- LOGIN
- MemberShip
- 2026-05-01 04:26:41
- Company
- 'Wegovy' dominating the South Korean obesity drug market
- by Chon, Seung-Hyun May 27, 2025 06:17am
- Novo Nordisk's Wegovy has dominated the South Korean obesity treatment market, establishing a monopolistic competition with over 70% market share. In just six months since its launch in Korea, Wegovy generated a sensation, surpassing KRW 100 billion in cumulative sales. Wegovy's success has expanded the obesity drug market to its largest size ever, while sales of Saxenda, which previously led the market, sharply declined. According to pharmaceutical market research firm IQVIA, on May 26, the obesity drug market reached KRW 108.6 billion in the first quarter of this year, a 162.3% increase compared to KRW 41.4 billion in the same period last year. This is the first time the quarterly obesity drug market size has exceeded KRW 100 billion. Quarterly Obesity Treatment Market Size and Key Product Sales. Legend: Bars-obesity market, Black-Wegovy, Purple-Qsymia, Green-Saxenda (Unit: KRW 100 million, Source: IQVIA) Novo Nordisk's Wegovy clearly stood out. In the first quarter, Wegovy's sales recorded KRW 79.4 billion, accounting for a dominant 73.2% of the entire obesity drug market. Wegovy, which was approved by the Ministry of Food and Drug Administration (MFDS) in April 2023, is a GLP-1 analogue containing semaglutide, known to reduce HbA1c. Novo Nordisk developed Wegovy as a once-weekly obesity treatment using semaglutide after observing weight loss effects in patients during clinical trials of GLP-1 class diabetes drug candidates. Wegovy gained immense popularity immediately after its domestic launch in October last year. Despite its high price, prescription demand surged due to its significant weight loss efficacy. Wegovy's sales reached KRW 60.3 billion in the fourth quarter of last year, propelling it to the top of the obesity drug market. The obesity drug market size was KRW 47.4 billion in the third quarter of last year, but it soared by 97.9% to KRW 93.8 billion in just one quarter following Wegovy's launch. Wegovy held a 63.4% market share in the obesity drug market in the fourth quarter of last year, and its share has continued to rise this year. Under six months since its domestic release, Wegovy's cumulative sales reached KRW 139.8 billion, exceeding KRW 100 billion. Wegovy is thriving globally due to its groundbreaking weight loss effects. Wegovy's sales last year reached DKK 58.26 billion (approximately KRW 11.7 trillion), an 85.7% increase from DKK 31.343 billion in 2023. Demand for Wegovy surged to the point of shortages after its launch in the U.S. market. Even before its domestic launch, Wegovy gained global fame for shortages, having been rumored as the weight loss secret of international celebrities like Tesla CEO Elon Musk. Despite its high price of around KRW 500,000, Wegovy gained explosive interest immediately after its domestic release, leading to supply shortages. Even with the suspension of non-face-to-face prescriptions, demand for Wegovy continued to rise. Initially, Wegovy was actively prescribed through non-face-to-face medical consultations. When concerns were raised that Wegovy was being indiscriminately prescribed via non-face-to-face consultations regardless of a person's weight or obesity status, health authorities suspended non-face-to-face prescriptions for obesity treatments from December 16 of last year. The introduction of Wegovy has led to a reduction in sales of Saxenda and Qsymia, which previously dominated the obesity drug market. The presence of Novo Nordisk's Saxenda has significantly declined in the obesity drug market. Saxenda's first-quarter sales were KRW 4.2 billion, down 72.3% from KRW 15.1 billion in the same period last year. Compared to its KRW 24.2 billion sales in the second quarter of last year, its sales have sharply declined to less than 20%. Saxenda, launched in Korea in 2018, was the world's first obesity drug approved as a GLP-1 analog. Saxenda's active ingredient, liraglutide, is identical to Victoza's, prescribed for type 2 diabetes patients, differing only in dosage and administration. The analysis suggests that Wegovy, a GLP-1 class drug similar to Saxenda, has further taken Saxenda's market. With reduced domestic supply since Wegovy's launch, there are rumors of Saxenda's production being discontinued. After becoming the obesity treatment market leader with sales of KRW 42.6 billion immediately after its launch in 2019, Saxenda maintained its lead for five consecutive years until 2023. Saxenda's sales reached KRW 66.8 billion in 2023, accounting for 37.5% of the obesity treatment market that year. However, following the introduction of Wegovy, Saxenda's sales sharply declined, and its market share in the first quarter of this year was only 3.8%. The first-quarter sales of Alvogen Korea's Qsymia decreased by 3.9% year-on-year to KRW 8.6 billion. Launched in late 2019, Qsymia is a combination drug containing 'phentermine' and 'topiramate'. Alvogen Korea secured domestic marketing rights from U.S.-based Vivus in 2017. Alvogen Korea entered a co-promotion agreement with Chong Kun Dang in late 2019 and began full-scale sales in Korea. Qsymia recorded KRW 10.2 billion in sales in the third quarter of last year but fell to 9.3 billion KRW in the fourth quarter, when Wegovy was launched, and has further decreased this year.
