- LOGIN
- MemberShip
- 2026-04-30 22:55:32
- Company
- Doctors ‘Reimb too slow for new drugs in Korea’
- by Eo, Yun-Ho May 15, 2025 06:23am
- Most doctors were found to believe that the speed of reimbursement for new drugs in Korea is too slow. The Korean Research-based Pharmaceutical Industry Association (KRPIA) released the results of a survey of 100 domestic medical professionals on the 14th. In January, the global polling agency Ipsos Research surveyed domestic clinical experts from various medical departments to ask their opinions on access to new drugs. According to survey results, all medical professionals unanimously answered that the period from the Ministry of Food and Drug Safety approval to health insurance reimbursement listing is “long,” with 74% stating it is “too long.” Regarding the appropriate period from approval to health insurance listing, 81% of medical professionals answered “up to 10 months,” with 41% deeming “within 6 months” as appropriate. As of 2022, it takes an average of 608 days (approximately 20 months) for innovative new drugs to be approved by the MFDS and listed for health insurance reimbursement in Korea. This is twice the appropriate period cited by most medical professionals (10 months) and significantly longer than in major overseas countries such as Germany (281 days), Japan (301 days), and France (311 days) during the same period. Furthermore, experts directly treating patients in the clinical settings anticipate that the swift and widespread introduction of innovative new drugs will provide substantial benefits for patient care. Eighty-three percent of medical professionals expected that “if drugs already in common use overseas are covered by health insurance in Korea, patient treatment outcomes will improve significantly.” A large proportion of medical professionals (85%) responded that “even for drugs already covered by health insurance, if reimbursement standards are eased to enable early or wider use, patient treatment outcomes will improve significantly.” In addition, 95% of medical professionals urged the MOHW to introduce a “fast-track listing procedure or system” for health insurance coverage, similar to the MFDS's Global Innovate Products on Fast Track (GIFT) system, which shortens the drug approval review period for severe or life-threatening diseases by up to 75%. Medical professionals who participated in the survey also expressed concerns about Korea's low access to new drugs. Ninety-four percent of medical professionals pointed out that “Korea's access to new drugs is lower than overseas,” and 97% answered that “the government must set appropriate and reasonable drug prices to prevent the ‘Korea passing’ phenomenon, where multinational pharmaceutical companies give up the launch of innovative new drugs in Korea due to domestic regulations on pharmaceuticals.” Seventy-six percent of medical professionals were concerned that the proportion of new drug expenditures (13.5%) in total domestic drug expenditures is 60% lower than the OECD average (33.9%), and 88% believed that reimbursement and access to new drugs in South Korea need to be improved to the level of the top 10 OECD countries. Medical professionals identified “enhancing access to innovative new drugs” as the top priority among the four key strategies of the government's Second Comprehensive National Health Insurance Plan (2024-2028). As the government pushes policies to reduce drug costs in response to an aging society, 67% of medical professionals expressed the view that “the budget savings should be reinvested into the health insurance fund.” As the survey respondents were clinical experts, they also requested that the opinions of those working on-site be more actively taken into account in the reimbursement decision-making process. Eighty-eight percent of medical professionals responded that “the opinions of medical professionals should be better reflected in the process of registering drugs for health insurance coverage,” and 80% said that “medical professionals should also be involved in the process of selecting patient population eligible for health insurance coverage.” A KRPIA official stated, “Medical professionals who care for patients on the front lines are deeply concerned about the difficulties patients face in receiving treatment due to delays in the introduction of innovative new drugs. They hope that new drugs will be listed for health insurance reimbursement more swiftly and with a broader scope. We anticipate that the results of this survey will contribute to the government’s fostering of a patient-centered treatment environment and policy design.”
