- LOGIN
- MemberShip
- 2026-04-30 19:06:46
- Company
- Interest in Lotte Biologcs’ partner Ottimo Pharma rises
- by Kim, Jin-Gu Jun 20, 2025 06:05am
- Interest is growing in Ottimo Pharma, a biotech company that has signed a contract manufacturing agreement for its antibody-drug with Lotte Biologics. Founded in 2017, this UK-based biotech venture owns a new drug candidate called ‘Jankistomig’. The drug candidate bifunctional antibody targeting solid tumors, and the company plans to submit an Investigational New Drug (IND) application to the US Food and Drug Administration (FDA) within this year. Lotte Biologics announced on the 19th that it has signed an antibody-drug contract manufacturing agreement with Ottimo Pharma at the BIO INTERNATIONAL 2025 (Bio USA) event. The contract covers the production of drug substance (DS) for Ottimo Pharma's antibody drug Jankistomig at the Syracuse Bio Campus in New York. Ottimo Pharma was founded in 2017 in Kent, England, under the name Ultrahuman Eight Limited. In October last year, the company changed its name to Ottimo Pharma. At that time, Medicxi Ventures UK, a UK-based life science venture capital, participated as an initial investor. Medicxi led Ottimo Pharma's Series A investment round. Ottimo Pharma successfully secured USD 140 million (approximately KRW 190 billion) in Series A investment in December last year. To date, Ottimo Pharma’s only pipeline is Jankistomig. This candidate drug works by simultaneously inhibiting PD-1 and VEGFR2. The drug candidate was known to be designed based on camrelizumab, developed by China's Jiangsu Hengrui Pharmaceuticals. It is designed to reduce VEGF-related side effects while providing immune checkpoint inhibition effects. Jankistomig is in the preclinical stage and is being tested in the UK for solid tumors. There is no clinical trial number registered on ClinicalTrials.gov, a clinical trial registration site. This suggests that no official clinical trials have been initiated in any country, including the US and the UK. The company announced last year that it had secured Series A investment and would submit an Investigational New Drug (IND) application to the US FDA by the end of this year. At the time, Ottimo mentioned its development of other pipelines in addition to Jankistomig, but did not disclose specific substance names or stages.
- Company
- New pneumococcal vaccine expected to be launched
- by Whang, byung-woo Jun 19, 2025 06:04am
- As 'Capvaxive,' a 21-valent pneumococcal conjugate vaccine (PCV21) developed by MSD, is anticipated to receive marketing authorization in South Korea, competition in the market is likely to heat up. Product photo of CapvaxiveAccording to pharmaceutical industry sources, MSD has filed with the Ministry of Food and Drug Safety (MFDS) for marketing authorization of Capvaxive. It is expected to be approved by the second half of 2025. Capvaxive is a vaccine designed to prevent adults from serotype that causes most of the invasive pneumococcal disease (IPD). The safety and immunogenicity of Capvaxive were cofirnmed based on the Phase 3 STRIDE clinical trial, comparing Capvaxive to PCV20 in adults aged 18 years and above who have no prior history of pneumococcal vaccination. Capvaxive was found to be nonequivalent to PCV20 regarding 10 serotypes (3, 6A, 7F, 8, 10A, 11A, 12F, 19A, 22F, 33F) that are commonly included in PCV20. 10 out of 11 serotypes (9N, 15A, 16F, 17F, 20A, 23A, 23B, 24F, 31, 35B) that are included in Capvaxive but not in PCV20 were demonstrated to be superior to PCV20. Capvaxive was approved in the United States in June 2024 based on these study results, and it also obtained European approval in March. There is growing attention on whether Capvaxive will obtain Korean approval during the second half of this year, as it will be the third consecutive year a new pneumococcal vaccine is approved in South Korea. In late 2023, MSD's 15-valent vaccine, 'Vaxneuvance,' was expedited for inclusion in the pediatric National Immunization Program (NIP). Then, a year later, in October 2024, Pfizer's 20-valent vaccine, Prevenar 20, won MFDS approval. As the 21-valent vaccine, which is the higher serotype vaccine, is expected to be introduced in less than a year, competition is likely to get intense. If there are no setbacks to the approval process for Capvaxive, it is expected to be launched at the very end of the first half of next year. For instance, Vaxneuvance was launched in late April, and Prevenar 20 was launched in June exclusively for adults aged 18 years and above. The market is also highly likely to be competitive, with Vaxneuvance and Prevenar 20 competing for the pediatric NIP and Prevenar 20 and Capvaxive competing for the adult NIP. Regarding this, MSD Korea is expected to employ a marketing strategy that differentiates its vaccine portfolio for pediatric (15-valent) and adult (21-valent) populations. Indeed, MSD previously announced the use of tailored strategies for pediatric and adult populations during its Vaxneuvance launch 1st-anniversary media seminar. In the long term, Pfizer's Prevenar 20, with its first-mover advantage, will be competing directly with MSD's 21-valent Capvaxive, which includes a higher number of serotypes. Capvaxive will reportedly be preventing approximately 84-85% of adult IPD. This estimate is higher than the coverage for Prevenar 20. In this case, Pfizer is expected to defend its position by highlighting the performance and extensive clinical experience of Prevenar 20. Currently, Pfizer emphasizes that Prevenar 20 offers safety and convenience based on the well-established technology of Prevenar 13, validated through long-term pediatric and adult vaccinations, and with its 20-serotype coverage. Additionally, potential competition against Sanofi-SK bioscience is another variable. Although their commercialization timeline is the latest, if they succeed in developing a 21-valent vaccine, another equally strong competitor will emerge. Notably, the Sanofi vaccine is being developed for pediatric use, suggesting that the company will employ a future strategy to cover all age groups, from infants and young children to adults.
