- LOGIN
- MemberShip
- 2026-04-30 19:06:55
- Company
- Metabolic disease drug development landscape evolves
- by Son, Hyung Min Jul 01, 2025 06:02am
- Innovative efforts in the development of new drugs for metabolic disorders such as obesity and diabetes are steadily gaining traction in Korea and abroad. The success of standalone GLP-1 therapies has driven the evolution toward multi-hormonal agents, and recently, major global pharmaceutical companies and biotech firms are accelerating new drug development by targeting not only GLP-1, GIP, and glucagon combinations but also new metabolic hormones such as FGF21 and IGF-1. In essence, a multi-target approach that goes beyond simple weight loss to regulate energy metabolism, insulin sensitivity, and liver fat improvement is becoming a reality. Development of multi-agonists starts with Mounjaro... Biomed announces Phase II trial According to industry sources on the 30th, the US pharmaceutical and biotech company Biomed Industries recently announced the results of its Phase II clinical trial for NA-931, an oral quadruple receptor agonist candidate for obesity and metabolic disorders. The results were presented at the American Diabetes Association's annual diabetic conference (ADA 2025) that was held in Chicago from the 21st to the 24th. NA-931 is a small-molecule drug that acts on four receptors: glucagon-like peptide (GLP-1), gastric inhibitory peptide (GIP), glucagon (GCG), and insulin-like growth hormone type 1 (IGF-1). Recently, research has also been conducted on quadruple receptor agonists that utilize additional metabolic-related hormones such as IGF-1, FGF21, and PYY, in addition to GLP-1, GIP, and GCG. FGF21 is a hormone produced in the liver that is involved in fat oxidation, insulin sensitivity, and temperature regulation. It is gaining attention as a next-generation target due to its ability to induce metabolic improvements similar to those observed with intermittent fasting. PYY is an appetite-suppressing hormone derived from the gut that can contribute to enhancing satiety by interacting with GLP-1. The Phase II clinical trial presented at the conference was a 13-week, randomized, double-blind, placebo-controlled, parallel-group study involving 125 adults with obesity (BMI ≥ 30 kg/m²) or overweight (BMI ≥ 27 kg/m²). The primary endpoints were the safety, tolerability, and weight loss efficacy of the investigational drug NA-931. Results showed that the group administered NA-931 at a dose of 150 mg once daily experienced an average weight reduction of 13.8% compared to baseline, with a statistically significant reduction of 12.4 percentage points compared to the placebo group. The incidence of treatment-emergent adverse events was also favorable. Gastrointestinal (GI) adverse events were mostly mild, with nausea and vomiting observed in 7.3% of participants and diarrhea in 6.3%. No muscle mass reduction was reported, and there was no clinically significant difference in the incidence of GI-related adverse events among subjects treated with NA-931 compared with placebo. The researchers explained, “NA-931 appears to induce weight loss while preserving muscle mass and has a lower incidence of side effects than existing drugs. It can be regarded as a viable alternative that can improve the shortcomings of existing GLP-1 class treatments.” Development of quadruple receptor agonists continue… to treat MASH in addition to obesity Clinical studies of quadruple receptor agonists targeting indications other than obesity are also actively underway. Among domestic companies, Wonjin Biotechnology has entered the clinical trial phase for a quadruple receptor agonist. Wonjin Biotechnology received approval in the US last year for the Investigational New Drug (IND) application for its candidate compound ‘OGB21502’ for the treatment of metabolic dysfunction-associated steatohepatitis (MASH) and fibrosis. OGB21502 is an innovative drug candidate that combines GLP-1, glucagon, FGF21, and an IL-1 receptor agonist (IL-1RA), an immune modulator, to simultaneously regulate metabolic disorders and chronic inflammation. OGB21502 induces appetite suppression and energy metabolism activation through GLP-1 and glucagon receptors while promoting lipid metabolism via FGF21 action. Additionally, by blocking IL-1 signaling involved in inflammation, the company expects it to demonstrate a multi-layered therapeutic effect by preventing inflammatory exacerbation of metabolic diseases. According to Wonjin Biotechnology, there is growing evidence that therapies blocking IL-1 signaling are effective in alleviating liver fibrosis. In fact, anakinra, a recombinant IL-1 receptor antagonist, has been shown in multiple studies to inhibit NLRP3 activation and reduce hepatic stellate cell (HSC) activity and blood-based fibrosis markers. In preclinical studies, OGB21502 demonstrated a reduction in lipid markers, including cholesterol and triglycerides, as well as the expression of fibrosis-related markers, compared to semaglutide and FGF21 analogs in obese mouse models. In a mouse model of non-alcoholic fatty liver disease (NAFLD), liver tissue staining analysis revealed that OGB21502 significantly reduced the non-alcoholic fatty liver disease activity score (NAS) and improved steatosis and lobular inflammation.