- Company
- Arexvy opening the era of RSV vaccine
- by Whang, byung-woo May 15, 2025 06:22am
- As GSK launches the respiratory syncytial virus (RSV) vaccine Arexvy in South Korea, it will challenge the market on a full-scale. Arexvy is already expanding its market dominance in the global market with its strength as the first RSV vaccine. The company will likely focus on expanding vaccine awareness as it opens the RSV vaccine market for the first time. Dr. Ji-Yong Moon, Professor of Konkuk University On May 14, GSK Korea convened a press conference celebrating the launch of Arexvy, the world's first RSV vaccine. The company showcased a preventative strategy for seniors against RSV and Arexvy's clinical significance. Arexvy received approval from the Ministry of Food and Drug Safety (MFDS) at the end of December 2024 for the 'Prevention of lower respiratory tract disease (LRTD) caused by RSV in adults over 60 years of age and older.' Approval of Arexvy was based on results from two Phase 3 studies, 'RSV OA=ADJ-006' and 'RSV OA=ADJ-004,' involving adults 60 years of age and older. The study results showed that during the first RSV season, Arexvy significantly lowered the RSV-LRTD risk by 82.6% and severe RSV-LRTD risk by 94.1% in participants 60 years of age and older compared to placebo. Furthermore, the efficacy of the vaccine regarding RSV-A-associated LRTD increases and RSV-B-associated LRTD increases were 84.6% and 80.9%, respectively. Dr. Ji-Yong Moon, Professor of Konkuk University's Department of Respiratory-Allergy and Clinical Immunology, explained, "RSV infection causes complications, such as pneumonia, in adults aged 60 years or older and it may require hospitalization or lead to death in severe cases," and added, "Based on a retrospective study, 56.8% of the hospitalized adults over age of 65 had pneumonia and 10.6% of those died." Dr. Moon added, "Despite the high disease burden, awareness of RSV infection is poor, and differential testing is not well implemented, so the disease burden of RSV infection has been underestimated." He said, "RSV infection is as contagious as influenza, but there is no specific treatment other than supportive care, so prevention is most important." (from left) Professor Jacob Lee, Professor of the Division of Infectious Disease at Hallym University Kangnam Sacred Heart Hospital, and, Dr. Ji-Yong Moon, Professor of Konkuk University Experts consider that the launch of the first RSV vaccine, Arexvy, is expected to be significant from a preventive standpoint. Professor Jacob Lee, Professor of the Division of Infectious Disease at Hallym University Kangnam Sacred Heart Hospital, reported that RSV vaccination is already recommended in the United States. Dr. Lee said, "Arexvy showed a preliminary efficacy of 94.6% against RSV-LRTD in adults with one or more comorbidities," and emphasized, "Considering that 84% of domestic adults aged 65 and over have one or more chronic diseases, these data are noteworthy." Dr. Lee stated, "Arexvy was approved in the U.S. in 2023, real-world data on its use have accumulated, and excellent preventive efficacy has been confirmed in actual clinical settings." He said, "The U.S. Advisory Committee on Immunization Practices (ACIP) recommends RSV vaccination for high-risk individuals aged 60–74 and all adults aged 75 and older." Cost and awareness remain challenges… "NIP is necessary from a long-term perspective" Regardless of the preventive value of RSV through Arexvy, it remains uncertain how much influence it will exert in the market. Currently, Arexvy remains non-reimbursed in South Korea. Without a recommendation like that of the U.S. ACIP, there is a need to improve awareness. In particular, the fact that the recommended vaccination age in the U.S. is higher at 75 years old, compared to the domestic approval age of 60 and over, also raises questions. Hyunji Kwon, Business Unit Head at GSKRegarding this, Dr. Lee explained, "In South Korea, the Korean Society of Infectious Diseases is expected to announce a recommendation within this year, but it is unlikely to differ significantly from the U.S.," and added, "To enter the National Immunization Program (NIP), sufficient disease burden research needs to be conducted." Dr. Lee continued, "There is a cost burden and domestic data are lacking, but as the population structure changes, the number of vaccination targets will also increase. Through research data and cost-effectiveness, we can hope for RSV prevention through the NIP in the long term." GSK Korea has stated that it will work to improve awareness through TV advertisements and other means alongside the launch of Arexvy. Hyunji Kwon, Business Unit Head at GSK, added, "RSV is a disease with a great unmet medical need yet remains unfamiliar, so we will work to improve awareness among medical staff and high-risk patients," and added, "In a super-aged society in which, for the first time, the population in their 60s exceeds that in their 40s, we will collaborate with the medical community and the government to increase patient access."
- Company
- Will a new trend emerge for liver cancer treatment?