- Company
- K-Bios face string of clinical failures in H1
- by Son, Hyung Min Jun 19, 2025 06:04am
- A series of clinical trial failures for new drug candidates under development by Korean biotech companies in the first half of the year have raised concerns about the feasibility of future technology exports. Orum Therapeutics halted a clinical trial due to safety concerns, while Genexine and Bridge Biotherapeutics both failed to demonstrate statistical significance in their respective Phase II trials for glioblastoma and idiopathic pulmonary fibrosis. Stem cell therapy developers such as Anterogen and SCM LifeScience are also struggling to prove efficacy in clinical settings. According to industry sources on the 19th, Orum Therapeutics recently suspended its Phase 1 trial of ORM-5029. ORM-5029 was the company’s only pipeline in clinical trials targeting human epidermal growth factor receptor 2 (HER2), a major biomarker for solid tumors. The company received IND approval for ORM-5029 from the U.S. FDA in 2022. However, a severe adverse event (sAE) occurred during the trial, upon which the company reported it to the FDA. Due to toxicity issues, administration had to be halted even at low doses. ORM-5029 is a Degrader Antibody Conjugate (DAC) candidate. DACs combine Targeted Protein Degradation (TPD) mechanisms with Antibody Drug Conjugates (ADCs) and are expected to offer higher safety due to the use of TPD, which are small-molecule degraders. Orum emphasized that the sAE was limited to only the ORM-5029 substance and that there were no issues with the company's technology or platform itself. Orum plans to focus its resources on its blood cancer candidate ORM-1153, which also utilizes the company’s DAC platform. The company explained that it has shown strong GSPT1 degradation and robust anti-proliferative effects in blood cancer cell lines. Genexine and Bridge Biotherapeutics fail Phase II trials Genexine and Bridge Biotherapeutics both faced setbacks in Phase II trials. In March, Genexine's GX-I7 (Interleukin-7), an immune-oncology drug candidate, failed to demonstrate efficacy in glioblastoma mulifrome (GBM) patients. GX-I7 is a new drug candidate that maximizes immune anticancer effects by inducing T-cell amplification in the body. GBM is a type of glioma, a malignant tumor that originates in the brain. Despite surgery and chemotherapy, the five-year survival rate for GBM is only 5%, with an average survival time of about a year. The Phase II trial for GX-I7 enrolled 20 patients with recurrent or progressive glioblastoma, and evaluated a combination of the GX-I7 and bevacizumab (Avastin), a VEGF inhibitor used as a targeted therapy. Bevacizumab inhibits angiogenesis to prevent tumor growth, and its combination with existing anticancer drugs was expected to enhance therapeutic efficacy. However, no significant improvement was observed in the primary endpoints of progression-free survival (PFS) and overall survival (OS). Meanwhile, Bridge Biotherapeutics announced in April that its top-line data analysis results showed that its idiopathic pulmonary fibrosis (IPF) candidate BBT-877 failed to demonstrate a statistically significant improvement in the primary endpoint of forced vital capacity (FVC) change at 24 weeks. BBT-877 is an innovative novel drug candidate that selectively inhibits the novel target protein autotaxin. Autotaxin is a protein known to bind to intracellular receptors and be involved in pathological mechanisms such as fibrosis and tumorigenesis. BridgeBio previously secured global exclusive rights to BBT-877 from LegoChem Bio (now LigaChem Bio) in 2017. In May, BridgeBio received a recommendation from the IDMC to continue the clinical trial. The Phase 2 clinical trial of BBT-877 was conducted in 5 countries - South Korea, the United States, Australia, Poland, and Israel - to evaluate the efficacy, safety, and tolerability of the drug in patients with idiopathic pulmonary fibrosis (IPF). A total of 129 patients participated, and the study results showed that changes in FVC were observed in both the drug group and the placebo group; however, there was no statistically significant difference between the two groups. Bridge Biotherapeutics licensed out BBT-877 to Boehringer Ingelheim in 2019 in a deal worth up to KRW 1.5 trillion. Upon transferring BBT-877, which was in Phase 1 clinical trials, the company received approximately KRW 600 billion in upfront and milestone payments (short-term milestones). In late 2019, following the completion of Phase I clinical trials for BBT-877, BridgeBio paid approximately KRW 50 billion to LigaChem Bio as milestone revenue sharing. However, in 2020, Boehringer Ingelheim returned the rights to BBT-877 due to potential toxicity issues. BridgeBio determined that the toxicity issues were caused by high-dose drug administration in additional experiments and decided to develop the candidate on its own, but failed to demonstrate its efficacy in trials. stem cell therapy developers also struggling with commercialization Stem cell therapy developers are also facing commercialization hurdles. SCM LifeScience failed to achieve statistical significance in Phase 2 clinical trials of its stem cell therapy candidate SCM-CGH. This is the company's second failed attempt at commercialization following the failure of its acute pancreatitis clinical trial in 2022. The trial, which targeted patients with steroid-resistant or steroid-dependent chronic graft-versus-host disease, was conducted from 2017 to 2024 at 11 hospitals in South Korea, including Seoul St. Mary's Hospital. The results of the Phase II clinical trial of SCM-CGH showed no statistically significant difference in the primary efficacy endpoint, the overall response rate (ORR) at 12 weeks. Upon closer examination, the ORR at 12 weeks was higher in the placebo group than in the SCM-CGH group, and the results were not statistically significant. Anterogen failed to demonstrate the efficacy of its stem cell therapy ALLO-ASC-DFU in the U.S. Phase III clinical trial. In the trial, ALLO-ASC-DFU recorded a complete wound closure rate of 46%, which was lower than the 60% in the control group that was treated with hydrogel sheets. The therapy had garnered attention as a treatment for diabetic foot ulcers (DFU), but its failure to meet the key primary endpoint has significantly reduced the likelihood of its FDA approval. Anterogen is conducting further analyses to revise its development strategy.