- Company
- AstraZeneca Korea launches ‘Lung Health Checkbus’ campaign
- by Whang, byung-woo Jul 01, 2025 06:00am
- Launch ceremony for AstraZeneca Korea AstraZeneca Korea announced on the 30th that it held a ceremony for the launch of its ‘Lung Health Checkbus’ campaign at the COEX Square in Seoul on the 27th. The campaign aims to help people detect lung nodules that they are not aware of at an early stage by operating buses equipped with AI-based chest X-ray imaging nationwide. AstraZeneca Korea, which has been making various efforts to create a world where lung cancer is no longer a cause of death, has partnered with the Korea National Tuberculosis Association and medical AI solution company Maihub to operate the ‘Lung Health Checkbus’ nationwide. Lung cancer is the leading cause of cancer-related deaths in Korea, according to 2023 data. Over the 5-year period from 2018 to 2022, the relative survival rate was 79.8% when the cancer was detected at an early stage, but the rate dropped sharply to 12.9% when the cancer had spread to distant parts of the body. However, it has been reported that more than 40% of patients are diagnosed with cancer at an advanced stage of metastasis, emphasizing the importance of regular screening. Low-dose chest CT is an effective screening method that can accurately detect lung cancer and reduce mortality, and AI-equipped chest X-rays are more effective than conventional X-rays in detecting lung nodules. According to a study comparing the lung nodule detection rates of AI-equipped chest X-rays and conventional X-rays at a single institution in Korea, the lung nodule detection rate in the AI group was more than twice that of the non-AI group. Se-Hwan Chon, General Manager of AstraZeneca Korea, said, “Lung cancer can affect anyone, so lung cancer screening is necessary even for non-smokers. In particular, lung cancer has a significantly higher survival rate when detected early, so it is important to detect it early through regular screening.” On the same day, AstraZeneca Korea signed a three-party memorandum of understanding (MOU) with the Korea National Tuberculosis Association and Maihub to ensure the successful operation of this campaign. Many citizens had chest X-rays taken at the lung health check bus set up on-site and received reports analyzed by artificial intelligence (AI) to check their lung health status for themselves. Min-Seok Shin, Chair of KNTA, said, “We find it meaningful that KNTA can expand its social responsibility beyond respiratory diseases to a broader range of diseases through the campaign. We plan to continue supporting customized health management programs to ensure that everyone, including medically vulnerable groups, can easily check their lung health. Hyuck Yang, CEO of Maihub, said, “This campaign is a meaningful example of public and private sectors coming together to create an AI-based chest X-ray examination environment that is easily accessible to everyone, going beyond simply providing technology. Maihub will contribute to the early detection of lung nodules through AI-based image interpretation solutions and strive to build a digital healthcare ecosystem.” Meanwhile, AstraZeneca Korea is a member of the Lung Ambition Alliance (LAA), a global non-profit collaboration organization, and is conducting various lung cancer awareness improvement activities to create a future where lung cancer is no longer a cause of death.
- Company
- New K-drugs for metabolic diseases make international debut
- by Son, Hyung Min Jun 30, 2025 06:07am
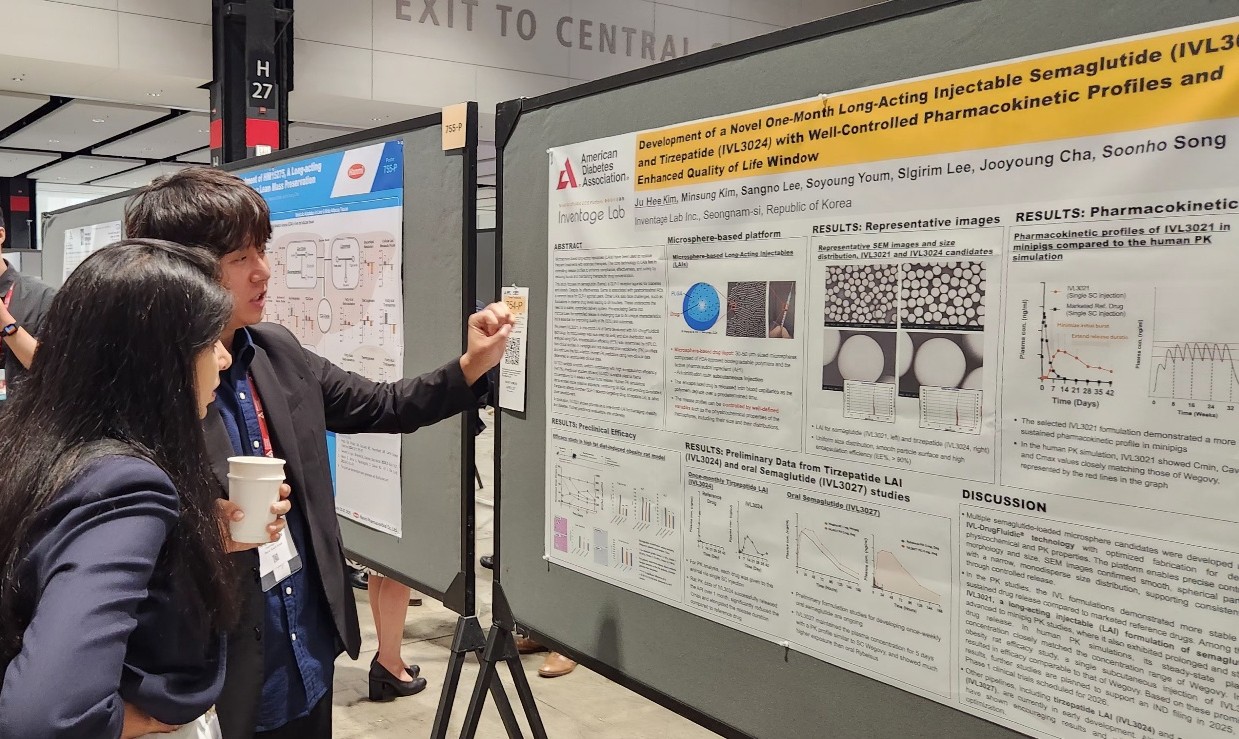
- Major Korean pharmaceutical and biotechnology companies have signaled their full-scale entry into global clinical trials, presenting new drug development results at overseas conferences. The companies presented their achievements in developing new drugs for various metabolic diseases, including obesity, type 2 diabetes, and metabolic dysfunction-associated steatohepatitis (MASH). Although most of the data disclosed is focused on early clinical or preclinical trials, the companies are attempting to diversify their mechanisms of action with triple agonists and oral small molecule formulations. Hanmi reveals triple agonist data... Oral and long-acting injectable formulations are also under development 30According to industry sources, the American Diabetes Association Diabetes Conference was held in Chicago, USA, from the 21st to the 24th. At the conference, various domestic companies, including Hanmi Pharmaceutical, Ildong Pharmaceutical, Dong-A ST, and Inventage Lab, revealed the clinical results of their novel drug candidates. Obesity drugs are rapidly emerging as a global R&D trend. With Novo Nordisk and Lilly's GLP-1-based obesity treatments becoming global blockbuster drugs, latecomers are also intent on developing their versions. Major domestic companies are conducting clinical studies on GLP-1 agonists in various areas, including obesity and MASH with drugs that have different methods of administration or focus on the quality of weight loss effects. Hanmi Pharmaceutical announced the results of its Phase I clinical trials and preclinical data for HM15275, a GLP-1 class triple agonist, and HM17321, a UCN2-based candidate substance, at the conference. First, Hanmi Pharmaceutical announced the results of the Phase I clinical trial for HM15275. HM15275 is a new obesity drug candidate that acts on glucagon-like peptide (GLP-1), gastric inhibitory polypeptide (GIP), and glucagon (GCG). No new drug with this mechanism targeting all three has been commercialized to date. The Phase I clinical trial was conducted on 74 healthy and obese adults. HM15275 was administered subcutaneously once a week for four weeks, followed by an evaluation of the candidate drug’s safety, tolerability, pharmacokinetics, and pharmacodynamics. The results confirmed