- by Moon, sung-ho May 15, 2025 06:22am
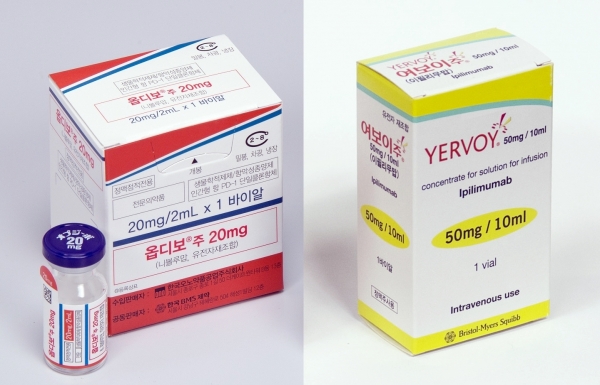
- With new anticancer drugs entering the liver cancer treatment market, where combination therapy has been gaining prominence, attention is focused on whether a paradigm shift will occur. This is because a new competitive landscape is forming with the arrival of newly approved drugs and newly reimbursed drugs. # According to industry sources on the 7th, the US Food and Drug Administration (FDA) recently approved the combination therapy of Bristol Myers Squibb (BMS) and Ono Pharmaceutical's Opdivo (nivolumab)+Yervoy (ipilimumab) combination as a first-line treatment for adult patients with unresectable or metastatic hepatocellular carcinoma (HCC). The approval was based on the results of a randomized, open-label, global Phase III clinical trial (CheckMate-9DW) comparing the combination therapy of Opdivo and Yervoy (335 patients) with either lenvatinib or sorafenib monotherapy (333 patients). The trial was conducted on patients with unresectable or metastatic HCC who had not previously received systemic therapy. The CheckMate-9DW results showed that the median overall survival (mOS) in the Opdivo+Yervoy combination therapy group was 23.7 months (95% CI: 18.8-29.4), compared to 20. 6 months (95% CI: 17.5-22.5) in the control group (n=333), reducing the risk of death by 21% (HR=0.79; P=0.0180). In addition, the 3-year survival rate was 38% in the Opdivo+Yervoy combination therapy group, higher than the 24% in the control group. The objective response rate (ORR) was also significantly higher in the combination therapy group at 36.1% (95% CI: 31–41.5) compared to 13.2% (95% CI: 9.8–17.3; P
- Company
- 'Adempas' for pulmonary hypertension closer to obtain reimb
- by Eo, Yun-Ho May 14, 2025 06:11am
- Product photo of Adempas A new reimbursable treatment option for pulmonary hypertension is anticipated to be introduced. According to industry sources, Bayer Korea has finally reached an agreement with the National Health Insurance Service (NHIS) for its 'Adempas (riociguat).' Accordingly, after 10 years since it was approved in South Korea, Adempas will likely be included in the reimbursement list. Adempas' negotiation was a dosage negotiation, rather than a ceiling price negotiation. Bayer accepted the price 100% below the weighted average price (WAP) of a substitute drug and passed the Drug Reimbursement Evaluation Committee (DREC) of the Health Insurance Review and Assessment Service (HIRA) in February. Adempas was exempted from the drug pricing negotiations. This drug obtained approval in South Korea as an orphan drug in June 2014. Five products with different doses are available, and it has the efficacy and the effect in ▲patients with persistent/recurrent chronic-thromboembolic pulmonary hypertension (CTEPH, WHO Group 4) after surgical treatment or inoperable CTEPH, to improve exercise capacity ▲adult patients with arterial pulmonary hypertension (PAH, WHO Group 1) who have WHO functional class 2-3, to improve exercise capacity. Adempas has been known as the first novel drug to treat CTEPH. CTEPH occurs in patients with chronic pulmonary embolism who progress to chronic obstructive pulmonary disease (COPD) and develop fibrotic stenosis and occlusion, leading to pathological vascular remodeling and increased resistance in the pulmonary arteries. CTEPH is a chronic disease that causes progressive shortness of breath and right heart failure. Symptoms include dyspnea, fatigue, chest pain, dizziness, peripheral edema, cough, and hemoptysis, significantly impacting quality of life. Ultimately, it can progress to heart, kidney, and liver failure, potentially leading to death. Meanwhile, Adempas is a stimulator of soluble guanylate cyclase (sGC), an enzyme found in cardiovascular organs. The efficacy of the drug in patients with chronic thromboembolic pulmonary hypertension (CTEPH) was confirmed in Phase 2 and Phase 3 clinical trials. The clinical trial results showed that Adempas improved the study's primary endpoint physical activity and demonstrated superior tolerability. No unusual adverse reactions were reported. In the CHEST-1 study, results from comparing the 6 Minute Walking Test (6MWT) at 16 weeks from baseline showed that the patient group treated with riociguat had statistically significant improvement compared to the placebo group. In the PATENT-1 study, comparison of changes in 6MWT values at 12 weeks to the placebo group demonstrated statistically significant improvement, meeting the primary endpoint.
