice"
- by Whang, byung-woo Mar 14, 2025 05:57am
- Changes to the treatment settings have been brought to Mantle cell lymphoma (MCL) with the launching of new treatments like BTK inhibitors and implementing reimbursement. As the 2nd-generation BTK inhibitors are introduced, treatment options have been broadened for relapsed or refractory MCL, where treatment options have been limited to 1st-generation BTK inhibitors. Dr. Youngwoo Jeon, a Professor in the Department of Hematology at Yeouido St MaryDuring a meeting with Daily Pharm, Dr. Youngwoo Jeon, a Professor in the Department of Hematology at Yeouido St Mary's Hospital, who runs the only Lymphoma and Cell Therapy-Research Center in South Korea, emphasized the need to discuss ways to effectively utilize treatment options. Mantle cell lymphoma (MCL) is a subtype of non-Hodgkin lymphoma (NHL), accounting for approximately 5% of all NHL cases. MCL shows aggressive clinical features. Unlike typical lymphoma that starts in lymph nodes, MCL occurs in organs where lymph nodes are not located. Patients often transfer from dermatology, oncology, and gastroenterology to hematology. Five years ago in South Korea, the annual new patient number was about 80. Now, that there are about 100-110 new patients each year. "According to international classification standards, extranodal T-cell lymphoma is typically considered indolent; however, once it surpasses a critical threshold, it can proliferate rapidly, like DLBCL, and requires immediate treatment," Dr. Jeon said. "Even if the disease initially progresses slowly, the short time between diagnosis and treatment means clinicians on the front lines inevitably perceive it as aggressive." In the case of MCL, the development of new treatment options, including standard therapies, has been slow, and treatment protocols or strategies have only been established relatively recently. As a result, treatment approaches have been sporadic and often based on available economic discretion, implying that patients have been treated drugs designed for other types of lymphoma treatments. Previously, combination treatments such as CHOP (rituximab+cyclophosphamide/doxorubicin/vincristine/prednisone) were primarily used. Patients with poor ECOG were administered treatments that have lower-toxicity, such as follicular lymphoma treatments like R-CVP (rituximab+cyclophosphamide/vincristine/prednisone). In addition to these options, patients have the choice to pay for BR treatment (bendamustine+rituximab) out-of-pocket. "Expanded option to include BTK inhibitors is regarded favorable, 2nd generation with reduced side-effects gains attention" The remaining issue lies in the relapse and treatment resistance seen in MCL. Many patients present with a poor prognosis at diagnosis, and given the nature of indolent lymphomas, which are notoriously difficult to cure, the sequencing of treatments becomes increasingly critical. Dr. Jeon explained, "Full recovery is challenging for indolent lymphomas like MCL. Thus, we adopt a strategy similar to that used in treating chronic lymphocytic leukemia, focusing on maximizing progression-free survival (PFS). Moreover, because relapse is frequent, it is difficult to aim for a cure from the very first treatment line, so we carefully plan the treatment sequence for when the disease relapses." In response to these unmet therapeutic needs, BTK inhibitors have emerged. In addition to the 1st-generation agents, a 2nd-generation treatment, like Brukinsa (zanubrutinib), has been introduced. Since June 2024, reimbursement for Brukinsa has been expanded to include its use as monotherapy for patients with MCL who have received at least one prior treatment. Dr. Jeon analyzed, "The introduction of oral targeted anticancer agents, such as first- and second-generation BTK inhibitors, has greatly improved patient treatment convenience. Under conventional cytotoxic chemotherapy, patients typically endured cycles of deterioration and improvement over more than a year of hospital admissions, often leading to death. Because patients can self-administer the medication and only need to manage side effects, treatment accessibility has been significantly enhanced." In fact, the 1st-generation BTK inhibitor, Ibrutinib, has shown outcomes that surpass those observed in clinical trials, as evidenced by over seven years of real-world data (RWD). "In about two years, we expect RWD on the second-generation BTK inhibitor, Brukinsa, will become available," Dr. Jeon said. "Based on results with 1st-generation BTK inhibitors, Brukinsa's data are expected to be very favorable. Although it has only been a short time, its satisfactory performance in the clinical setting is very encouraging." Dr. Jeon has highlighted that a characteristic of 2nd-generation BTK inhibitors is their enhanced selectivity for BTK. Typically, when a drug affects unintended targets, it can lead to adverse events such as cardiac hemorrhage, referred to as an off-target effect. 