HM15275’s tolerability and safety. Specifically, the average weight loss rate at day 29 in the highest dose group was 4.8%. At day 43, the maximum weight loss rate was 10.6%. Preclinical data for HM15275 demonstrated greater weight loss efficacy compared to semaglutide (brand name Wegovy) and tirzepatide (Zepbound), which are currently marketed as obesity treatments. In animal models, switching from tirzepatide to HM15275 resulted in additional weight loss effects. Hanmi Pharmaceutical also announced the results of preclinical studies on HM17321, a new drug candidate that simultaneously targets weight loss and muscle gain. HM17321 is a UCN-2 analogue that selectively targets the CRF2 (corticotropin-releasing factor 2) receptor rather than GLP-1 or other incretin receptors. It is being developed as an innovative first-in-class drug that selectively reduces fat while increasing muscle mass. HM17321 demonstrated weight loss effects and improved body composition in both mouse models and non-human primate models. Yunovia, a subsidiary of Ildong Pharmaceutical Group specializing in new drug research and development, is conducting Phase I clinical trials for ID110521156, a GLP-1 receptor agonist class new drug candidate targeting metabolic disorders such as diabetes and obesity. ID110521156 is a low-molecular-weight compound-based drug, and the company aims to develop it as an oral synthetic new drug for diabetes and obesity with distinct advantages such as superior productivity and excellent ease of use over existing representative treatments like peptide injections. Previously, Yunovia confirmed the efficacy of insulin secretion and blood glucose control through preclinical efficacy and toxicity evaluations. It also demonstrated superior safety compared to competing drugs in the same class and confirmed the drug's characteristics in a recently completed Phase 1 single-ascending dose (SAD) trial. According to the study poster presented at the conference, in the single-dose escalation trial, ID110521156 demonstrated good tolerability with fewer gastrointestinal side effects across the entire effective dose range, unlike existing GLP-1 class drugs. Inventage Lab also introduced preclinical data for its 1-month long-acting injectable formulations ‘IVL3021’ and ‘IVL3024’ based on semaglutide and tirzepatide, as well as its oral semaglutide formulation ‘IVL3027.’ After GLP-1 class obesity treatments such as Saxenda, Wegovy, and Zepbound emerged as global blockbuster drugs, the pharmaceutical industry has been actively pursuing formulation changes. The existing drug Saxenda requires a once-daily injection, while Wegovy and Zepbound require weekly injections. Oral formulations or long-acting injectables are expected to gain a competitive edge in terms of convenience of administration if commercialized. According to the company's preliminary preclinical results, IVL3021 showed stable drug release in the blood over a one-month period. Also, the long-acting injectable suppressed initial over-release and maintained stable drug release. IVL3027 demonstrated high bioavailability compared to existing oral formulations and sustained drug release over a one-week period. Poster presentation by Inventage Lab (Source=Inventage Lab). MASH clinical trial results also announced Dong-A ST and its subsidiary MetaVia announced the results of non-clinical studies on DA-1241, which is being developed as a treatment for MASH, and combination therapy that uses efruxifermin, a fibroblast growth factor (FGF21) analog. Metabolic dysfunction-related fatty liver disease was previously referred to as non-alcoholic steatohepatitis (NASH), but overseas academic societies such as the American Association for the Study of Liver Diseases have decided to change the name to metabolic dysfunction-associated steatohepatitis (MASH). To date, Madrigal's rezdiffra is the only new drug for MASH that has cleared regulatory hurdles. The U.S. Food and Drug Administration (FDA) approved rezdiffra in March last year for the treatment of adult patients with non-cirrhotic MASH in combination with diet and exercise. Rezdiffra is a selective thyroid hormone receptor (THR)-β agonist designed to target the core pathophysiological mechanisms of MASH within the liver. The pharmaceutical industry is also developing new drugs for MASH that target not only THR-β but also GLP-1 and FGF21, which influence lipid metabolism. DA-1241 is a synthetic new drug that activates GPR119. Preclinical results have confirmed that DA-1241 improves blood sugar and lipid levels and directly acts on the liver to improve inflammation and fibrosis, making it a promising candidate for MASH treatment. A Phase 2a clinical trial targeting patients with estimated MASH was completed in December last year. Efruxifermin is a recombinant protein designed based on FGF21 (Fibroblast Growth Factor 21), a hormone secreted by the liver. FGF21 is involved in energy consumption and the regulation of glucose and lipid metabolism in the body and is used as a target for the development of treatments for MASH, obesity, diabetes, and other conditions. According to the study results presented at the conference, in the DA-1241+efruxifermin combination therapy group, approximately 94% of subjects showed an improvement of 2 points or more in NAS (Non-Alcoholic Steatohepatitis Activity Score) compared to baseline. Additionally, the DA-1241+efruxifermin group showed a significant reduction in liver fibrosis area compared to the MASH control group that did not receive combination therapy, and in some individuals, a decrease in fibrosis stage was observed compared to pre-treatment levels. Dong-A ST is currently conducting clinical studies on the combination therapy of DA-1241 with semaglutide, a GLP-1 agonist, in addition to a Phase II clinical trial on DA-1241 as monotherapy.