- Company
- U.S. executive order on drug price cuts raises hope
- by Kim, Jin-Gu May 14, 2025 06:10am
- U.S. President Donald Trump has signed an executive order to significantly reduce drug prices in the United States. This measure, which focuses on significantly reducing U.S. drug prices in line with overseas prices, is expected to have a positive effect on domestic biosimilars. However, some predict that the expected effects of this executive order will not be sufficient and that there will be no significant impact. Trump signs executive order to lower drug prices in the US... “Up to 90% reduction” According to local media reports on the 13th, President Trump signed an executive order on the 12th (local time) to lower the prices of prescription drugs in the US to the same level as other countries. The order essentially applies the most-favored-nation policy to drug prices in the US. Under the executive order, the US Secretary of Health and Human Services must promote a program that allows US patients to purchase drugs directly from pharmaceutical companies at a “most-favored-nation” price. In addition, the Secretary of Health and Human Services must impose the most-favored-nation pricing on companies within the US pharmaceutical industry within 30 days. This measure is interpreted as a move made to present a kind of price cap to pharmaceutical companies compared to drug prices overseas, and pressure them to lower their prices accordingly. Ultimately, this is expected to improve the intermediate distribution structure of Pharmacy Benefit Managers(PBMs) and further reduce the prices of expensive drugs. President Trump said at a press conference, “What we are trying to do is level the playing field for drug prices,” adding, “The American people will be able to purchase drugs at the lowest prices in the world.” He did not specify which drugs would be subject to price reductions. However, the administration has hinted that the price reduction could be as high as 90%. President Trump said at the press conference, “Drug prices in the United States could be reduced by 59%, 80%, or even 90%.” A day earlier, he had posted on his Truth Social account, “Prescription Drug and Pharmaceutical prices will be REDUCED, almost immediately, by 30% to 80%.” “Original drug price cuts expected to expand market for Korean biosimilars” The domestic pharmaceutical and biotech industry has responded with more optimism than concern to these measures. There are expectations that the price cuts targeting original drugs will have a positive impact on biosimilars. Celltrion said it expects the measure to create “a better business environment.” With the simplification of the intermediate distribution structure, it is expected that “this will provide positive opportunities for Celltrion's U.S. business activities,” and that “the dominance of pharmaceutical companies making high profit selling original products is expected to weaken, which could present market expansion opportunities for biosimilar companies.” Furthermore, it is anticipated that “biosimilar manufacturers will be able to negotiate drug prices directly with the government rather than through intermediaries such as PBMs, which could benefit both the government and manufacturers.” Additionally, it predicted that the prescription of biosimilars will increase as the prices of high-priced drugs are reduced. Currently in the US, through insurance companies and PBM systems, high-priced original drugs are prioritized for inclusion in formularies, followed by limited competition among biosimilar products, resulting in the addition of 2–3 products. During this process, rebates are paid to intermediary distributors, so the burden associated with these rebates will be significantly reduced with the new system. Celltrion stated, “Previously, biosimilar prices were set at the same high level as originals, making it impossible to provide substantial benefits to patients. However, if the intermediate distribution structure is improved through this executive order, the actual prescription prices of biosimilars will decrease, ultimately expanding biosimilar prescriptions to European levels.” Furthermore, it is also expected to provide an opportunity to launch new products in the US market. Celltrion explained, “If parallel imports are activated to supply medicines at most-favored-nation prices in accordance with this executive order, Celltrion will secure the opportunity to launch additional products that have not yet been introduced in the US market.” Another biosimilar company, Samsung Bioepis, is said to have shown a similar response. A positive outlook on the newly announced measure is dominant both inside and outside the company. The background is the expectation that the preferential policy for biosimilars will be strengthened to reduce medical costs. However, a Samsung Bioepis official said, “We are closely monitoring this policy,” without further elaboration. “It is premature to make specific judgements… Its impact on the Korean pharmaceutical industry will be limited The key issue is the strong opposition from the U.S. pharmaceutical and biotech industry. The U.S. pharmaceutical and biotech industry has consistently opposed the U.S. government's repeated attempts to lower drug prices, either by blocking them altogether or minimizing their impact. As a result, the actual evaluated effect of the drug price reductions was then minimal. In fact, President Trump signed an executive order for drug price cuts during his first term in 2018, but it was ultimately scrapped due to opposition from the pharmaceutical industry. At the time, an attempt was made to lower drug prices through an international reference pricing system, but a federal court raised procedural issues and put the brakes on the plan. President Joe Biden also conducted drug price negotiations with major pharmaceutical companies under the Inflation Reduction Act (IRA). However, the actual drug price reductions were limited to 10 drugs, including Eliquis, Xarelto, Januvia, Forxiga, Entresto, Enbrel, Imbruvica, Stelara, and Fiasp. Even these are limited to those who have Medicare - people aged 65 and older - in the United States. The drug price reduction measures will take effect in 2026, and the annual savings in medical costs are estimated at USD 6 billion (approximately KRW 8 trillion). This is considered insignificant compared to the total drug costs in the United States, which amounted to USD 805.9 billion (approximately KRW 1,142 trillion) last year. In this situation, the Trump administration's push for more stringent drug price reduction policies in the second term is expected to face strong opposition from the U.S. pharmaceutical industry. Additionally, criticism has emerged locally that the executive order lacks specific policies. The New York Times (NYT) criticized the executive order, stating that it “did not include specific policies such as pushing for legislation to reduce drug prices or revising drug payment regulations under government health programs.” In the same line, there are also projections that its impact on the domestic pharmaceutical and biotech industry will be limited. A representative from the pharmaceutical industry stated, “So far, only the direction to pursue drug price cuts has been outlined; there are no specific methods for how the cuts will be implemented, nor have the target products been determined.” The official added, “Its impact on domestic pharmaceutical and biotech companies is currently unclear, and even if there is an impact, it is expected to be minimal.” Another industry insider noted, “The recent administrative order requiring the manufacture of pharmaceuticals within the United States, as well as the upcoming pharmaceutical tariff policy expected next week, are likely to have a greater impact on the domestic pharmaceutical and biotech industry than the recent drug price reduction administrative order.”