- Company
- KOR-JPN jointly launches Healthcare Distribution Alliance
- by Son, Hyung Min Jun 19, 2025 06:03am
- (From the left) Jun-Jae Hyeon (CEO, Dongwon Healthcare), Jun-ho Hyun (CEO, Dongwon Pharmaceutical Wholesale), Seung-Uk Eom (CEO, Boksan Nice), and Seongwook Cho (Country Manager, Suzuken Korea) Three pharmaceutical distribution companies in Korea and Japan have joined forces to launch the Healthcare Distribution Alliance to lead the domestic market by introducing advanced overseas models. The alliance aims to go beyond simple logistics agreements – it seeks to build an innovative cooperation structure where companies can share capital and operational know-how, and combine each company's strengths to transform the pharmaceutical distribution market. Jun-Jae Hyeon (CEO, Dongwon Healthcare), Jun-ho Hyun (CEO, Dongwon Pharmaceutical Wholesale), Seung-Uk Eom (CEO, Boksan Nice), and Seongwook Cho (Country Manager, Suzuken Korea) recently met with reporters to explain the alliance's goals. Eight affiliates of Dongwon Pharmaceutical Group, Boksan Nice, and Suzuken have signed a business partnership agreement and established an organizational framework for cooperative operations at the alliance level. As part of the collaboration, the companies also entered into a capital partnership, with Suzuken acquiring a 33.6% stake in Gyeongnam Dongwon Pharmaceutical, and Boksan Nice acquiring a 3.4% stake in Gyeongnam Dongwon Pharmaceutical. This alliance goes beyond simple logistics cooperation by sharing capital and strategy direction of the companies. With the direct participation and investment of Suzuken, a major Japanese pharmaceutical distribution company, the alliance aims to pursue a long-term model that pursues the maximization of distribution productivity, supply chain stability, and function as part of a social infrastructure. CEO Seung-Uk Eom said, “We decided to pursue this alliance to survive in the rapidly changing pharmaceutical distribution industry and create growth opportunities through innovation. We aim to realize economies of scale through the alliance between Dongwon Pharmaceutical, Boksan Nice, and Suzuken and maximize productivity in the pharmaceutical distribution market while driving innovation for mutual growth.” Industry observers expect synergy from the partnership. Dongwon Pharmaceutical Group and Boksan Nice each reported over KRW 1 trillion in annual sales last year. Suzuken, one of Japan's top three pharmaceutical distributors, posted annual revenue exceeding JPY 2 trillion (approx. KRW 19 trillion) in 2023. CEO Jun-ho Hyun emphasized, “As the pharmaceutical distribution environment evolves and capital requirements grow, scaling up is no longer an option – it’s a necessity. We aim to establish a Korean-style large-scale distribution model and guide the future direction of the market.” The alliance anticipates increased distribution-related costs and volatility in the coming years. To address this, it aims to build a robust infrastructure and reduce labor dependency. Plans include exploring hospital market strategies, logistics outsourcing services, private-label (PB) healthcare products, and the potential introduction of Suzuken’s current Japanese business operations into the Korean market. CEO Seung-Uk Eom noted, “In the short term, we’ll focus on collaboration between logistics centers within the alliance, which is expected to reduce stockouts and delivery lead times through optimal inventory and shipping operations.” He added, “In the long term, we plan to build systems such as enterprise resource planning (ERP), web order systems (WOS), and customer relationship management (CRM). Given the significant time and cost required for IT system development, combining the long-standing expertise and ideas of Suzuken, Boksan Nice, and Dongwon Pharmaceutical will not only facilitate joint development but also greatly contribute to future logistics innovations such as the modernization of logistics and improvements in operational efficiency involving robots and AI. CEO Seung-Uk Eom added, “Beyond transportation management systems (TMS) and quality control standards, we will also build a foundation system for environmental, social, and governance (ESG) and seek ways to advance them.” CEO Jun-ho Hyun said, “Profit margins for pharmaceutical distributors have been steadily shrinking. Survival through sales promotion activities alone is becoming difficult. We must scale up and differentiate through cost reduction and pharmaceutical partnerships.” ”Will seek to implement Japanese-style wholesale structure in Korea" The alliance is eyeing the Japanese model, where pharmaceutical distribution is treated as a core part of national infrastructure, with government, pharma companies, wholesalers, and hospitals working in unison. Even logistics center placements are coordinated with government authorities, and disaster response systems are embedded into the distribution network. Japan’s market is dominated by major distributors like Medipal, Alfresa, and Suzuken, which fulfill roles as social infrastructure through close cooperation across the pharmaceutical supply chain. CEO Jun-jae Hyeon noted, “In Japan, systems are in place to ensure medicine continues to flow even during national disasters like earthquakes and tsunamis. We aspire to build such a socially integrated distribution system here in Korea.” Country Manager Seongwook Cho said, “, “Japan has established a virtuous cycle model that contributes to the national health insurance budget by minimizing the deterioration of medicine quality and the amount of expired medicines through infrastructure development. Although this may not be immediately achievable in South Korea, we will do our best to prepare for it." In addition, as more and more pharmaceutical companies are expected to develop new drugs such as biological agents, anticancer drugs, and orphan drugs, it is necessary to establish a system to manage and deliver these drugs. The association aims to provide one-stop services tailored to their needs. Country Manager Seongwook Cho said, “Suzuken already communicates and conducts business with many multinational pharmaceutical companies in Japan. We are aware that the companies have high standards for quality control and other global requirements. Our association’s goal is to meet the standards set by such global companies in various areas, including logistics and cold chain.” CEO Jun-jae Hyeon said, “In Japan, there are various pharmaceutical platforms, with distribution companies at the center of each. All transactions between healthcare institutions and related organizations are conducted through distribution companies. We will strive to establish a similar structure in Korea, where distribution companies play a central role in facilitating various activities.”