- Company
- Adempas may be prescribed at general hospitals in KOR
- by Eo, Yun-Ho Jun 30, 2025 06:06am
- Adempas, a new treatment for pulmonary arterial hypertension that has emerged after a long wait, is now available for prescription at general hospitals in Korea. According to industry sources, Bayer Korea's Adempas (riociguat) has been approved by the Drug Committee (DC) of tertiary hospitals in Korea, including Samsung Medical Center and Seoul National University Hospital. As it has been listed for reimbursement since this month (June), the number of medical institutions that can prescribe it is expected to continue to increase. Adempas was approved in Korea as an orphan drug in June 2014 and is available in 5 dosage forms. It is indicated for: ▲Improvement of exercise capacity in adult patients with chronic thromboembolic pulmonary hypertension (CTEPH, WHO Group 4) who are unable to undergo surgery or who have persistent or recurrent symptoms after surgery ▲Improvement of exercise capacity adult patients with pulmonary arterial hypertension (WHO Group 1) who are classified as having functional class II or III. In particular, it was known as the first new drug for CTEPH. CTEPH is caused by patients who develop chronic pulmonary embolism, which leads to fibrotic stenosis and occlusion, resulting in pathological vascular remodeling and increased resistance in the pulmonary artery. CTEPH is a chronic disease that causes progressive dyspnea and right heart dysfunction, which weakens the heart. Symptoms include dyspnea, fatigue, chest pain, dizziness, peripheral edema, cough, and hemoptysis, which reduces the patient’s quality of life. Ultimately, it can progress to heart, kidney, and liver failure, which can lead to death. Meanwhile, Adempas is a stimulator of soluble guanylate cyclase (sGC), an enzyme found in the heart and lungs, and its efficacy has been confirmed in two Phase III clinical trials in patients with chronic thromboembolic pulmonary hypertension. Results showed improvement in exercise capacity, which was the primary endpoint, and good tolerability. No unexpected adverse reactions were reported. In the CHEST-1 study, when comparing the 6-minute walking test (6MWT) results after 16 weeks with the baseline, results showed that the group of patients who received riociguat showed statistically significant improvement compared to the group of patients who received placebo. In the PATENT-1 study, the change in the 6MWT score after 12 weeks of treatment, showed statistically significant improvement in the riociguat arm compared to placebo, meeting the primary endpoint.
- Company
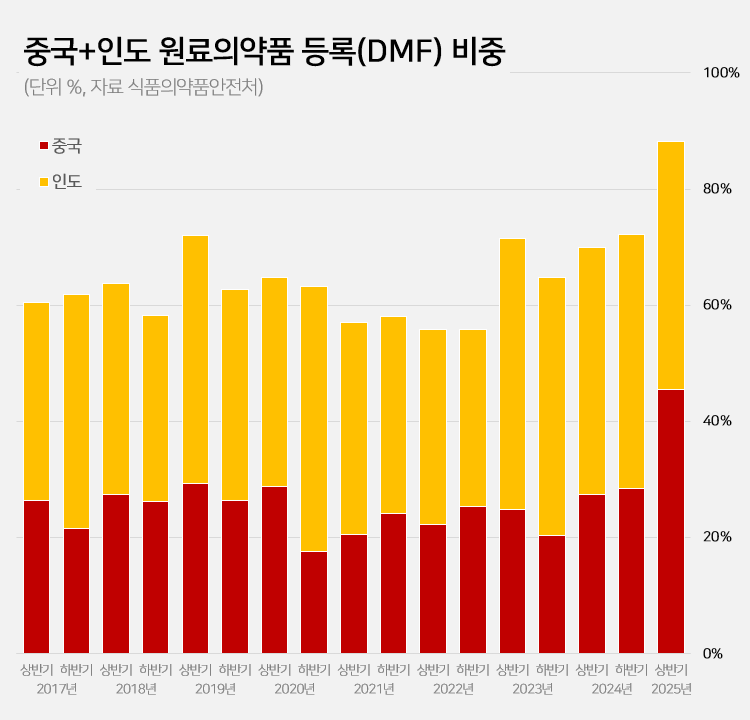
- 88% registered APIs imported from China or India
- by Kim, Jin-Gu Jun 27, 2025 06:04am
- Amid a surge in the number of drug master file registrations in the first half of this year, the share of raw materials from China and India rose to 88.2%. This is a sharp increase compared to the average of 62.1% share the two countries had during the past 5 years. This is attributed to the large number of previously delayed raw material drug registrations that had been made upon the relaxation of DMF regulations, as well as the domestic pharmaceutical and bio industry's increased use of raw materials from China and India to reduce costs. 606 DMF registration from China and India source materials made in the first half of this year... accounts for 88%, which is the highest-ever share According to the Ministry of Food and Drug Safety on the 26th, 687 DMF registrations were made in the first half of this year. Among them, 313 raw materials were from China and 293 were from India. The two countries combined accounted for 606 cases or 88.2% of the total DMF registrations. This is the highest proportion ever recorded for a half-year period. Until last year, the proportion of Chinese and Indian raw materials had never exceeded 75%. The average proportion of Chinese and Indian DMF imports over the past 5 years was 62.1%, which is more than 26 percentage points higher than in the first half of this year. The proportion of Chinese and Indian imports in the DMF has increased rapidly over the past 3 years. After steadily declining since the first half of 2019, the share of Chinese and Indian DMF rose to 55.8% in the first half of 2022 and then began to increase. It reached 69.9% in the first half of last year and 72.2% in the second half. In the first half of this year, it soared to nearly 90%. Pharmaceutical industry's dependence on Chinese and Indian raw materials deepens amid cost pressures The rapid increase in Chinese and Indian DMF is attributed to cost reduction pressures in the pharmaceutical and bio industry. China and India are representative “low-cost mass production bases” in the global raw material drug market. Following the global economic downturn after the pandemic, the pharmaceutical and bio industry in general faced a decline in profitability. As a result, attempts to reduce costs were made, which led to an increase in the use of Chinese and Indian DMF. An industry insider explained, “Domestic pharmaceutical and biotechnology companies are feeling a significant burden from manufacturing costs due to high exchange rates, rising labor costs, and declining profitability. Chinese and Indian raw materials are sometimes almost half the price of domestically produced materials, leading to increased use of imported materials.” Additionally, the relaxation of DMF regulations has further increased the use of Chinese and Indian raw materials. The government eased DMF requirements earlier this year by replacing on-site GMP inspections with the submission of GMP certificates and reducing the administrative processing period from 120 days to 20 days. As a result, imported raw materials for which registration had been delayed were registered en masse. In particular, it is analyzed that the abolition of on-site inspections has led to a significant increase in Chinese and Indian raw materials. In the past, inspections in these two countries were physically difficult, and administrative procedures complex, often causing delays in registration. This year, however, registration became possible with only a GMP certificate, significantly lowering barriers, and leading to a significant increase in the registration of Chinese and Indian raw materials. Domestic raw material share in DMF only 5%, raising concerns about increased dependence on Chinese and Indian products On the other hand, the share of domestically produced raw materials registered in the DMF has decreased significantly. In the first half of this year, the share of domestically produced raw materials registered was only 4.9% (34 cases). This is less than half of the 12.6% recorded in the second half of last year. The share of DMF registrations of raw materials from Europe and Asia also decreased sharply. The share of European raw materials decreased by more than 10 percentage points from 14.5% in the first half of last year to 4.4% in the first half of this year. The number also decreased from 37 to 30. The share of DMF registrations of raw materials from Asian countries other than China and India also decreased from 3.9% to 1.5%. Concerns have been raised that the dependence on raw materials from China and India may become excessively high. If this trend intensifies, it could pose a threat to the stability of domestic drug supply. In fact, during the early stages of the COVID-19 pandemic, export restrictions imposed by China and India directly impacted domestic drug production. A pharmaceutical industry insider stated, “If raw material production becomes overly concentrated in specific countries, it becomes vulnerable to external factors such as export restrictions, logistics disruptions, and sharp exchange rate fluctuations. In the long term, policies are needed to strengthen domestic raw material production capabilities and provide various incentives for the use of domestically produced raw materials.”