- Company
- Re-evaluation possibility of 'Bylvay' gathers attention
- by Eo, Yun-Ho May 26, 2025 05:57am
- Product photo of Ipsen KoreaAttention has been drawn to when 'Bylvay Cap,' the first medicine chosen for the 'Pilot Project for Integration of Product Approvals, Reimbursement Coverage Reviews, and Drug Price Negotiations,' will be re-evaluated. Bylvay (odevixibat), Ipsen Korea's treatment option for progressive familial intrahepatic cholestasis (PFIC) in patients aged 3 months or older, received a decision of reevaluation at the Health Insurance Review and Assessment Service (HIRA)'s Drug Reimbursement Evaluation Committee (DREC) meeting held in April. After that, the drug had not been considered for the DREC review in May so it will likely be considered for the upcoming review. As part of implementing the first concurrent approval-evaluation-negotiation pilot project since October, the government selected two medicines, including 'Qarziba (dinutuximab)' for treating rare disease in children and Bylvay, as the first drugs for the pilot project. The concurrent approval-evaluation-negotiation pilot project conducts the Ministry of Food and Drug Safety (MFDS)'s approval, HIRA's drug evaluation, and drug pricing simultaneously, expediting the insurance listing process, including approval, drug evaluation, and the Ministry of Health and Welfare (MOHW) reporting. However, Bylvay not being approved for reimbursement has raised questions about the validity of the expedited listing of selected drugs. Furthermore, Bylvay was challenged at the stage of setting the reimbursement criteria. It has been reported that the company poorly took the expert advice while setting the reimbursement criteria because the expert advice gathering process was reduced to merely a 'formality.' In response, the HIRA stated, "To facilitate DREC's efficient evaluation, HIRA operates a small committee for setting reimbursement criteria before a review. At the small committee meeting, HIRA was reviewed in depth by gathering advice from experts and academics regarding clinical usefulness and cost-effectiveness. It was not indeed a thorough process rather than merely a formality." It is to be watched whether Bylvay, which was selected for the concurrent approval-reimbrusement-drug price pilot project, is to be included in the reimbursement list. Meanwhile, the efficacy of Bylvay was demonstrated through the Phase 3 ASSERT study involving children and adolescent patients aged 17 years and below. The study results demonstrated that Bylvay reduced itchiness statistically significantly compared to a placebo, meeting the primary endpoint. Furthermore, Bylvay statistically improved the average serum bile acid concentration at the primary endpoint target time-points, at week 20 and week 24, than the placebo. This effect of Bylvay continued through 24 weeks of treatment.
- Company
- Pfizer’s Lorviqua is granted reimbursement in Korea
- by Whang, byung-woo May 26, 2025 05:54am
- Lorviqua (lorlatinib), a treatment for ALK-mutated non-small cell lung cancer (NSCLC), has been approved for reimbursement as a first-line treatment, heralding a tectonic shift in the field. Experts saw this as a positive development in addressing unmet patient needs and improving access to treatment. In this sense, the presence of third-generation treatments for ALK-positive metastatic non-small cell lung cancer is expected to grow further. On the 21st, Pfizer Korea held a press conference to commemorate the expanded reimbursement of Lorviqua (lorlatinib), a first-line treatment for ALK-positive metastatic non-small cell lung cancer (NSCLC), highlighting the significance of the milestone. Ji-Youn Han, Professor of Hematology and Oncology at the National Cancer Center Lorviqua is a third-generation ALK tyrosine kinase inhibitor (TKI) designed to be effective against ALK mutations and to effectively penetrate the blood-brain barrier (BBB). In May 2022, it was granted reimbursement as a first-line treatment, 3 years after the indication was expanded to include ALK-positive metastatic non-small cell lung cancer as a first-line treatment. Patients with ALK-positive non-small cell lung cancer account for over 80% of all lung cancer cases and are characterized by relatively young age and a history of minimal or no smoking. Lorviqua’s efficacy as a first-line treatment was confirmed in the global Phase III CROWN trial. According to the 5-year follow-up results of the global Phase III CROWN clinical trial, Lorviqua demonstrated an 81% reduction in the risk of disease progression or death compared to the crizotinib group in patients with no prior treatment experience. Also, the median progression-free survival (PFS) for Lorviqua was not reached at 60.2 months of follow-up, regardless of brain metastasis status, while the median PFS for crizotinib was 9.1 months at 55.1 months of follow-up. According to a 5-year analysis of the CROWN trial, this is the longest progression-free survival rate achieved among ALK-positive non-small cell lung cancer treatments to date. Ji-Youn Han, Professor of Hematology and Oncology at the National Cancer Center who presented at the event, said, “Among non-small cell lung cancer, which accounts for the majority of lung cancer cases, treatment for ALK-positive non-small cell lung cancer, a major genetic mutation, has progressed from the first-generation crizotinib to the third-generation Lorviqua. According to the five-year follow-up analysis of the CROWN study, this is the treatment with the longest median progression-free survival (mPFS) among ALK-positive NSCLC treatments to date.” Han added, “There are studies showing that 25-30% of patients with ALK-positive metastatic non-small cell lung cancer do not receive later-line treatment and that the main cause is rapid clinical deterioration due to tumor progression. The reimbursement of Lorviqua as a first-line treatment is significant in that it addresses unmet needs and greatly improves access to treatment.” Furthermore, the efficacy and safety profile of Lorviqua was consistently demonstrated in studies involving patients in Asia, including Korea. In a study of Asian patients, after 5 years of follow-up, the median PFS for Lorviqua was not reached, while the median PFS for crizotinib was 9.2 months. Furthermore, Lorviqua demonstrated a 99% reduction in the risk of disease progression and death compared to crizotinib at the 5-year mark. However, some have expressed concerns about the risk of central nervous system side effects associated with Lorviqua. One percent of patients who experienced cognitive impairment as an adverse reaction after receiving Lorviqua discontinued treatment. Nevertheless, given that Lorviqua demonstrates superior efficacy compared to existing treatments, Han believes it can become the preemptive choice as a first-line treatment. He stated, “Based on the data, Lorviqua shows positive results in patients with over 5 years of follow-up, so there seems to be no room for second-generation treatments. While there may be cases where the drug is replaced due to CNS side effects, using Lorviqua, which maintains PFS for over 5 years, as a backup and using second-generation treatment first is something patients would not agree to.” In fact, Lorviqua was included in the 2025 National Comprehensive Cancer Network (NCCN) guidelines (recommendation level: Category 1), the 2024 American Society of Clinical Oncology (ASCO, recommendation level: Strong), and the 2023 European Society for Medical Oncology (ESMO, recommendation level: Tier I-A) guidelines as one of the first-line treatments for ALK-positive metastatic non-small cell lung cancer.