- Company
- How has Leqembi changed the clinical site in 6 months?
- by Moon, sung-ho May 14, 2025 06:09am
- The overall treatment system on-site has been changing with the introduction of Leqembi, a new dementia drug that was released 6 months ago in Korea. In response, experts are evaluating the therapeutic effects and side effects of Leqembi (lecanemab, Eisai Korea) while demanding improvements to the health insurance reimbursement system in various areas, such as testing costs. At the same time, they are expressing anticipation for other new dementia drugs from global pharmaceutical companies that are likely to be introduced in Korea. The introduction of just one new global drug has changed the atmosphere on-site and in academic activities. #According to the medical community on the 12th, Eisai Korea’s Leqembi (Lecanemab), which was launched in Korea at the end of last year, has passed the Drug Committees (DCs) of major university hospitals and is rapidly expanding its prescriptions in Korea. Leqembi is a new treatment that removes amyloid beta (Aβ), one of the main causative substances of Alzheimer's disease. It specifically binds to soluble amyloid beta protofibrils and insoluble amyloid beta fibrils—the most toxic forms of amyloid beta—to reduce amyloid beta plaques in the brain. In particular, it is the first antibody therapy to receive full approval from the FDA in July 2023 for its efficacy and safety in delaying the progression of Alzheimer's disease and cognitive decline by removing the causative agent. In Korea, it was approved by the MFDS in May last year and has been prescribed in general hospitals since the end of November. However, there are limitations in that it can only be used in medical institutions that meet certain standards, as it requires collaboration with radiologists, neurologists, or other specialists to assess amyloid-related imaging abnormalities (ARIA) such as cerebral vascular lesions and cerebral hemorrhage, as well as facilities capable of administering Leqembi intravenously every 2 weeks and personnel to monitor adverse drug reactions. Nevertheless, Lecanemab is being rapidly adopted and utilized on-site not only at university hospitals but also at regional hub general hospitals, due to the disease's characteristic lack of a fundamental cure. According to an internal evaluation by the Korean Dementia Association, approximately 700 cases have been treated using the medication 6 months after its introduction in Korea. Seong Hye Choi, president of the Korean Dementia Association (Department of Neurology, Inha University Hospital), stated, “A patient weighing 50kg bears approximately KRW 1 million for a single administration of Leqembi. Considering that it is administered every 2 weeks, this amounts to KRW 2 million per month, and KRW 24 million per year for the patient. For a 40kg patient, the cost per administration is approximately KRW 800,000. Some patients are covered with indemnity insurance, and the treatment being well received onsite, even better than expectations.” Additionally, in clinical settings, there is a projection that the incidence of adverse reactions related to brain edema, particularly ARIA, observed during MRI scans in the course of Leqembi’s introduction may be relatively lower than what was shown in clinical studies. Kee Hyung Park, Chair of the Strategy and Planning Committee (Department of Neurology, Gachon University Gil Medical Center), said, “Japan introduced Leqembi before South Korea, and the drug is being actively used as it is covered by reimbursement. Among approximately 8,000 cases, ARIA occurred in 537 cases, which is 6.7% of all patients.” He added, “During the initial clinical trials, the incidence rate of ARIA was reported to be between 12% and 17%.” Park added, “In Korea, there have been no cases of patients experiencing severe side effects after receiving Leqembi. While it is still too early to draw definitive conclusions, it is reasonable to expect that side effects may be less common in Asians compared to Westerners. Amidst this, doctors have suggested that reimbursement for Leqembi itself is necessary, but that reimbursement for tests such as MRI is more urgent. Currently, MRI tests are required to assess cerebral vascular lesions and ARIA in patients receiving Leqembi. The logic is that tests that bring less health insurance burden should be reimbursed first, followed by a discussion on reimbursement for the drug itself. Choi said, “According to MFDS guidelines, patients receiving Leqembi must undergo MRI scans. This means that MRI scans must be performed before the fifth, seventh, and fourteenth injections. If reimbursement is applied to this part of the treatment, it will be able to somewhat reduce the burden on patients.” Choi also pointed out that long-term support for the costs of injection rooms and dedicated nurses (coordinators) provided for severe cancer patients is also necessary for dementia. Recently, Ewha Womans University Mokdong Hospital and Ewha Womans University Seoul Hospital introduced dedicated nurses following the adoption of Leqembi, but many medical institutions find it difficult to