- Company
- Takeda launches new drug 'Fruzaqla' in Korea
- by Whang, byung-woo Jun 18, 2025 10:28am
- Product photo of Takeda Pharmaceutical's Fruzaqla Takeda Pharmaceutical Korea (CEO Kwang-kyu Park) announced on June 16 that the company has officially launched its 'Fruzaqla (fruquintinib)' in South Korea. Fruzaqla is the first new treatment for metastatic colorectal cancer. This drug selectively inhibits Vascular Endothelial Growth Factor Receptor (VEGFR)-1,2, and 3. It is expected to provide a new treatment option for patients subjecting to fourth-line or later treatment who had limited treatment options previously. According to the 2024 statistics, colorectal cancer is one of the cancer types with prevalence ranking No.2 in South Korea. Approximately 20% of the patients are found to be metastatic at diagnosis. It has been reported that 50-60% of the patients who do not have metastasis during the initial diagnosis experience metastasis to other organs. In such cases, the survival rate is only 20.6%. However, treatment options for third-line treatment and above in metastatic patients are limited. Thus, many patients and doctors have voiced high demands for new treatment options that are effective and less burdening. Fruzaqla is a new treatment option for metastatic colorectal cancer that has emerged after over a decade and can be used regardless of specific biomarker status. Fruzaqla was designed to be effective by selectively inhibiting VEGFR-1, 2, and 3. It also minimizes off-targeted toxicity, thereby avoiding unnecessary targets. It has the mechanistic advantage of high-level drug exposure and continuous target inhibition. The efficacy·effectiveness of Fruzaqla has been demonstrated for adult patients with metastatic colorectal cancer who have previously been treated with a chemotherapy containing fluoropyrimidines, oxaliplatin, and irinotecan plus an anti-VEGF or anti-EGFR agent (for patients with wild-type RAS), and whose disease has progressed or who does not show tolerability following treatment with trifluridine/tipiracil and/or regorafenib. The basis of approval was the Phase 3 FRESCO-2 clinical study. The study results showed that the Fruzaqla group had a median overall survival (mOS) of 7.4 months, which was higher than the 4.8 months mOS of the placebo group, and also had a 34% lower mortality risk. Additionally, the Fruzaqla group had a median progression-free survival (mPFS) of 3.7 months, more than double the 1.8 months in the placebo group, corresponding to a 68% reduction in disease progression or death risk. Furthermore, Fruzaqla is an oral treatment that can be taken once daily with convenience without complicated mean conditions. It is expected to yield a positive impact on improving quality of life in addition to the treatment effects. Dr. Sang Cheul Oh, Korea University Guro Hospital (Korean Cancer Study Group's Colorectal Cancer Division Head), said, "Metastatic colorectal cancer, despite its high prevalence and aggressiveness, had been the key cancer type with unmet needs due to limited treatment options for fourth-line or later treatments." Dr. Oh added. "Fruzaqla works by selectively inhibiting VEGFR-1, 2, and 3, and is highly effective but reduced toxicity. It will be a significantly meaningful option for patients at later cancer stages who are undergoing fourth-line or later treatments." Kim Mi-seung, Takeda Pharmaceutical's oncology business unit, said, "Fruzaqla is an innovative new drug that can be used regardless of the specific biomarker status, emerged to the metastatic colorectal cancer treatment setting after 10 years, based on the FDA record. It is expected to solve unmet needs for a wide range of patients," and added," Takeda Pharmaceutical will continue to put efforts in providing improved treatment options for Korean patients, including those for metastatic colorectal cancer."