- Company
- Accomplishments of the approval-drug price pilot project
- by Eo, Yun-Ho May 23, 2025 05:52am
- Winrevair·Fintepla·Limcato expected to be commercialized With the drugs that were reviewed for approval and drug prices simultaneously about to be commercialized, the industry gathers attention. The Ministry of Health and Welfare (MOHW) has been running the 'Pilot Project for Integration of Product Approvals, Reimbursement Coverage Reviews, and Drug Price Negotiations' since 2023 to improve treatment access for life-threatening severe·rare diseases. The project conducts approval, reimbursement evaluation, and drug price negotiations simultaneously, aiming to shorten the time required for new drugs to be included in the National Health Insurance list. The first pilot project is at the final stage, showing accomplishments. Three drugs were selected in the second pilot project, which recently completed the selection of items. Typically, it takes approximately 330 days for a new drug to be introduced into the market. This pilot program aims to significantly reduce the time for ▲Product Approval (Ministry of Food and Drug Safety, MFDS) for 120 days ▲DrugReimbursement Evaluation (Health Insurance Review & Assessment Service, HIRA) for 150 days ▲ Drug Price Negotiation (National Health Insurance Service, NHIS) for 60 days. The two drugs selected for the first pilot program (2023) are either already on the reimbursement list or nearing their evaluation process's completion. Following this, ten drugs were submitted for the second pilot program, launched in 2024, and three new drugs were selected. These three are 'Winrevair (sotatercept),' a pulmonary hypertension treatment from MSD Korea; 'Fintepla (fenfluramine),' a Dravet syndrome treatment from UCB Pharma Korea; and 'Limcato,' a large B-cell lymphoma treatment from the Korea-based company Curocell. All three drugs are currently expected to be commercialized in Korea this year. Among them, Winrevair is receiving particular attention. This drug is a first-in-class innovative new drug with a novel mechanism, marking its appearance 20 years after 'Sildenafil', which targeted the NO-sGC-cGMP pathway, in 2005. As of 2023, the number of pulmonary hypertension patients in Korea is approximately 3,600. The average age of these patients is in their 40s, a demographic that plays a crucial role in society and family. Although the 5-year survival rate has significantly improved compared to the past, 3 out of 10 Korean pulmonary hypertension patients still die within 5 years. Furthermore, most patients experience significant difficulties performing daily activities such as housework, childcare, and light outings. Pulmonary hypertension is a rare, intractable, and progressive disease, where delaying the worsening of the condition directly impacts patients' quality of life and survival. To date, no cure through drug treatment has been discovered, and the mechanism of existing drugs primarily aims to alleviate symptoms by relaxing thickened pulmonary arteries. It remains to be seen how quickly drugs undergoing the concurrent approval-evaluation process, including Winrevair, will be listed. The selection criteria for the second pilot program considered drugs that met all of the following conditions: ▲ drugs for which approval and reimbursement decisions can be applied by June 2025 ▲ drugs with sufficient efficacy intended for treating life-threatening diseases with a life expectancy of less than one year, or for rare diseases ▲drugs for which no existing treatment is available or that show clinically significant improvement over current treatments ▲drugs that have been designated for or are eligible to apply for, MFDS's Global Innovative Products on Fast Track (GIFT) program. The MOHW announced December 2024 that it had selected three drugs for the second pilot program. This selection considered factors such as disease severity, availability of alternative drugs, urgency, treatment efficacy, and expert opinions from the submitted applications.
- Company
- CKD-Novartis new drug advances to the next phase trial
- by Chon, Seung-Hyun May 23, 2025 05:52am
- Chong Kun Dang (CKD)'s new drug candidate out-licensed to Novartis is entering the next clinical stage. CKD has secured its first milestone payment of KRW 7 billion, 1 year and 6 months after the technology export contract. CKD announced on May 22 that it expects to receive a milestone payment of USD 5 million (KRW 7 billion) from Novartis, upon the achievement of a stage-based milestone for CKD-510. As Novartis submitted its Investigational New Drug (IND) application for a Phase 2 clinical trial of CKD-510 to the U.S. Food and Drug Administration (FDA), the condition for the milestone payment, as per the contract, was met. CKD is set to receive an additional milestone payment for the first time since the CKD-510 technology export. CKD-510 is a new drug candidate that CKD out-licensed to Novartis in November 2023. It was a blockbuster technology export agreement with a non-refundable upfront payment of USD 80 million. The maximum milestone payment, contingent on development and approval stages, reaches up to USD 1.225 billion. CKD-510 is a new drug candidate researched and developed by CKD. It is an HDAC6 inhibitor developed using a highly selective non-hydroxamic acid (NHA) platform technology. In preclinical studies, the efficacy of the candidate was confirmed in various HDAC6-related diseases, including cardiovascular diseases. Its safety and tolerability were demonstrated in Phase 1 clinical trials conducted in Europe and the United States. CKD has completed the European Phase 1 trial of CKD-510 for Charcot-Marie-Tooth (CMT) disease. CMT is a hereditary peripheral neuropathy where motor and sensory nerves are damaged due to gene mutations, making normal walking and daily life difficult. It is a rare disease, but there is currently no definitive treatment. CKD has strategized to derive optimal drugs for various diseases based on the fundamental structure of HDAC6 inhibitors. CKD is developing new drugs applying its HDAC6 platform technology to CMT disease, Huntington's disease, Alzheimer's disease, hematologic cancers, and autoimmune diseases. Novartis enters a Phase 2 clinical trial for the first time since in-licensing CKD-510. However, Novartis has not disclosed the target indication for CKD-510.