operate such programs due to the burden of labor costs. Jee Hyang Jeong of the Department of Neurology at Ewha Womans University Seoul Hospital (Chair, Public Relations Committee, KDA) explained, “When patients and their families request Leqembi treatment, it is necessary to provide detailed explanations about the treatment process, side effects, MRI scans, and other related matters. This entire process typically takes 30 to 40 minutes. We recognize this as a critically important process and have begun operating with dedicated staff internally. This was a big and dedicated decision on the hospital’s part.” Jeong added, “Ultimately, dementia is similar to cancer. We need to hire dementia specialists to educate patients and their families. The same applies to infusion rooms. Comprehensive improvements are needed, including reimbursement for the use of infusion rooms and labor costs, similar to cancer treatment.” Following the use of Leqembi on-site, expectations are also rising for another new dementia drug, Lilly's Kisunla (donanemab). Lilly's Kisunla cleared the regulatory hurdle - FDA - last year. Kisunla demonstrated efficacy in delaying cognitive decline in patients with early-stage Alzheimer's disease in the Phase III TRAILBLAZER-ALZ2 study. In the clinical trial, it delayed cognitive decline regardless of the disease's progression or pathological stage. Particularly, compared to Leqembi, it is evaluated as having a high potential for utilization in Korea due to improved convenience in patient administration. Choi said, “Kisunla has the advantage of a relatively long administration cycle and the possibility of discontinuing treatment midway through the course. I understand that Lilly, the manufacturer of Kisunla, is in discussions with the MFDS for its introduction in Korea. With mixed results in global markets, I expect the MFDS to make a decision based on the domestic situation.” Park also noted, “Rather than focusing on why approval was granted in the U.S. but not in Europe, we should examine the characteristics of the drug and its differences from existing medications. We need to assess this and consider how patients can individually select the appropriate medication for treatment when approval is granted domestically.”
- Company
- Vyloy quickly lands in general hospitals in Korea
- by Eo, Yun-Ho May 13, 2025 06:07am
- Vyloy, a targeted anticancer drug for gastric cancer, may be prescribed at general hospitals in Korea. According to industry sources, Vyloy (zolbetuximab), a Claudin 18.2-positive gastric cancer targeted therapy developed by Astellas Korea, is now available for prescription at the Big 5 tertiary hospitals in Korea, including Samsung Medical Center, Seoul National University Hospital, Seoul St. Mary's Hospital, Asan Medcial Center, and Sinchon Severance Hospital. The drug has passed the regular or emergency drug committees (DCs) at each hospital. Additionally, prescription codes have been generated at other medical institutions such as Ajou University Hospital, Chilgok Kyungpook National University Hospital, and Hwasun Chonnam National University Hospital. Approved in Korea last September, Vyloy is the world's first claudin 18.2-targeted therapy, an immunoglobulin (IgG) monoclonal antibody that binds to Claudin 18.2, a protein expressed on the surface of cancer cells in gastric epithelial cells. The SPOTLIGHT study, which became the basis of Vyloy’s approval, showed that Vyloy+ mFOLFOX6 (fluorouracil, leucovorin, oxaliplatin) combination therapy’s progression-free survival (PFS) was 10.6 months, compared with the 8.67 months in the placebo arm. Also, the median overall survival (OS) was 18.23 months, compared with the 15.54 months in the placebo arm. Additionally, in the GLOW study, the combination therapy group of Vyloy and CAPOX (capecitabine and oxaliplatin) achieved a median progression-free survival (mPFS) of 8.21 months, reducing the risk of disease progression or death by approximately 31%. However, Vyloy has not yet been reimbursed by health insurance. The drug was submitted to the Health Insurance Review and Assessment Service's Cancer Disease Review Committee in February, but failed to set the reimbursement criteria. Additionally, due to companion diagnostic issues last year, Vyloy was only officially launched in Korea in March. To use Vyloy, patients must be diagnosed as Claudin 18.2 positive, and the companion diagnostic (CDx) device used for Claudin 18.2 diagnosis was considered for evaluation as a new health technology in the reimbursement review process. In response, Astellas has been conducting an EAP program even before Vyloy’s approval to allow sooner access to the treatment, and currently, 51 patients are enrolled across 10 institutions. SunYoung Rha, a professor of medical oncology at Yonsei Cancer Center, stated, “Approximately 90% of metastatic gastric cancer patients are HER2-negative, rendering a dire need for treatment options targeting this biomarker. Given that about 40% of HER2-negative patients are reported to be Claudin 18.2-positive, the release of Vyloy, which selectively binds to Claudin 18.2, presents a new treatment possibility.”