- Company
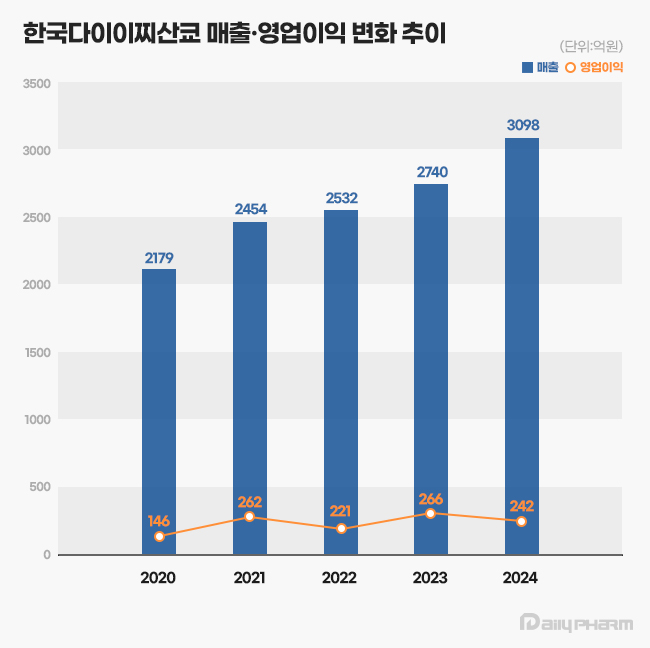
- Daiichi Sankyo exceeds ₩300B in sales…new drug drives
- by Son, Hyung Min Jun 18, 2025 06:01am
- Daiichi Sankyo Korea has exceeded KRW 300 billion in sales for the first time, led by its cardiovascular products, Antibody-Drug Conjugate (ADC), and new anticancer drugs. The company is successfully transitioning its portfolio towards new ADC drugs while maintaining robust growth from established cardiovascular products like Sevikar, Lixiana, and Olmetec. According to the Korea Financial Supervisory Service (FSS)'s electronic disclosure system on June 16, Daiichi Sankyo Korea's sales last year reached KRW 309.8 billion, a 13% increase from the previous year. During the same period, operating profit decreased by 9%, from KRW 26.6 billion to KRW 24.2 billion. Daiichi Sankyo Korea considers its sales for 2024 based on the Japanese fiscal year, covering April of last year to March of this year. Daiichi Sankyo Korea Daiichi Sankyo Korea's sales have been steadily increasing since 2020. The company first surpassed KRW 200 billion in revenue in 2020 with KRW 217.9 billion, followed by a continuous upward trend, reaching KRW 245.4 billion in 2021, KRW 253.2 billion in 2022, and KRW 274.0 billion in 2023. Notably, an analysis suggests that collaboration with the domestic pharmaceutical company Daewoong Pharmaceutical on some cardiovascular products, such as Lixiana and Sevikar, has created a synergistic effect. Daiichi Sankyo Korea signed co-promotion agreements for Sevikar in 2013 and Lixiana in 2015 with Daewoong Pharmaceutical, and this partnership continues to date. Among them, the highest revenue generator is the Direct Oral Anticoagulant (DOAC), Lixiana. According to market research firm UBIST, Lixiana's prescription sales last year was KRW 117.5 billion, a 12% increase compared to KRW 105.3 billion in 2023. DOACs are anticoagulants that prevent blood clots by directly acting on blood coagulation factors. They are increasingly being used in clinical settings as they replace warfarin, which inhibits Vitamin K metabolism. In Korea, Xarelto was approved in 2009, followed by Pradaxa and Eliquis in 2011, and Lixiana in 2015. Despite being the last to be launched among DOACs, Lixiana has rapidly increased its prescription performance, backed by demonstrated clinical data, and has maintained its market dominance since 2019. With annual growth of around 10%, its prescription performance nearly doubled in five years, from KRW 60.4 billion in 2019. Its market share in the overall DOAC market also expanded from 33% in 2019 to 45% last year. Sevikar, an olmesartan-based combination therapy for hypertension, continues to maintain its strong performance in prescription revenue. Sevikar's prescription revenue last year was KRW 68.8 billion, a 4% increase from the previous year. Despite numerous global and domestic pharmaceutical companies entering this market, Sevikar's prescription sales continue to grow. Sevikar's prescription revenue, which was KRW 53.4 billion in 2019, surpassed KRW 60 billion in 2022. In 2023, it recorded KRW 65.9 billion, demonstrating five consecutive years of increased prescription sales. The triple combination hypertension drug Sevikar HCT also maintained its growth trajectory. Sevikar HCT's prescription sales for the last year totaled KRW 42.1 billion, representing a 4% increase from the previous year. Daiichi Sankyo Korea generated approximately KRW 140 billion in prescription sales solely from olmesartan-based hypertension treatments, including Sevikar HCT (KRW 42.1 billion), Olmetec (KRW 30.6 billion), and Sevikar (KRW 68.8 billion). The 5 ADC Strategy...Will it achieve R&D success after Enhertu? Daiichi Sankyo Korea is working towards transitioning from a cardiovascular-focused company to a leader in oncology. The company is particularly concentrating its R&D capabilities on the ADC field, focusing on new growth engines. Following the already approved Enhertu, it is pursuing a '5 ADC strategy' and preparing for the launch of various other therapeutic agents, including Datroway, patritumab deruxtecan, DS-7300, DS-700, and DS-6000. An ADC is a new anticancer drug designed by linking an antibody that binds to a specific target antigen on the surface of cancer cells with a drug that has cell-killing capabilities using a linker. The advantage of ADCs is their ability to selectively target cancer cells by utilizing the antibody's target specificity and the drug's cytotoxic activity, thereby maximizing therapeutic efficacy while minimizing side effects. ADC anticancer agentWhile first-generation ADCs, such as Roche's Kadcyla, were initially limited to breast cancer indications, second-generation ADCs are successfully securing various other indications. Among these, Enhertu is a second-generation new ADC drug introduced by Daiichi Sankyo Korea. Enhertu is a next-generation ADC that links a monoclonal antibody with the same structure as trastuzumab (which binds to specific target receptors overexpressed on the surfaces of cancer cells) and a highly potent, novel topoisomerase I inhibitor payload via a tumor-selective, cleavable linker. Currently, Enhertu won domestic approval for HER2-positive breast cancer, gastric cancer, and non-small cell lung cancer, and is primarily used as a second-line treatment. Its potential as a first-line treatment for breast cancer is also currently being investigated. Daiichi Sankyo is also preparing to launch its second new ADC drug, Datroway. This ADC targets the Trop-2 protein and has been approved in the U.S. for the treatment of breast cancer. The Trop-2 protein is a cell membrane antigen overexpressed in breast cancer, particularly in over 90% of triple-negative breast cancer cases. Datroway binds to the Trop-2 protein and delivers cytotoxic substances into the cancer cells. It has the advantage of maximizing the benefits of targeted therapy and cytotoxic chemotherapy while minimizing damage to healthy cells. Currently, Daiichi Sankyo is co-developing and co-marketing Enhertu and Datroway with AstraZeneca. Daiichi Sankyo is also developing an ADC with Merck. patritumab deruxtecan, which targets HER3, showed efficacy in EGFR-mutated patients compared to platinum-based chemotherapy in the Phase 2 HERTHENA-Lung01 study. Daiichi Sankyo continues to conduct research for subsequent ADC candidates after Enhertu, which targets the HER2 biomarker. The company is also jointly conducting clinical studies with Merck on DS-7300, which targets B7-H3 (an emerging new biomarker in solid tumors), and DS-6000, a CDH6-targeting ADC.