- Company
- Tevimbra adds esophageal, gastric, lung cancer indications
- by Whang, byung-woo Jun 27, 2025 06:02am
- Pic of TevimbraBeiGene Korea (Name to be changed to BeOne Medicine Korea) announced that its immuno-oncology drug Tevimbra (tislelizumab) has been approved by the Ministry of Food and Drug Safety for additional indications for esophageal cancer, gastric cancer, and non-small cell lung cancer. With the additional approval, Tembriva can now be used as a first- or second-line treatment for a total of 5 indications across 3 solid tumor types. The approved cancer types are: esophageal squamous cell carcinoma (ESCC), gastric or gastroesophageal junction adenocarcinoma (G/GEJ), and non-small cell lung cancer (NSCLC). Tevimbra demonstrated efficacy and safety for the indications in the RATIONALE clinical trial series (RATIONALE-303, 304, 305, 306, 307), which served as the basis for this approval. In particular, the drugs’ clinical benefits were observed in the overall patient population for esophageal squamous cell carcinoma and gastric or gastroesophageal junction adenocarcinoma and showed consistent results in pre-specified subgroups based on PD-L1 expression levels. Such benefits of Tevimbra were also reflected in global treatment guidelines and is recommended at a high level by the National Comprehensive Cancer Network (NCCN) and the European Society for Medical Oncology (ESMO). Tevimbra employs a dual mechanism of action that effectively blocks PD-L1 while minimizing binding to Fc-gamma receptors (FcγR), thereby inducing potent antitumor responses through a mechanism distinct from that of existing immuno-oncology agents. Also, the drug demonstrated superior PD-1/PD-L1 blocking efficacy (>99%) compared to other immunotherapy agents of the same class, and according to the company, it has a higher binding affinity and a half-life 30-80 times longer than existing drugs, suggesting a more sustained therapeutic effect. In addition, by minimizing binding to the Fc gamma (Fcγ) receptor of the antibody, the drug enhanced the sustainability and stability of the immune response. Ji-Hye Yang, General Manager of BeOne Medicines Korea, explained, “Based on its differentiated mechanism and long-term clinical data, Tevimbra is gaining attention as a new standard of care that surpasses the limitations of existing immunotherapy drugs by offering treatment performance comparable to global benchmarks, as well as treatment sustainability and financial predictability.” Yang added, “We are particularly pleased to offer new treatment opportunities for underserved patients. in the first-line setting for esophageal squamous cell carcinoma, as Tevimbra is the only approved immunotherapy in Korea that can be used regardless of PD-L1 expression levels.” Meanwhile, BeiGene changed its corporate name to BeOne Medicines under the vision of “Overcoming Cancer Together” and is accelerating the expansion of its next-generation anti-cancer portfolio, including ADC (antibody-drug conjugates) and protein degraders, with Switzerland as its global hub. The domestic subsidiary will also complete its rebranding as ‘BeOne Medicines Korea’ by June 30 and plans to accelerate its growth in the solid tumor and blood cancer treatment markets based on its 2 products and 11 indications.