2nd-generation agents like Brukinsa have been designed to minimize these off-target effects. Dr. Jeon said, "These agents have reduced the incidence of hematologic bleeding and cardiac issues seen in clinical trials to around 7–8%, less than half of previous rates, which is very encouraging," adding, "However, in real-world clinical practice, we focus more on managing gastrointestinal disturbances and onycholysis, side effects that can significantly impact patient adherence during long-term treatment." "The criteria for BTK inhibitors are limiting…patient's life-long use of treatments must be considered" Despite both 1st- and 2nd-generation BTK inhibitors being eligible for reimbursement, a significant limitation remains: if a patient fails on a 1st-generation BTK inhibitor, subsequent BTK inhibitor therapy is not reimbursable. Patients only have one opportunity for BTK inhibitor treatment when considering treatment sequencing. Dr. Jeon had about five to six patients on 1st-generation BTK inhibitors that have been switched with Brukinsa. Dr. Jeon explains that while patients already stabilized on 1st-generation therapy typically continue, new patients are generally started on Brukinsa whenever possible. Yet, Dr. Jeon stressed that even though switching between 1st- and 2nd-generation therapies that both target the same molecule can be justifiable, it is critical to remember that these drugs are intended for lifelong use. "Patients on 1st-generation BTK inhibitors often suffer from severe nausea and vomiting that disrupt their daily lives. In such cases, switching to Brukinsa, which has reduced off-target effects, has produced notably positive outcomes," Dr. Jeon said. "It appears that the Health Insurance Review and Assessment Service (HIRA) considers switching unnecessary because the treatment offers a refined version of the same target," Dr. Jeon said. "Because forcing patients to endure painful side effects for a lifetime is like torture, 2nd-generation BTK inhibitors will represent an excellent alternative." Finally, Dr. Jeon emphasized the urgent need to adopt CAR‑T therapies as a third-line treatment for MCL quickly. "While CAR‑T therapy is already being implemented as third-line treatment overseas, it is not yet approved domestically, so they are available for use. However, the HIRA must propose an alternative so Korean patients could eventually receive treatment on par with those in other G20 countries," Dr. Jeon said. "Approximately 85% of patients are filtered out during second-line BTK inhibitor treatment, leaving very few who progress to third-line treatments," Dr. Jeon said. "Although these treatments are high-cost, the financial burden is likely minimal due to the small patient population."
- Company
- Takadea's metastatic colorectal cancer drug Fruzqla wins nod
- by Whang, byung-woo Mar 14, 2025 05:57am
- Takeda Pharmaceutical Korea announced on March 13 that its metastatic colorectal cancer treatment, Fruzqla (fruquintinib), received domestic marketing approval from the Ministry of Food and Drug Safety (MFDS) on March 6. The efficacy·effectiveness of Fruzqla has been demonstrated for adult patients with metastatic colorectal cancer who have previously been treated with a chemotherapy containing fluoropyrimidines, oxaliplatin, and irinotecan plus an anti-VEGF or anti-EGFR agent (for patients with wild-type RAS), and whose disease has progressed or who does not show tolerability following treatment with trifluridine/tipiracil and/or regorafenib. With this approval, a new treatment option is now available to improve overall survival for metastatic colorectal cancer patients who, due to limited options in treating later stages of cancer, have not received adequate treatment. Fruzqla is a small-molecule VEGFR-TKI that selectively targets vascular endothelial growth factor receptors (VEGFR)-1, 2, and 3, thereby selectively inhibiting the neoangiogenesis and lymphangiogenesis of cancer cells. By avoiding unnecessary targets other than VEGFR-1, 2, and 3, the drug achieves high exposure and sustained target inhibition, while its once-daily oral administration enhances patient convenience. The approval of Fruzqla was based on a Phase 3 clinical trial involving 691 adult patients with metastatic colorectal cancer who have received prior treatments. Study results showed that the median overall survival (mOS) in the Fruzqla group was 7.4 months, which is significantly longer than the 4.8 months observed in the placebo group, resulting in a 34% reduction in the risk of death. Additionally, the Fruzqla group had a median progression-free survival (mPFS) of 3.7 months, more than double the 1.8 months in the placebo group, corresponding to a 68% reduction in disease progression or death risk. Mahender Nayak, Takeda Pharmaceutical Korea' Senior Vice President for Growth & Emerging Market, stated, "Fruzqla is a new treatment option for metastatic colorectal cancer that has emerged after over a decade and can be used regardless of specific biomarkers. We expect it to offer favorable tolerability and to improve both quality of life and survival outcomes for patients who needed additional treatment options in the later stages of anticancer therapy." Nayak added, "Fruzqla is already approved in several countries worldwide, including South Korea, the United States, Europe, and Japan. Takeda Pharmaceuticals Korea will continue to work to supply innovative new drugs and enhance treatment accessibility to meet the unmet needs of cancer patients."