- Company
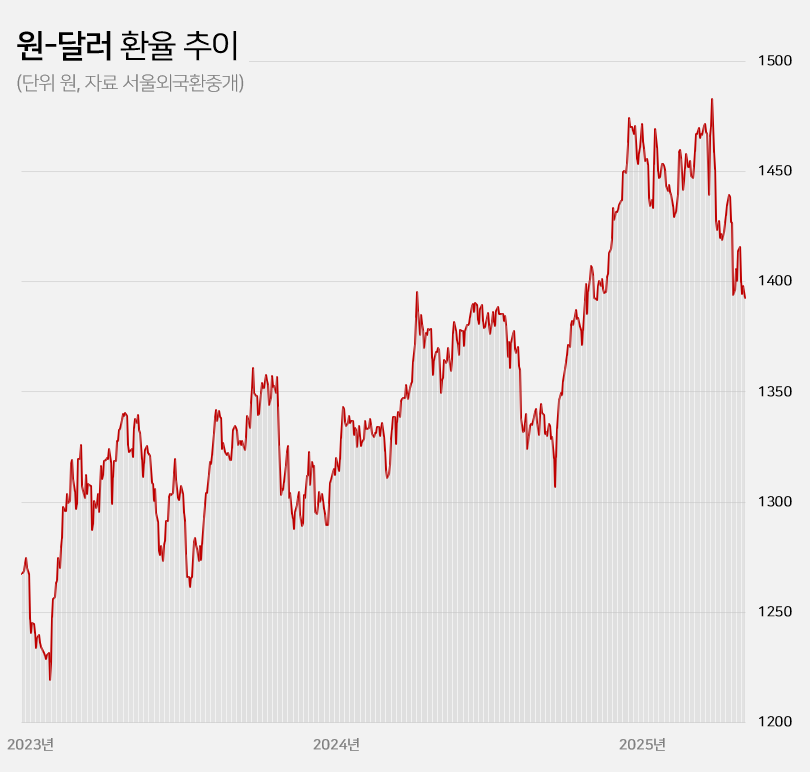
- Won-dollar rate lowest in 6mths... industry mixed
- by Kim, Jin-Gu May 23, 2025 05:51am
- With the won-dollar exchange rate falling below KRW 1,400, pharmaceutical and biotech companies are experiencing a mix of anticipation and concern. If the prolonged high exchange rate returns to previous levels, API imports and overseas clinical trial costs are expected to decrease, leading to an improvement in the cost structure. On the other hand, some predict that the asset value of pharmaceutical and biotech companies with a high proportion of drug exports will decline. Won-dollar exchange rate falls below KRW 1,400... Will the burden of API purchases and overseas clinical trials decrease? According to industry sources on the 22nd, the won-dollar exchange rate closed at KRW 1,392.60 on the 21st, down KRW 5.40 from the previous trading day. This is a 6.1% (KRW 90.30) decrease from the KRW 1,482.90 on the 10th of last month when concerns over the U.S.’s mutual tariffs were at their peak. The won-dollar exchange rate has remained high for a long time since entering the KRW 1,300 range in March 2023. It rose further to over KRW 1,350 from the middle of last year and broke through KRW 1,400 after the martial law crisis at the end of last year. With mutual tariffs by the US adding to the concern, the exchange rate soared to exceed KRW 1,480 at the beginning of last month. However, the won-dollar exchange rate has been falling this month amid expectations that trade tensions between the US and China will ease. In particular, the exchange rate has remained in the KRW 1,300 range for 4 consecutive trading days since the 16th, raising expectations that the prolonged high exchange rate trend will return to normal levels. KRW to USD exchange rate The pharmaceutical industry is hopeful that the decline in the won-dollar exchange rate will continue, leading to an improvement in the cost structure. Over the past 2 years, the rise in the won-dollar exchange rate has had a negative impact on the cost structure of domestic pharmaceutical and biotechnology companies. Due to their high dependence on imported APIs, the exchange rate increase directly led to higher manufacturing costs. Additionally, since these companies purchase APIs from China and India, with a high import dependency, they were significantly affected by the rise in the won-dollar exchange rate. As of 2023, the self-sufficiency rate for domestically produced APIs for pharmaceuticals stands at 25.4%. Among these, imports of APIs from China account for 30.5% of the total, while those from India make up 15.2%. Together, these two countries account for nearly 50% of the total API imports. Although manufacturing costs rose due to the increase in the won-dollar exchange rate, unlike other consumer goods, the price of finished drugs cannot be arbitrarily set by companies. As a result, the deterioration of the cost structure in the pharmaceutical industry has become more pronounced over the past 2 years. Additionally, the burden of clinical trial costs conducted overseas has steadily increased during the prolonged high exchange rate environment. Most clinical trials targeting the US and European markets are conducted locally. When the won-dollar exchange rate rises, global clinical trial costs also increase accordingly. In this situation, there are expectations that a decline in the won-dollar exchange rate will reduce the cost of importing APIs and the burden of global clinical trials, ultimately improving cost burdens. In the long term, improvements in the cost structure are expected to contribute positively to performance recovery. Asset value decline is inevitable for companies with high export ratios... Samsung Biologics to lose KRW 91.6 billion if exchange rate falls by 10% However, companies with high export ratios, such as Samsung Biologics and Celltrion, are expected to see their asset values decline due to the decline in the won-dollar exchange rate. These companies earn a large amount of assets in dollars overseas, so when the exchange rate rises, their asset values increase. Conversely, when the exchange rate falls, their asset values decline. Samsung Biologics had an overseas sales ratio of 96.5% in the first quarter. Of the KRW 1.2983 trillion in sales rendered in the first quarter, KRW 1.2528 trillion came from overseas. As the proportion of overseas sales is high, its performance is expected to be greatly affected by fluctuations in the exchange rate. Samsung Biologics explained in its quarterly report that a 10% increase or decrease in the won-dollar exchange rate would result in a KRW 91.6 billion increase or decrease in pre-tax income. Considering how the current won-dollar exchange rate has fallen by 1.5% compared to the average exchange rate (KRW 1,452.66) at the time of the quarterly report, the recent decline in the exchange rate is estimated to have reduced asset value by approximately KRW 14 billion. Samsung Biologics has seen an increase in cash and cash equivalents over the past 2 years due to the impact of high exchange rates. The figures were KRW 12.2 billion in 2023 and KRW 41.6 billion last year. This was due to the average won-dollar exchange rate rising from KRW 1,291.95 in 2022 to KRW 1,307.90 in 2023 and KRW 1,363.09 in 2024. The same goes for Celltrion and SK Biopharm. Given their high proportion of overseas sales, they are relatively more affected by fluctuations in the exchange rate. Celltrion saw its cash and cash equivalents increase by KRW 52.4 billion last year due to exchange rate changes. The company explains that, assuming all other variables remain constant, an 8% change in the won-dollar exchange rate would result in a KRW 33.8 billion increase or decrease in pre-tax profit. For SK Biopharm, the change in cash and cash equivalents due to exchange rate fluctuations last year amounted to KRW 3.6 billion. Based on the exchange rate at the end of last year, a 10% change in the exchange rate would result in an increase or decrease of KRW 14.1 billion in the company's pre-tax profit. It is expected that the fluctuation in pre-tax profit due to exchange rate changes will widen further this year if sales of Xcopri increase in the United States.