- Company
- KPTA ‘KOR-CHN-JPN supply cooperation to bring $12B effect'
- by Kim, Jin-Gu Jun 27, 2025 06:02am
- The Korea Pharmaceutical Traders Association (KPTA), China Chamber of Commerce for Import & Export of Medicines & Health Products (CCMPHIE), and Japan Pharmaceutical Traders Association (JPTA) announced on June 25 that they signed a memorandum of understanding (MOU) for the stabilization of the pharmaceutical supply chain at the Korea Pavilion in the CPHI & PMEC CHINA exhibition hall in Shanghai, China. The signing ceremony was attended by Hyung-seon Ryu, Chairman of the KPTA; Zhou Hui, President of CCCMHPIE; Ichiro Fujikawa, President of JPTA; Young-soo Jeong, Director of the KOTRA Shanghai Trade Office; and over 20 representatives and officials from each country. The MOU was signed to improve the global pharmaceutical supply chain, which has become unstable following the COVID-19 pandemic, and to enhance the three countries' capability to respond to health crises through cooperation. The three associations play leading roles in the pharmaceutical trade and distribution sectors. Through the MOU, the parties agreed to collaborate on ▲ the export, import, development, and supply of essential and active pharmaceutical ingredients; ▲ exchange of pharmaceutical research personnel, technology, and information; and ▲ joint hosting of seminars, academic conferences, and workshops to promote pharmaceutical trade. They plan to concretize the outcomes of the MOU through the implementation of practical joint projects. The KPTA stated that if supply chain stabilization is realized through this agreement, it will bring an annual economic effect of approximately USD 12 billion through ▲the reduction of raw material inventory costs ▲reduction of procurement costs through joint purchasing ▲reduction of health crisis response costs ▲trade creation effects from the activation of pharmaceutical trade among the three countries ▲trade diversion effects from replacing overseas pharmaceutical imports with intra-regional trade. Also, based on this agreement, it is anticipated that a three-country contract manufacturing (CDMO) model may be forged, enabling Korean companies to produce pharmaceuticals at competitive prices in China, register them in Japan, and enter the European market. Cases where domestic pharmaceutical companies experienced disruptions in imported raw material supplies also also expected to decrease, as the partnership will allow Korean companies to secure stable supply through emergency supply contracts with Chinese and Japanese companies. Hyung-seon Ryu, Chairman of the KPTA, stated, “Korea has strengths in producing high-quality drugs, Japan in precision manufacturing technology and rare drug raw material technology, and China in large-scale production and supply capabilities. This MOU will greatly contribute to the stability of the entire Northeast Asian supply chain and the enhancement of global competitiveness. We ask Korean companies to actively participate in various areas, including follow-up technical collaboration, research and development, and contract manufacturing.” Zhou Hui, Chairman of CCCMHPIE, said, “This agreement is the result of close collaboration between the 3 countries and will contribute not only to the stable supply of essential and raw pharmaceuticals but also to the creation of new business opportunities. It will serve as a leading model for cooperation in the East Asian pharmaceutical industry.” Ichiro Fujikawa, President of the JPTA, commented, “Korea and China are Japan’s core partners for raw materials and finished pharmaceutical products. This agreement will help alleviate the shortage of pharmaceutical supplies in Northeast Asia and positively impact the growth and development of each country’s pharmaceutical industry.”
- Company
- ‘Wegovy, a game-changer for high-risk obesity patients’
- by Whang, byung-woo Jun 26, 2025 06:08am
- Obesity is a cause of various metabolic syndromes and a major risk factor for cardiovascular disease. In fact, approximately 80% of patients hospitalized for cardiovascular disease are obese, and studies have shown that the risk of cardiovascular events in obese patients is up to twice as high as in those of normal weight. Over the past 20 years, the mortality rate from obesity-related cardiovascular diseases has increased significantly, with approximately two-thirds of obesity-related deaths attributed to cardiovascular diseases. Recently, semaglutide (brand name: Wegovy), a GLP-1 receptor agonist, has opened a new treatment paradigm in obesity treatment, demonstrating efficacy in reducing the risk of major cardiovascular events in high-risk obese patients. Kim Kyung-hee, professor of cardiology at Incheon Sejong Hospital (Director of the Heart Transplant Center), met with Dailypharm and emphasized the importance of obesity treatment for the prevention of cardiovascular disease. Obesity increases the risk of early onset of cardiovascular disease... “The number of young patients is also on the rise” Obesity is a precursor to various metabolic diseases, and inflammatory substances secreted from visceral fat reduce blood vessel elasticity. According to Professor Kim, these changes lead to hypertension, diabetes, and hyperlipidemia, which in turn greatly increase the risk of early onset of coronary artery disease and heart failure even in younger age groups. Kyung-hee Kim, Professor of Cardiology, Incheon Sejong Hospital (Director of the Heart Transplant Center) Professor Kim said, “Obesity can be a cause of all cardiovascular diseases, and there is a recent trend of an increase in patients with high blood pressure or symptoms of heart failure from a young age. Recently, the prognosis of severely obese patients is generally worse than that of the general population, but even in lean individuals, and those with sufficient muscle mass tend to have a better prognosis.” Kim further explained, “In cases of cardiovascular diseases such as angina or heart failure, there is a tendency for weight loss and reduced muscle mass due to decreased appetite and nutrient intake. Therefore, obesity typically occurs first, followed by cardiovascular diseases in most cases.” In other words, obesity often acts as a precursor to cardiovascular disease. In this regard, semaglutide is regarded a game changer in the fields of obesity treatment and cardiovascular disease prevention. Semaglutide was approved by the Ministry of Food and Drug Safety in April 2024 as an anti-obesity treatment for patients with a BMI of 27 kg/m² or higher (with comorbidities) or 30 kg/m² or higher, and in July of the same year, it was additionally approved for reducing the risk of cardiovascular events in overweight and obese adult patients with confirmed cardiovascular disease. Professor Kim emphasized the clinical value of semaglutide not merely as a weight-loss aid but as a preventive therapy for cardiovascular disease. In particular, KIM highlighted findings from the SELECT trial, a pivotal clinical study on semaglutide, where the drug demonstrated an additional 20% reduction in the risk of major adverse cardiovascular events (MACE) when added to standard care in patients already receiving conventional treatments. Professor Kim explained, “In the SELECT study, approximately 90% of participants were already receiving standard treatment, but when they added semaglutide, an additional 20% reduction in MACE risk was observed. This result demonstrates that semaglutide can make a substantial contribution to improving outcomes in high-risk patient populations where existing treatments have limitations.” According to the detailed results of the SELECT trial, over