- Company
- Zejula, a new standard ovarian cancer maintenance therapy
- by Whang, byung-woo Jun 18, 2025 06:00am
- Ovarian cancer is often diagnosed at an advanced stage due to the difficulty of early detection, and it is known for its high recurrence rate even after initial treatment. First-line maintenance therapy aimed at delaying recurrence as much as possible after surgery and chemotherapy became a key strategy that determines treatment outcomes for ovarian cancer. Recently introduced PARP inhibitors have emerged as a standard option for maintenance therapy, and the use of biomarkers to guide patient selection has been a major advancement, enabling better prediction of which patient subgroups are likely to benefit the most. In an interview with Dailypharm, Professor Jae Kwan Lee of the Department of Obstetrics and Gynecology at Korea University Guro Hospital, and Dr. Bradley Monk of the Florida Cancer Specialists & Research Institute stressed the need for institutional support for personalized treatment of ovarian cancer. Long-term efficacy of Zejula as first-line maintenance therapy for ovarian cancer proven Ovarian cancer is difficult to diagnose at an early stage and often recurs after initial treatment, raising the importance of maintenance therapy. This is why first-line maintenance therapy to delay recurrence as much as possible after surgery and chemotherapy has become a key strategy in ovarian cancer treatment. Professor Lee said, "First-line maintenance therapy is becoming a critical turning point in ovarian cancer treatment. HRd (homologous recombination deficiency)-positive patients showed an average progression-free survival period extension of approximately 2 years when receiving first-line maintenance therapy after surgery. Given the high recurrence rate of ovarian cancer, maintaining remission for as long as possible is key to successful outcomes, and first-line maintenance therapy serves as a highly effective strategy in this regard." Jae Kwan Lee, Professor of Obstetrics and Gynecology, Korea University Guro Hospital (President, Korean Society of Gynecologic Oncology)One of the changes in the domestic treatment environment for ovarian cancer came with the expansion of reimbursement criteria for the PARP inhibitor Zejula (niraparib) to HRd-positive ovarian cancer in October last year. The reimbursement extension of the PARP inhibitor Zejula was significant because of its biomarker. Approximately 50% of all ovarian cancer patients are HRd-positive, and about half of them, or 25%, have BRCA gene mutations. In addition, studies continue to demonstrate the efficacy of PARP inhibitors in HRd-positive patients. Professor Lee said, “In the past, reimbursement was limited to patients with BRCA mutations, so HRd-positive patients who were BRCA-negative could not choose to use Zejula due to the financial burden. However, since the reimbursement criteria were extended to include HRd-positive patients, many patients are actively starting Zejula treatment.” Zejula is currently one of the most promising PARP inhibitors for first-line maintenance therapy for ovarian cancer. In particular, the long-term follow-up data from the PRIMA study published last year has enhanced the reliability of Zejula. In the PRIMA trial, Zejula increased progression-free survival (PFS) by more than twofold in HRd-positive ovarian cancer patients compared to placebo. Additionally, at the time of clinical confirmation, the median PFS in the Zejula treatment group was 24.5 months, compared to 11.2 months in the placebo group, showing a significant difference. The 5-year PFS rate was also 35%, approximately twice as high as that of the placebo group. Dr. Monk stated, “Previously, there were concerns that long-term use of PARP inhibitors could lead to drug resistance, but this data confirms that such a possibility is low. These long-term follow-up results will serve as a strong source of reliability for doctors who have been hesitant about prescribing Zejula in the long term." He further explained, “Zejula can be used as a first-line maintenance therapy for all patients who respond to platinum-based chemotherapy (all-comer), but it is known to show the most effective results in HRd-positive patients. Since approximately half of all ovarian cancer cases are classified as HRd-positive, Zejula is increasingly being considered as a key option when setting treatment strategies.” “Diagnostic hurdles remain despite Zejula’s extended reimbursement for HRd-positive ovarian cancer” One of the main reasons for the popularity of Zejula is its ease of administration. While other PARP inhibitors require twice-daily dosing, Zejula can be taken once daily, improving medication adherence. Professor Lee said, “For patients to adhere to long-term maintenance therapy without becoming fatigued, treatment convenience is crucial. Zejula’s once-daily dosing regimen has had a positive impact on patients' ability to remain on therapy over the long term without discontinuation." Bradley