- Company
- Will Tibsovo be reimbursed for cholangiocarcinoma this time?
- by Eo, Yun-Ho May 22, 2025 06:10am
- Attention is focused on whether the targeted anticancer drug “Tibsovo” for cholangiocarcinoma and acute myeloid leukemia will be successful in its attempt to be reimbursed by health insurance in Korea. Servier Korea’s IDH1 (isocitrate dehydrogenase 1) genetic mutation targeting therapy recently passed the Health Insurance Review and Assessment Service's Cancer Disease Review Committee. The drug is indicated for use in IDH1-mutation-positive patients in combination with azacitidine for newly-diagnosed acute myeloid leukemia (AML) with a susceptible isocitrate dehydrogenase-1 (IDH1) mutation in adults aged ≥75 years, or who have comorbidities that preclude use of intensive induction chemotherapy, or as monotherapy for locally advanced or metastatic cholangiocarcinoma in previously treated adults with IDH1 mutation. The dual AML indication passed the Health Insurance Review and Assessment Service's Cancer Disease Review Committee’s review in October last year, but the cholangiocarcinoma indication had not been approved until now. It remains to be seen whether Tibsovo will be submitted to the Drug Reimbursement Evaluation Committee and complete the reimbursement process in the field of cholangiocarcinoma, where treatment options are limited. Cholangiocarcinoma is a highly aggressive cancer with a poor prognosis, with a 5-year relative survival rate of just 28.9%. In particular, 65% of patients with intrahepatic cholangiocarcinoma are diagnosed at an advanced stage where surgery is not feasible. Tibsovo is the only targeted therapy recommended by the National Comprehensive Cancer Network (NCCN) in the highest category (Category 1) as a second-line treatment for bile duct cancer. According to the Phase III ClarlDHy trial, Tibsovo reduced the risk of disease progression by 63% compared to placebo, with a median progression-free survival (PFS) of 2.7 months (1.4 months in the placebo group). Additionally, the median overall survival (mOS) was 10.3 months in the Tibsovo group, more than twice as long as the 5.1 months in the placebo group. Do-Youn Oh, Professor of Hematology-Oncology at Seoul National University Hospital, stated, “The development of drugs for cholangiocarcinoma has accelerated significantly in recent years. As new drugs are being developed, many companies are also actively working on the development of treatments for cholangiocarcinoma. Patients with cholangiocarcinoma must follow the doctors’ guidance and receive appropriate treatment rather than feeling discouraged, so they can participate in clinical trials and access new treatment opportunities.” Meanwhile, in the Phase III AGILE study that enrolled patients with acute myeloid leukemia, Tibsovo demonstrated improved event-free survival (EFS) when used in combination with azacitidine, and also significantly improved overall survival (OS). The median OS in the Tibsovo-treated group was 24.0 months (7.9 months in the placebo group), and long-term follow-up results showed that the median OS with Tibsovo combination therapy was 29.3 months, over 3.7 times longer than with placebo combination therapy. Kyu-Pyo Kim, Professor of Oncology at Asan Medical Center, said, “Tibsovo is indicated for IDH1 mutation-positive cholangiocarcinoma and acute myeloid leukemia, and has demonstrated excellent efficacy and safety in clinical studies. Considering that both studies allowed crossovers, the significant improvement in survival is a very meaningful result.” He added, “Given the limited treatment options for cholangiocarcinoma and acute myeloid leukemia, the emergence of a new targeted therapy with promising efficacy is expected to contribute to improving treatment outcomes and quality of life for patients with these diseases in Korea.”