an average follow-up period of approximately 3.3 years, the semaglutide 2.4 mg group showed a statistically significant 20% reduction in the risk of cardiovascular death, nonfatal myocardial infarction, or stroke compared to the placebo group. Professor Kim added, “Semaglutide regulates the appetite center in the brain, delays gastric emptying to induce weight loss, and further improves cardiovascular risk factors through its anti-inflammatory effects. While weight loss may also play a role, we believe that the drug’s anti-inflammatory effect plays a very significant role in cardiovascular health.” In particular, Professor Kim emphasized that semaglutide is not simply a weight loss drug, but a scientifically proven cardiovascular treatment option. He said, “Semaglutide is a must-use drug for patients who are severely obese or have a BMI of 27 kg/m² or higher with cardiovascular disease.” “Limitations remain on its reimbursement... Selective reimbursement support needed for high-risk obese patients” Although semaglutide has emerged as an important drug that should be considered as part of a standard treatment for obese patients at high risk of cardiovascular disease, there are currently practical limitations to its access in Korea. This is because it is not yet covered by insurance in Korea. Professor Kim said, “Drugs such as semaglutide carry a certain risk of misuse, so caution should be exercised when expanding reimbursement to all patient groups. However, I believe it is desirable to allow reimbursement through strict criteria and screening procedures for high-risk groups, such as patients with severe obesity and cardiovascular complications, for whom clear therapeutic effects can be expected.” In fact, limiting reimbursement to groups with a clear clinical need for reimbursement, such as patients with a body mass index (BMI) of 27 kg/m² or higher and obesity-related complications or cardiovascular disease, may be a realistic alternative. Professor Kim also predicted that discussions on whether to continue reimbursement will be necessary when semaglutide significantly improves a patient’s BMI. He said, "If patients with a BMI of 30 kg/m² or higher are administered semaglutide and their BMI falls below 26 kg/m² due to weight loss, it may be possible to consider limiting the reimbursement period to the initial 4-6 months. However, since there is currently insufficient long-term data in Korea and some patients experience weight regain after 6 months of treatment based on clinical experience, further review of long-term management strategies is necessary.” To address current issues surrounding reimbursement coverage and costs, Professor Kim is currently conducting an economic evaluation study. “We are analyzing how much the number of medications taken by patients can be reduced when they lose weight after 6 months or a year through bariatric surgery or semaglutide treatment, and we expect this to be significant in terms of establishing future treatment strategies and fiscal efficiency.” Ultimately, Professor Kim believes that obesity treatment should not end with medication alone but must include comprehensive management to help patients fundamentally improve their lifestyle habits. Professor Kim emphasized, “Patients with a BMI of 30 kg/m² or higher often find exercise difficult, so they should be actively educated to combine medication with walking exercises, maintain a high-quality diet, reduce carbohydrate intake, and abstain from alcohol and smoking.” “Early intervention in obesity treatment is necessary to maximize preventive effects” Professional counseling and lifestyle education support are essential to increase the effectiveness of obesity treatment. However, the reality of how difficult it is to provide sufficient counseling in the current outpatient setting is also pointed out as an issue. Professor Kim said, “It is difficult to check blood pressure, assess the patient's condition, perform a physical examination, and explain lifestyle correction measures within the 5 minutes of consultation time allocated per patient. At least 7-10 minutes are necessary for proper treatment.” In this regard, Professor Kim proposed the establishment of a lifestyle education program and a new fee schedule to hire dedicated personnel to overcome such limitations. He explained, “Under the current system, separate reimbursement rates for education provided by specialized nurses need to be introduced, and institutional and financial support would also be needed to manage these personnel. Overall, I believe that establishing an environment where lifestyle education can be systematically implemented is essential to improving the quality of care for obese patients. In particular, Professor Kim emphasized, “Patients with hypertension are at high risk of developing heart failure over time. Starting medication early and educating young obese patients with hypertension on proper lifestyle habits can prevent serious complications and repeated hospitalizations.” In other words, Kim believes preventive treatment and lifestyle improvement efforts targeting young obese patients are expected to lead to macro-level medical cost savings in the future. Finally, Professor Kim emphasized that “Patients should always be treated with scientifically validated medications first. Drug treatment alone is not sufficient and must be accompanied by lifestyle modifications and proper dietary management.”
- Company
- The 2nd KRAS-targeted cancer drug 'Krazati' expected
- by Eo, Yun-Ho Jun 26, 2025 06:08am
- Product photo of Krazati The second KRAS inhibitor is expected to be commercialized in South Korea. Bristol Myers Squibb (BMS) Korea recently submitted a marketing authorization application to the Ministry of Food and Drug Safety (MFDS) for its anti-cancer drug, Krazati (adagrasib). Krazati was also designated as an orphan drug in January. It is indicated for the treatment of 'locally advanced or metastatic non-small cell lung cancer (NSCLC) with a KRAS G12C mutation, previously treated with at least one prior therapy.' Krazati received accelerated approval from the U.S. FDA in December 2022. It is the second KRAS inhibitor receiving approval, following Amgen's 'Lumakras (sotorasib),' which was approved in 2021. The development of KRAS-targeted anti-cancer drugs has come approximately 40 years after the initial discovery of the oncogene. Amgen and BMS are engaged in fierce competition to dominate this new market. Lumakras and Krazati share many similarities, including their target mutation and indications. Both target the KRAS G12C mutation, and their initial approved indication is NSCLC. Both companies are also conducting clinical trials in combination with compounds with different mechanisms of action developed in-house or through collaborations. Meanwhile, Krazati's initial approval was based on cohorts from the KRYSTAL-1 study who are eligible for Phase 2 trials. Last year, the primary analysis results of the confirmatory Phase 3 study were disclosed. This study compared Krazati with docetaxel in 301 patients with previously treated KRAS G12C-mutated locally advanced or metastatic NSCLC. These patients had previously received platinum-based chemotherapy and anti-PD-1/PD-L1 immunotherapy. They were randomized 1:1 to either the Krazati treatment group or the docetaxel treatment group. The primary endpoint was progression-free survival (PFS) as assessed by blinded independent central review (BICR). After 9.4 months of follow-up, the median PFS for Krazati was 5.49 months, which met the primary endpoint by reducing the risk of disease progression or death by 42% compared to 3.84 months for the docetaxel group.