- Company
- Mounjaro provides more weight loss than Wegovy
- by Eo, Yun-Ho May 13, 2025 06:06am
- Research results found Mounjaro brings more weight loss than its competitor Wegovy. On the 12th, Lilly announced the detailed results of the SURMOUNT-5 Phase IIIb open-label clinical trial directly comparing Mounjaro (tirzepatide), a dual GIP/GLP-1 receptor agonist, and Wegovy (semaglutide), a GLP-1 receptor agonist. The study, which compared the weight loss effects of the two drugs that have garnered global attention as obesity treatments, was simultaneously presented at the 32nd European Congress on Obesity (ECO) and published in the international academic journal The New England Journal of Medicine (NEJM). The study enrolled adult patients with a body mass index (BMI) of 30 kg/m² or higher, or overweight patients (BMI 27 kg/m² or higher but less than 30 kg/m²) with at least one weight-related comorbidity, excluding diabetes. In the trial, Mounjaro demonstrated superiority over Wegovy, meeting all the primary endpoints and five key secondary endpoints at week 72. The primary endpoint, the mean percent change in weight from baseline to week 72 in the Mounjaro group (10 mg or 15 mg), was 20.2%, compared to 13.7% in the Wegovy group (1.7 mg or 2.4 mg), representing a 47% relative weight loss improvement. Based on treatment regimen estimands, the mean weight loss was 22.8 kg in the Mounjaro group and 15.0 kg in the semaglutide group. Mounjaro also outperformed Wegovy in all key secondary endpoints of weight loss. In key secondary endpoints, Mounjaro was superior across all weight reduction targets, with 64.6% of patients in the Mounjaro group achieving at least 15.0% weight loss compared to 40.1% in the Wegovy group. Additionally, patients in the Mounjaro group achieved a superior average waist circumference reduction of 7.2 in (18.4 cm), while in the Wegovy group saw an average reduction of 5.1 in (13.0 cm). Min-Seon Kim, President of the Korean Society for the Study of Obesity and Professor of Endocrinology at Asan Medical Center, stated, “In the head-to-head trial (SURMOUNT-5), Mounjaro demonstrated a superior weight loss effect compared to Wegovy. I expect the introduction of this medication to contribute to obesity treatment in Korea.”
- Company
- U.S. pharmaceutical imports rise twofold amid tariff concern
- by Kim, Jin-Gu May 13, 2025 06:05am
- U.S. drug imports have nearly doubled in a year. This is believed to be the result of frontline pharmaceutical and biotech companies focusing on securing inventory in expectation of U.S. President Donald Trump's drug tariff imposition. According to Korea Biotechnology Industry Organization (KoreaBIO) on the 12th, the U.S. The United States Census Bureau and the U.S. Bureau of Economic Analysis recently announced the results of U.S. trade in goods and services. According to the data, U.S. imports of goods in the first quarter totaled USD 997.8 billion. This is a 26.6% (USD 209.8 billion) increase from the USD 788 billion in the first quarter of last year. In particular, imports of pharmaceuticals increased significantly. In the first quarter, U.S. imports of pharmaceuticals amounted to USD 108.2 billion, a 97.2% (or USD 53.3 billion) increase from the USD 54.9 billion in the same period last year. Pharmaceutical imports surged in March during the first quarter. This coincides with the spread of concerns over President Trump's pharmaceutical tariffs. In fact, U.S. pharmaceutical imports, which stood at USD 29.5 billion in February, rose to USD 50.4 billion in March, an increase of 70.9% in just one month. Considering that total imports increased by 5.5% (from USD 326.5 billion to USD 344.3 billion) during the same period, the increase in pharmaceutical imports was particularly notable. The share of pharmaceuticals in total U.S. imports rose from 9.0% in February to 14.6% in March, an increase of 5.6 percentage points in a single month. KoreaBIO explained that the increase in U.S. pharmaceutical imports was due to the proactive response of local pharmaceutical and biotech companies to concerns over President Trump's import tariffs. KoreaBIO analyzed that “local pharmaceutical and biotech companies appear to be focusing more on securing inventory in the U.S. this year as a short-term response.” Domestic companies are also focusing on securing inventory in the U.S. As of the 7th of this month, Celltrion has completed the transfer of approximately 15 months' worth of inventory for products scheduled for sale in the U.S. Celltrion expects that this will minimize the impact of tariffs not only this year but also in the first half of next year. Celltrion plans to minimize risk in the mid- to long-term by producing finished drug products (DP) through local contract manufacturing organizations (CMOs) in the US. The company explained