- Company
- SK Bioscience wins the '8-year Prevenar patent dispute'
- by Kim, Jin-Gu May 22, 2025 06:09am
- In a patent infringement trial against Pfizer involving SK Bioscience's export of 'SKYPneumo' raw solution, SK Bioscience has won after eight years. The latest ruling is expected to enable SK Bioscience's global export of its pneumococcal conjugate vaccine 'SKYPneumo' raw solution. Ruling that 'Exporting SK Bioscience's SKYPneumo raw solution does not infringe on the patent'…an opportunity has been presented to export According to SK Bioscience, on May 21, the Supreme Court of Korea ruled in favor of SK Bioscience in a legal dispute filed by Pfizer for 'Cessation of infringement of Pfizer's Prevenar 13 (PCV13) pneumococcal conjugate vaccine patent.' The Supreme Court recently upheld the second trial ruling, which ruled in favor of SK Bioscience. SK Bioscience and Pfizer long fought over whether the overseas supply of SKYPneumo's raw solution infringes on Pfizer's 'Prevenar 13.' This eight-year legal battle began in 2017 when Pfizer filed a patent infringement lawsuit. The Supreme Court ruled that "Producing and supplying PCV13 finished products for research purposes does not infringe on the patent," adding, "Individual conjugates that make up PCV13 do not fall within the scope of the patent claim." It is analyzed that the Supreme Court's final ruling favoring SK Bioscience is expected to open the door for SK Bioscience to export SKYPneumo raw solution overseas. SK Bioscience says, "While supplying PCV13 finished products is not permitted, the latest ruling presents an opportunity for supplying individual conjugates overseas," and added, "We plan to initiate a new project to supply raw solution of individual conguates to regions with high vaccine demand, such as Southeast Asia and Central and South America. We also intend to pursue technology transfer through local partnerships." SK Bioscience vs. Pfizer: Patent Invalidation·Infringement Lawsuits…3 out of 4 cases transferred to the Supreme Court In addition to this case, the dispute between SK Bioscience and Pfizer surrounding the Prevenar 13 patent has been ongoing for a long time, including 'patent invalidation' lawsuits filed by SK Bioscience and 'cessation of patent infringement' lawsuits counter-filed by Pfizer. First, SK Bioscience challenged two related patents after developing SKYPneumo, a follow-up product to Prevenar 13. SK Bioscience challenged two of Pfizer's Prevenar 13-related patents: ▲Expiring in March 2026, 'a novel formulation that stabilizes immunogenic compositions and prevents precipitation' patent ▲Expiring in April 2027, 'a multivalent pneumococcal polysaccharide-protein conjugate composition' patent. SK Bioscience filed invalidation trials for both patents. Among these, the patent expiring in 2026 was successfully invalidated. The Intellectual Property Trial and Appeal Board (IPTAB) ruled in favor of SK Bioscience in January 2020, and the decision was finalized without an appeal from Pfizer. On the other hand, the patent expiring in 2027 ultimately failed to be invalidated. SK Bioscience argued for the invalidation of this patent, but received a dismissal from IPTAB in June 2015. SK Bioscience also lost in the second trial, which proceeded with SK Bioscience's appeal, in November 2017. SK Bioscience decided to appeal to the Supreme Court. However, they again received a losing ruling from the Supreme Court in 2018. In conclusion, the patent expiring in March 2026 was successfully invalidated, while the patent expiring in April 2027 was not. Consequently, domestic sales of SKYPneumo are prohibited until April 2027. Pfizer counter-filed a patent infringement prohibition lawsuit against SK Bioscience during this process. In 2017, Wyeth LLC, the patent holder of Prevenar 13, and Pfizer Korea, the domestic distributor, filed a lawsuit claiming that SKYPneumo infringed on their patent rights. This lawsuit also went to the third trial. Ultimately, the Supreme Court issued a recommendation for settlement in 2019, essentially siding with Pfizer. The Supreme Court ruled that Pfizer's patent was valid and prohibited the production and launch of SK Bioscience's SKYPneumo until 2027. SK Bioscience voluntarily withdrew the product approval for SKYPneumo. However, they re-obtained approval for SKYPneumo in June 2021. The conflict between the two companies continued thereafter. Facing difficulties with the domestic launch, SK Bioscience signed a license agreement to transfer related technology to a Russian pharmaceutical company to develop vaccines in that region. SK Bioscience supplied raw solution for its self-developed pneumococcal vaccine and finished pharmaceutical products for research. Pfizer and Wyeth LLC also interfered with this, filing a cession of patent infringement lawsuit in 2020. SK Bioscience countered, arguing that supplying raw solution for research and testing purposes, not finished products, to overseas markets was outside the scope of patent infringement. In August 2023, Pfizer won in the first trial. SK Bioscience appealed, and ultimately, the Patent Court overturned the first trial ruling and sided with SK Bioscience. This time, Pfizer appealed to the Supreme Court after losing in the second trial. Finally, the Supreme Court upheld the original ruling in the appeal. The two companies engaged in a long-term battle, going to the Supreme Court for three out of four cases, including patent invalidation lawsuits and patent infringement lawsuits. With this, most disputes between the two companies concerning the Prevenar 13 patent have been concluded. However, a trade commission dispute remains. The Korea Trade Commission under the Ministry of Trade, Industry and Energy is investigating SK Bioscience's unfair trade practices. This is because Pfizer and Wyeth LLC have filed a complaint requesting a cessation on exporting SK Bioscience's pneumococcal raw solution. In the pharmaceutical industry, there is an outlook that the trade commission dispute surrounding the export of pneumococcal raw solution will also be concluded soon due to this Supreme Court ruling.