- Company
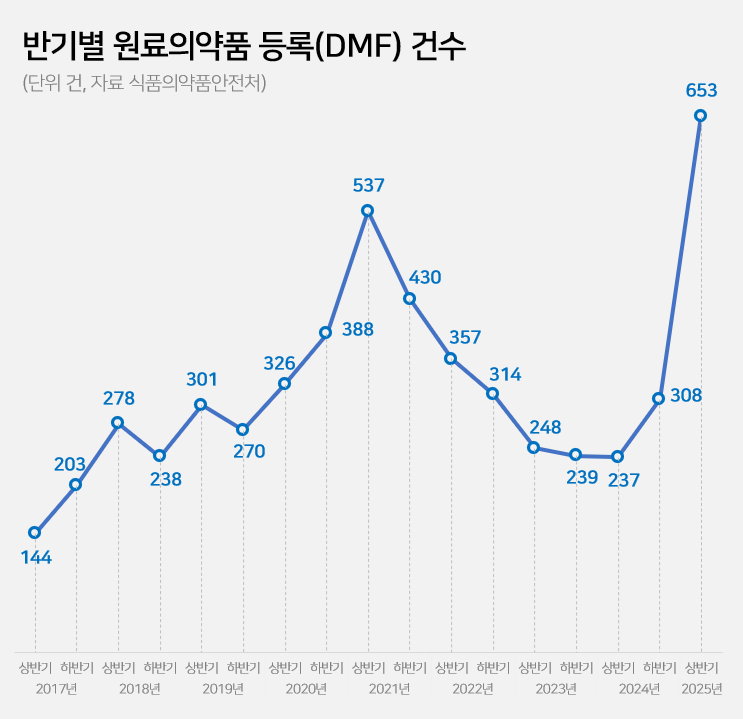
- Drug Master File for API 237→653…'easing of regulation'
- by Kim, Jin-Gu Jun 26, 2025 06:07am
- The number of Drug Master File, DMF, cases in the first half of this year surged by 2.8 times compared to the same period last year. This is the highest for a half-year period. Analysis suggests that this is due to the easing of Active Pharmaceutical Ingredient (API) registration requirements at the beginning of the year. The government had previously eased regulations to allow GMP evaluation to be replaced by GMP certificates for API registration starting this year. 'Record-High' DMF Registrations of 653 Cases in the first half of 2025...Up 2.8x YoY According to the Ministry of Food and Drug Safety (MFDS) on June 26, the number of API registrations by Korean pharmaceutical and biotech companies in the first half of this year reached 653 cases. Compared to 256 cases in the first half of last year, this year's marks a 2.8-fold increase in one year. It has already surpassed the total number of API registrations for the entire 2024 (545 cases). In terms of half-yearly API registrations, it has exceeded the 537 cases in the first half of 2021, reaching an all-time high. Half Yearly Cases of Drug Master Files (DMFs) for API (unit: number of cases, source: MFDS) The robust increase in DMF cases is attributed to the government's easing of regulations. Earlier this year, MFDS reformed the DMF system to replace on-site inspections with GMP certificates. Previously, DMF applications required on-site inspections, along with manufacturing facility data, production country manufacturing certificates, and 11 types of GMP documents. From this year, on-site inspections have been abolished. Additionally, documents can now be replaced by GMP certificates issued by the government agency of the production country or a PIC/S member country. The administrative processing period has also been shortened from 120 days to 20 days. An MFDS official explained, "Previously, to register API, the applying company had to undergo a GMP on-site inspection, but from this year, it can be substituted with a certificate," and added, "It seems that nearly 1,000 piled-up DMF applications were processed in large numbers this year, leading to a surge in DMF cases." Concerns over API quality verification...MFDS states "On-site inspections maintained for high-risk Items" Regarding this deregulation, some in the pharmaceutical industry express concerns that API quality control could become lax. Critics argue that, with registration now possible solely based on GMP certificates, it will be challenging to identify quality issues beforehand through document-based evaluations. This easing of regulation is a complete reversal from MFDS's previous stance. Since the introduction of the DMF system in 2002, MFDS has consistently strengthened quality control. In 2014, GMP evaluation standards were reinforced with PIC/S membership. At this time, 11 types of GMP documents and on-site inspection standards were introduced. In 2019, DMF registration became mandatory not only for new items but also for previously approved items. In 2021, the on-site inspection system was further strength with a focussing on high-risk items. During a briefing last year, MFDS explained that they adjusted the evaluation system in response to the administrative bottleneck caused by a surge in DMF applications, which also delayed the review of finished pharmaceutical products (FPP). Overall, MFDS's policy is to shift its GMP approach to be 'FPP-centric.' Regarding concerns about API quality, MFDS states that on-site inspections are exceptionally maintained for high-risk items, and GMP certificate requirements have been strictly set in line with international standards. Indeed, for high-risk items such as biopharmaceuticals and sterile APIs, on-site inspections and submission of evaluation data are still required. Furthermore, on-site inspections are maintained as before for drug approval and suitability judgments. They also plan to introduce the concept of a 'Site Master File' to understand the quality management system of manufacturing sites comprehensively. Up and down of cases based on regulatory changes...Decline after 2021 peak→ rebounding This Year The number of DMF registrations by Korean pharmaceutical and bio-companies has fluctuated significantly each year due to system changes and policy factors. Over the past eight years, DMF cases have exhibited fluctuating trends: ▲347 in 2017 ▲516 in 2018 ▲571 in 2019 ▲714 in 2020 ▲967 in 2021 ▲671 in 2022 ▲487 in 2023 ▲545 in 2024. With 653 cases in the first half of this year alone, there is a possibility of exceeding 1,000 cases by year-end. The surge in DMF cases in 2021 coincided with the implementation of a policy that made API registration mandatory, even for previously approved items. In 2019, MFDS expanded the scope of DMF to include 'previously approved items' from the original 'newly approved items.' It is analyzed that commercial drugs were required to complete registration by 2021, leading to a concentrated influx of DMF applications. A reform of the drug pricing system around the same time also influenced the increase in DMFs. In July 2019, the government introduced a 'step-wise drug pricing system.' Generics that did not meet the highest price criteria could maintain their previous drug prices if they submitted data from bioequivalence tests and demonstrated the use of registered APIs. This led to a surge in DMF applications from pharmaceutical and biotech companies seeking to maintain drug prices. After 2023, the situation changed. In February 2023, the submission of DMF documents for drug price maintenance concluded. With most DMFs for previously approved items also finalized, the number of registrations began to decline. Indeed, DMFs, which had reached 967 cases in 2021, nearly halved to 487 cases by 2023. However, with the lowering of DMF hurdles this year, the number of registrations is showing a rebounding trend.