Monk, MD, Medical Director of Late-Phase Clinical Research Program, Florida Cancer Specialists & Research Institute Dr. Monk added, "Zejula has the advantage of having relatively low drug-drug interactions, which makes it a safer option when used in combination with other drugs. This is a significant advantage for elderly patients with comorbidities or those receiving complex medication regimens.” Meanwhile, with the expansion of reimbursement criteria for HRd-positive ovarian cancer, it has become essential to determine whether a patient is HRd-positive before establishing a treatment strategy, but access to such HRd diagnostic tests remains a barrier. Currently, BRCA1/2 mutation testing for ovarian cancer patients is relatively affordable through national support programs and partial health insurance coverage. However, genomic panel testing required to confirm HRd status is not covered by insurance, leaving patients to bear the full cost of approximately KRW 2.5 million. Professor Lee pointed out, “HRd testing is essential for HRd-positive patients to receive Zejula treatment, but the fact that the test is not covered by insurance and must be paid out of pocket is a major institutional contradiction. Policy improvements should be made so that HRd diagnostic tests can settle as a diagnostic tool accessible under the same criteria as BRCA tests.” In contrast, access to HRd tests has been rapidly improving overseas. Dr. Monk stated, “Currently, more than 10 companies in the United States offer HRd tests, and some provide the service at very low costs. HRd diagnostic tests can serve as an important basis for predicting treatment response to PARP inhibitors such as Zejula.” For this reason, the Korean Society of Gynecologic Oncology is also known to be actively collecting supporting data to officially propose reimbursement for HRd tests to Korean health authorities. If HRd tests are promptly covered by health insurance, patients will be able to receive the necessary testing without financial burden and fully enjoy the benefits of targeted maintenance therapy such as Zejula. In addition, Professor Lee, who has been appointed as the President of the Korean Society of Gynecologic Oncology, emphasized his commitment to advancing precision medicine based on the genetic profiling of ovarian cancer. Professor Lee stated, “The society plans to focus on how to diagnose and manage the genetic characteristics of ovarian cancer. In other countries, there are already detailed clinical guidelines in place for individuals with BRCA mutations, and I believe similar protocols are needed in Korea as well." He concluded, "Since ovarian cancer often occurs alongside other cancers such as breast or endometrial cancer, collaboration with other specialties, including surgical departments, is essential. Establishing a multidisciplinary, patient-centered integrated care system through close coordination with various medical fields is another key priority for the society."
- Company
- Adstiladrin receives orphan drug designation in Korea
- by Eo, Yun-Ho Jun 18, 2025 05:59am
- The new bladder cancer drug Adstiladrin has been designated as an orphan drug in Korea. The Ministry of Food and Drug Safety recently announced the news in a orphan drug designation announcement. The specific indication for designation is “treatment of BCG-refractory high-risk non-muscle-invasive bladder cancer (NMIBC) with carcinoma in situ (CIS) with or without papilloma.” Adstiladrin (nadofaragene firadenovec-vncg) received FDA approval in the United States in 2022. This drug uses a non-replicating adenovirus vector to deliver the human interferon alpha-2b gene, inducing an immune response by directly expressing the protein within the bladder epithelium. Adstiladrin demonstrated efficacy through the NCT02773849 clinical trial that involved 157 patients with bladder cancer. In the study, 51% of the 98 patients treated with Adstiladrin achieved complete response (CR). The median duration of response was 9.7 months. In addition, 46% of patients who achieved CR remained recurrence-free at 12 months after treatment. The most commonly reported side effects were instillation site discharge (33%), fatigue (24%), bladder spasms (20%), urinary urgency (19%), and hematuria (17%). The rate of discontinuation due to side effects was 1.9%. Non-muscle-invasive bladder cancer (NMIBC) is an early-stage bladder cancer confined to the bladder mucosa, accounting for approximately 70–80% of all bladder cancers. Among these, high-risk patients include those with carcinoma in situ (CIS) or multifocal high-grade tumors, which have a high risk of recurrence and invasion. Although BCG instillation therapy is used as first-line treatment, approximately 30–50% of patients eventually experience recurrence or become resistant within a few months. While radical cystectomy is considered the standard treatment thereafter, as it is a highly invasive surgery, there has been a continued demand for bladder-preserving therapeutic alternatives.