- Company
- KPBMA proposes policies ahead of the presidential election
- by Kim, Jin-Gu May 22, 2025 06:08am
- The Korea Pharmaceutical and Bio-Pharma Manufacturers Association (KPBMA) proposes the "Pharmaceutical and Biotech Policies for Korea Ahead of South Korea's 21st presidential election, the Korea Pharmaceutical and Bio-Pharma Manufacturers Association (KPBMA) has proposed "The Ten Pillars of Pharmaceutical and Biotechnology Policies." KPBMA's proposal can be summarized in three keywords: supporting R&D, establishing an AI-based new drug development platform, and improving the drug pricing system. The KPBMA proposed initiatives, such as establishing performance-based R&D policies, creating a compensation system for domestically developed novel drugs, and supporting domestically-produced active pharmaceutical ingredients (APIs). The KPBMA also asked the government to develop an AI-driven drug discovery big data platform and cultivate specialized professionals in this field. Furthermore, they asked to establish a reinvestment cycle where drug price reduction exemptions based on R&D investment ratios are reinvested. They also urged the creation of a predictable drug pricing roadmap according to the balanced post-market management policies. In its policy report published on May 19, the KPBMA proposed its "Pharmaceutical and Biotech Policies for Korea's Healthy Future for the 21st Presidential Election." ◆Performance-Based R&D Policy Establishment = According to the KPBMA, the Korean government's 2023 R&D budget for the pharmaceutical and biotech sector was KRW 2.5826 trillion. However, only KRW 347.7 billion, or 13.5%, was allocated to direct corporate support. This differs significantly from the IT sector, where the proportion of corporate support reached 44.5%. In response, the KPBMA called for the establishment of a strategic R&D investment system. They stressed the need for institutional improvements, including expanding the 'Korean ARPA-H' project (a reference to the U.S. Advanced Research Projects Agency for Health) and applying exemption from preliminary feasibility studies. Furthermore, they urged for the continuous expansion of mega-funds to foster the creation of blockbuster novel drugs and to broaden tax credits for pharmaceutical and biotech companies' investments in bio-ventures. The KPBMA also stressed a shift in R&D budget policy towards an 'performance-based' approach. They emphasized the need to increase the corporate support ratio of government R&D budgets to over 30%. Additionally, they stressed the importance of strengthening support for Phase 2 and 3 clinical trials and global market entry to enhance the success rates of new drug development and commercialization. ◆Domestically Produced Vaccines·Treatments for Infectious Disease = The KPBMA asked for expanded support for research and development into next-generation vaccine platforms, such as mRNA and synthetic antigen technologies. They stressed the urgent need for swift countermeasures to the infectious disease crisis, citing initiatives like a '100-Day Vaccine Development Project' or 'Large-scale Rapid Global Clinical Projects.' Furthermore, the KPBMA proposed introducing a 'loss compensation system' and establishing pre-purchase schemes and long-term procurement contract systems to accelerate the development of national essential vaccines and treatments. For instance, countries like the United States and Japan actively utilized pre-purchase systems during the COVID-19 pandemic to shorten development timelines. ◆Establishing a Compensation System for Domestically Developed Novel Drugs = The KPBMA urged an improved drug pricing compensation system for domestically developed innovative new drugs. Currently, preferential drug pricing is only applied to novel drugs produced by innovative pharmaceutical companies or those that conduct domestic clinical trials. The KPBMA argued this should be expanded to pharmaceutical and biotech companies that contribute to the supply of essential medicines or job creation. The KPBMA also called for expanding the scope of the refund system, also known as a dual pricing system, to enhance global competitiveness. They anticipate that the refund system could be applied when the market launch plans for new drugs developed overseas or those subject to technology export are identified. ◆Supporting domestically produced APIs = According to the KPBMA, the number of domestic API manufacturers in Korea decreased significantly over the last decade, from 381 in 2013 to 296 in 2023. During the same period, API items sharply declined from 10,341 to 6,244. This trend is attributed to the lower price competitiveness of domestically produced raw materials, leading to an increased reliance on APIs from China·India. The KPBMA also pointed out the severe lack of policy support, such as preferential reimbursement drug pricing and tax incentives, as a reason for the lack of revitalization in API development. While the government introduced a system for preferential reimbursement pricing (68%, 5 years + 5 years) for using domestically produced raw materials, its application is limited to newly listed national essential medicines. Concerns remain regarding potential drug price reductions due to post-market management. In response, the KPBMA urged the establishment of a control tower to build a stable drug supply system and called for real-time investigation, analysis, and monitoring of drug supply and demand. In the mid-to-long term, they recommended developing a supply stability plan and establishing a government preferential procurement system for domestically produced essential medicines. Furthermore, they proposed expanding tax benefits for APIs under Korea's 'Act on Restriction on Special Cases Concerning Taxation.' They also emphasized the need for incentives, such as preferential drug pricing or manufacturing facility support, when diversifying supply sources or achieving self-sufficiency. ◆Accelerating AI-Driven Drug Discovery Initiatives = Korea's AI-driven drug discovery technology is estimated to be at 74% of the level of the United States, indicating a technology gap of approximately five years. While over 50 domestic AI drug discovery specialized companies demonstrate capabilities, a diagnosis suggests limitations in competing with global companies due to insufficient infrastructure, including data, specialized personnel, and computing resources. Accordingly, the KPBMA called for expanding federated learning and establishing a collaborative AI drug discovery acceleration project (AIDA). They proposed expanding the concept of the 'K-MELLODDY Project' to foster a nationwide data-driven open innovation ecosystem. ◆Developing a New Drug Discovery Big Data Platform = The KPBMA recommended establishing a government-funded 'New Drug Discovery Big Data Platform.' This platform would collect 'bio R&D public data' tailored to the specific needs of each drug discovery area and support industry access. Additionally, they called for developing an 'AI-based intelligent autonomous laboratory optimization model' to generate and accumulate high-quality experimental data for new drug development purposes. ◆Training AI-based Biotech Specialized Talent = Currently, the government is running an 'AI-based New Drug Development Education and Promotion Project.' The KPBMA proposed expanding this initiative to develop an 'AI Drug Discovery Industry-Academia Cooperative Convergence Talent Training Program,' where industry and universities jointly produce talent. They anticipate that establishing this program would significantly alleviate the shortage of AI talent in Korea. ◆Operating Industry-Led Advanced Industry Academies = The KPBMA proposed designating and operating an industry-led 'Pharmaceutical and Bio-Pharma Advanced Industry Academies.' This initiative would involve leading industry organizations coordinating government, industry, and academia to support practical education and employment linkages. Through this, they emphasized the importance of building a sustainable talent supply system and nurturing talent to prepare for future new technology demands. ◆Reinvestment of Drug Price Reduction Exemptions Based on R&D Ratios = The KPBMA pointed out that existing post-market drug price management systems, such as the drug price-volume agreement (PVA) program and actual transaction price-based drug price reductions, operate inefficiently. They highlighted that drug price reduction risks frequently arise, leading to adverse effects such as a decrease in companies' R&D investment capacity. Therefore, they called for strengthening compensation, including expanding exemptions based on R&D investment ratios during drug price reductions. They argued for establishing a virtuous cycle where the reduced drug prices are reinvested into R&D, leading to new drug creation. Specifically, they suggested Belgium's 'R&D Incentive Drug Pricing Model' as an alternative. ◆Establishing a Predictable Drug Pricing Roadmap = The KPBMA proposed establishing an intuitive and predictable long-term roadmap through balanced post-market management policies. They argued for rationalizing the fragmented post-market management systems and integrating the implementation schedules for drug price reductions to enhance predictability for the pharmaceutical industry. Furthermore, they also urged that pediatric and orphan drugs should be excluded from post-market management systems. The KPBMA argued for excluding pediatric and rare disease drugs, which are difficult to produce and have low profitability, from preferential drug pricing and post-market management selection.