that it has completed negotiations to secure additional quantities in preparation for the possibility of increased tariff burdens after next year. At the same time, it is pushing ahead with securing active pharmaceutical ingredient (API) manufacturing facilities in the US. It has completed preliminary reviews and has entered the stage of detailed reviews. A Celltrion official said, “We have been devising various strategies in advance to respond to the possibility of changes in President Trump's tariff policy,” adding, “Once the policy is finalized, we will promptly share a comprehensive response plan with our shareholders.” The U.S. government plans to make a separate announcement on whether to impose tariffs on pharmaceuticals. Although it was not included in the mutual tariffs announced on the 2nd of last month, expectations are growing that separate tariffs on specific items will be imposed in May. Prior to this, the U.S. Department of Commerce has been assessing the impact of imports of pharmaceuticals and pharmaceutical raw materials on national security under Section 232 of the Trade Expansion Act, and requested public comments from interested parties from the 16th of last month to the 7th of this month. It is reported that a total of 957 companies and institutions worldwide, including the United States, submitted public comments. South Korea also submitted comments through the government, KoreaBIO, and the Korea International Trade Association(KITA). U.S. companies and institutions, including the Pharmaceutical Research and Manufacturers' Association, are said to have submitted comments stating that “imposing tariffs on pharmaceuticals is not the answer.”
- Company
- 'Retevmo' also seeks thyroid cancer reimbursement
- by Eo, Yun-Ho May 12, 2025 05:59am
- Product photo of Retevmo The RET-targeted cancer therapy 'Retevmo' seeks insurance reimbursement listing of its medullary thyroid cancer indication. According to sources, Lilly Korea recently submitted reimbursement applications simultaneously for the non–small cell lung cancer indication and medullary thyroid cancer indication of the RET (REarranged during Transfection) inhibitor Retevmo (selpercatinib). Having failed in its previous two attempts to secure reimbursement listing for lung cancer, the company is now showing its determination to gain listing by adding a new indication. The prevalence of medullary thyroid cancer is only 1–2% of all thyroid cancers worldwide and just 0.6% in South Korea. However, 60% of cases harbor RET gene mutations. In other words, RET-targeted therapies can be used to treat medullary thyroid cancer. First-line treatments for medullary thyroid cancer currently consist of multitargeted kinase inhibitors such as vandetanib and cabozantinib, which inhibit multiple kinases non-selectively and can therefore be associated with higher rates of adverse events. In contrast, Retevmo is a highly selective RET inhibitor. Retevmo demonstrated efficacy in medullary thyroid cancer in the Phase 3 LIBRETTO-531 study. The LIBRETTO-531 study is a Phase 3 open-label trial comparing Retevmo against the approved first-line multikinase inhibitors (cabozantinib or vandetanib) in patients with progressive or metastatic RET-mutant medullary thyroid cancer. A total of 291 medullary thyroid cancer patients with no prior multikinase inhibitor therapy were randomized (193 Retevmo treatment group, 98 control group). At the investigators' discretion, patients in the control group received cabozantinib in 73 cases and vandetanib in 25 cases. The progression-free survival (PFS) comparison yielded positive data in the interim analysis. At a median follow-up of approximately 12 months, the Retevmo treatment group had not yet reached median PFS by blinded independent central review (BICR), while the control group had a median PFS of 16.8 months. The hazard ratio was 0.280. In preplanned subgroup analyses, BICR-assessed and investigator-assessed PFS were longer with Retevmo than with the control. Meanwhile, although Retevmo was approved in South Korea in March 2022, it failed to pass the National Health Insurance Service (NHIS) Cancer Disease Review Committee (CDRC) in May of the same year. Still, it cleared the CDRC in November and passed the Drug Reimbursement Evaluation Committee (DREC) in May 2023. After passing the reimbursement committee, negotiations with the NHIS on pricing began in June, raising hopes for listing, but ultimately, no agreement was reached. The case was the only failed price negotiation of that year. A Lilly representative said, "The company has been continuously preparing for the reapplication process and has submitted all required documents to the NHIS. The government is currently reviewing the application. We will do our best to ensure that more patients can benefit as soon as possible."