- Company
- Long-acting HIV treatment shifts HIV treatment paradigm
- by Whang, byung-woo Jun 18, 2025 05:59am
- With insurance reimbursement now available for the long-acting HIV (human immunodeficiency virus) treatment Vocabria+Rekambys injection therapy, expectations are high on how it will meet the unmet demand. Compared to existing treatments that require daily administration, the new treatment is administered only 6 times a year, offering overwhelming convenience. With its accessibility barriers removed with reimbursement approval, the treatment is expected to gain influence quickly in the market. On the 17th of this month, GSK Korea held a meeting to commemorate the domestic launch of the Vocabria+Rekambys injection therapy and highlighted the treatment's effects and significance. The Vocabria+Rekambys combination was approved by the Ministry of Food and Drug Safety in February 2022 as a combination therapy for the treatment of HIV-1 infection in adult patients who are virologically suppressed, have no history of virological failure, and have no known or suspected resistance to cabotegravir or rilpivirine. Jae-Phil Choi, Professor of Infectious Diseases at Seoul Medical Center Jae-Phil Choi, Professor of Infectious Diseases at Seoul Medical Center, who presented at the event, stated, “Thanks to advancements in treatment that allow effective suppression of the virus, HIV has become a chronic condition that can be managed for life, similar to diabetes or hypertension. However, despite advancements in HIV treatment, social discrimination and stigma against people living with HIV remain widespread in South Korea.” According to Professor Choi, negative perceptions toward HIV influence treatment adherence among infected individuals, leading many to hesitate about seeking active early treatment and serving as a barrier to continuing treatment. According to the 2024 HIV Treatment Awareness Survey conducted by Love4One, a group representing people living with HIV, 73% of respondents in Korea reported feeling anxious that taking HIV medication might expose their status to others or attract unwanted attention. While conventional HIV treatments require daily oral dosing (365 days a year), injectable therapy using Cabotegravir (Vocabria) can reduce the dosing frequency to as little as once a month or once every two months—up to just 6 times a year. These advantages are expected to be effective in alleviating the anxiety caused by social stigma, which is one of the major challenges faced by HIV-infected individuals. Professor Choi emphasized, “Domestic HIV-infected individuals feel a significant psychological burden when taking medications, and as a result, they prefer long-acting HIV injections over oral medications that require daily intake. The Vocabria+Rekambys injection therapy could serve as an option that reduces anxiety about disclosure of infection status and alleviates the inconvenience and concerns associated with daily dosing, thereby providing high treatment adherence and satisfaction.” “High demand for bi-monthly dosing, sufficient flexibility in administration” Next, Professor Yeon-Sook Kim of the Department of Infectious Diseases at Chungnam National University Hospital, who participated in the clinical trial of the Vocabria+Rekambys injection therapy, stated that considering the treatment efficacy of the regimen, it may be possible to adjust treatment options according to the lifestyles of people living with HIV. Yeon-Sook Kim, Professor of Infectious Diseases at Chungnam National University HospitalProfessor Kim stated, “Analysis of data from HIV-infected individuals in Asia (n=41), including Korean HIV-infected individuals in the Phase IIIb clinical trial of the Vocabria+Rekambys injection therapy, showed that 83% of participants maintained viral suppression at Week 96 of treatment, with no reported cases of defined virological failure. This suggests that the Vocabria+Rekambys injection therapy could be an effective treatment option for HIV-infected individuals in Korea.” Professor Kim added, “A survey of HIV-infected individuals in Korea showed a high demand for ‘long-acting treatment with less frequent dosing’ for HIV treatment. With the recent reimbursement approval, it is worth considering changing the treatment option to a long-acting HIV injection according to the lifestyle of infected individuals.” However, along with expectations for reimbursement for the Vocabria+Rekambys injection therapy, there is also the concern that patients may feel burdened by having to visit the hospital more frequently. In the case of the Vocabria+Rekambys injection therapy, there is a 7-day period before and after the standard administration cycle, allowing for a total of 14 days of flexibility in administration, but this means that some patients may find it difficult to switch depending on their circumstances. “From the patient's perspective, there are cases where it is difficult to take time off work, and these patients receive prescriptions for oral medication once every 6 months. In such cases, it may be difficult to recommend switching to an injection therapy. However, for patients who typically receive a prescription every 3 months, we can consider switching to a long-acting injection administered every 2 months.” Professor Kim added, “Additionally, in cases where it is difficult to align the administration schedule due to overseas business trips, it is possible to reintroduce oral therapy. Since it is possible to switch back to oral medication after using the injection therapy, we believe there is sufficient flexibility in its administration.”
- Company
- Lotte-Axcelead-Kanaph signs MOU for joint ADC development
- by Kim, Jin-Gu Jun 17, 2025 05:58am
- Lotte Biologics announced on the 16th that it has signed a three-party memorandum of understanding (MOU) with global new drug development company Axcelead and innovative new drug development company Kanaph Therapeutics to establish an ‘ADC Toolbox’ for the development of antibody-drug conjugates (ADC). Under the agreement, the three companies will collaborate on joint research and development of linkers and payload technologies, which are key for the development of antibody-drug conjugates, which are regarded as next-generation anticancer drugs. Axcelead is a global contract research organization (CRO) spun off from Takeda Pharmaceutical in Japan. It will utilize Takeda's library of over 1.2 million compounds and more than 1,000 new drug development data to identify novel payload candidates that have not been applied to existing ADCs. Kanaph Therapeutics will focus on building an innovative platform that overcomes the limitations of existing linkers and payloads in ADC development. The developed linkers, payloads, and other results will be transferred to Lotte Biologics, based on which the company will strengthen the competitiveness of its ADC platform, including SoluFlex Link. Through the collaboration, Lotte Biologics plans to provide an ADC toolbox service that allows customers to select and utilize various technologies according to their needs. This is expected to further strengthen its one-stop platform service, which covers everything from research and development to GMP production for ADC modalities. A Lotte Biologics representative said, “The agreement marks another step forward in establishing a differentiated ADC platform and toolbox. We will continue to strengthen our partnership with both companies to enhance our ADC competitiveness in the global market and provide patients with more innovative treatments.” An Axcelead representative said, “We are very pleased to be able to forge a strategic partnership for the development and advancement of ADC platform technology and services. Based on our proprietary new drug development platform, we will contribute to the development of innovative treatments.” A Kanaph Therapeutics representative added, “Through this collaboration, we will strive to build a diverse toolbox of linkers and payloads that can overcome the limitations of existing ADC drugs and accelerate the development of innovative new drugs.”