- Company
- "Aiming the Alzheimer's market with ultrasound"
- by Whang, byung-woo May 22, 2025 06:07am
- Korea's biotech company Deepsonbio is gaining interest for its ultrasound-based brain disorder treatment technology. The company's recent exploratory clinical trials demonstrated the effect of improving cognitive function for patients with Alzheimer's disease and those with normal pressure hydrocephalus. Deepsonbio plans to enter the market on a full scale. Based on its multiple-low intensity pilant ultrasound (M-LIPUS) technology, Deepsonbio is projected to revolutionize treatments for brain disorders, such as Alzheimer's disease. DeepsonbioDaily Pharm met with Deepsonbio, a member of the KoreaBIO organization, to listen to the company's potential and vision. Deepsonbio is led by CEO Lee Dong-hyuk, who is currently a professor at Gachon University of Medicine and Science's Department of Biomedical Engineering. The company's key technology is its proprietary M-LIPUS. Unlike existing high-intensity focused ultrasound (HIFU) or transcranial magnetic stimulation (TMS), M-LIPUS uses safe, low-intensity ultrasound to non-invasively clear waste from the brain. Notably, the technology utilizes multiple ultra-sound generating devices, stimulating brain areas with ultrasound energy and promoting the cerebrospinal fluid (CSF) flow, ultimately increasing clearance of amyloid-β (Aβ), known as the pathogenesis of Alzheimer's disease. Deepsonbio CEO Lee said, "Disorders like Alzheimer's disease are caused by the accumulation of waste in the brain due to decreased CSF flow," and added, "M-LIPUS has been demonstrated to promote the elimination of Aβ and tau proteins, which are key waste in the brain. Clinical results showed that M-LIPUS improved cognitive function and neural functions." The company's recently finalized exploratory clinical results demonstrated Deepsonbio's technological advancement. It was found that within a month of ultrasound treatments, patients' cognitive function significantly improved. Furthermore, clinical evaluation showed a clear improvement in cognitive function with the trail making test (TMT) performance time, which is used to evaluate cognitive function in patients with Parkinson's disease, reduced by up to half. Additionally, biological indicators observed positive changes, including activated glucose metabolism in the brain and a significant reduction in beta-amyloid levels. When asked, "Are there any barriers to entering the brain disease treatment market using ultrasound technology?" CEO Lee emphasized that a shift in perspective led to the company's unique technology. Lee said, "Previous research focused more on opening the blood-brain barrier (BBB) rather than improving CSF fluidity. The ultrasound used in those studies was also less effective in humans, so no cases showed the same efficacy as Deepsonbio's low-intensity ultrasound." Global market entry and strategy to achieve 'KRW 100 Billion' mark Deepsonbio is targeting the global Alzheimer's treatment market. The global Alzheimer's treatment market is valued at approximately KRW 20 trillion as of 2023, showing rapid annual growth of over 20%. Deepsonbio aims to complete confirmatory clinical trials by the end of this year and secure additional medical device indications both domestically and internationally by early next year, thereby officially entering the global market. The company's strategy, led by CEO Lee, a regulatory expert, is expected to facilitate rapid market penetration. Considering that current Alzheimer's new drugs focus on early treatment to slow cognitive decline rather than fundamentally improving symptoms, there is an expectation that Deepsonbio's role could expand significantly. The company is actively pursuing domestic and international patent applications to enter the global market, including the United States. The company plans to secure a unique global medical device market position. Specifically, assuming a 6.4% market share of the domestic dementia population after receiving approval this year, Deepsonbio aims for approximately KRW 80 billion in revenue by 2028. CEO Lee stated, "Deepsonbio's device is not large, so there's no need to secure a separate space, which we predict will allow us to expand our influence in the non-reimbursable market rapidly." He added, "Our goal is to quickly generate revenue through the commercialization of our ultrasound technology." Expected to expand indications beyond Alzheimer's disease to normal pressure hydrocephalus Another area where Deepsonbio anticipates treating with its ultrasound technology is normal pressure hydrocephalus (NPH). The company has demonstrated that even with ultrasound treatment alone, patients with NPH significantly improved walking ability within a week, especially when existing treatments are limited. These results are evaluated to suggest the possibility of future indication expansion. Lee described Deepsonbio's technology as 'the technology of happiness.' In a reality where brain diseases like dementia and hydrocephalus severely impair the quality of life for entire families, he believes it is an innovative and valuable technology that can restore happiness to both patients and their families. Finally, Lee presented a vision to expand indications beyond the Alzheimer's disease treatment market to various brain disease treatment areas and to establish strong competitiveness in the global medical device market. Lee remarked, "Deepsonbio's technology can be a game-changer in the Alzheimer's disease treatment market," and added, "We will overcome the limitations of existing treatments, and our medical device will provide real and sustainable happiness to patients and their families."