- LOGIN
- MemberShip
- 2026-05-02 18:21:30
- Policy
- Drug crime regulation,
- by Lee, Jeong-Hwan Sep 05, 2023 05:40am
- It is expected that a bill that would impose an obligation on doctors and pharmacists who prescribe and dispense drugs with a risk of misuse, such as narcotics, to check medication history through the Drug Safety Information System (DUR) will quickly pass the National Assembly. This is because a large number of drug-related crimes have occurred, such as the so-called 'Rolls Royce Man on Drug Suspicion' incident, and social consensus on the need for stronger regulations has grown, and the Yoon Seok-yeol government has also announced mandatory DUR confirmation of medication history as a measure to strengthen surveillance of medical drugs. On the 4th, Jeon Hye-sook, an official from the Democratic Party of Korea's office, explained, "We have secured a consensus not only on the government but also on the ruling and opposition parties on the need to make it mandatory to check the medication history of patients who prescribed and dispensed narcotics through DUR." The amendment to the Medical Service Act and the Pharmaceutical Affairs Act, proposed this time by Rep. Jeon Hye-sook, are a method of establishing new regulations on medicine and pharmacists in the 'confirmation of drug information' provision commonly stipulated in both laws. Specifically, the 'confirmation of drug information' clause stipulated in Article 18-2 of the Medical Service Act and Article 23-2 of the Pharmaceutical Affairs Act will be revised. Rep. Jeon Hye-sook's bill stipulates that in order to ensure the safe use of medicines, the Minister of Health and Welfare or the Minister of Food and Drug Safety must check whether the same ingredients have been administered in the past when prescribing, directly dispensing, or dispensing medicines that are recognized as having a risk of abuse, such as narcotics. In addition, DUR was proposed as a method for doctors, dentists, and pharmacists to check a patient's medication history for the same narcotic ingredient, and a fine of up to 1 million won could be imposed if the verification obligation was not followed. The Yoon Seok-yeol government declared war on drugs last April and announced comprehensive government-wide measures. At that time, as the Gangnam Academy District drug drink incident and famous celebrities' drug use crimes such as propofol and methamphetamine continued to occur, the Office for Government Policy Coordination announced that it would be mandatory to check prescription drug history using DUR, starting with drugs with a high risk of abuse such as fentanyl. With administration consistent with the purpose of the bill proposed by former lawmakers, an environment has been created in which the speed of processing can be accelerated when the bill is later reviewed by the National Assembly. There is a possibility that the medical community will oppose legislation mandating drug use history DUR. In the past, medical and hospital groups strongly opposed the amendments to the Medical Service and Pharmaceutical Affairs Act in the 20th National Assembly, which mandated the use of DUR when prescribing and dispensing medicines and imposed fines on violators. At the time, the medical and hospital communities considered the fact that punitive regulations were created without support measures such as fees or compensation for medical institutions, that doctors' judgments regarding drug prescriptions and dispensing could be excessively restricted, and that there were limits to clinical usefulness. rebelled against. The Ministry of Health and Welfare, the responsible ministry, and pharmacists' organizations supported the bill to strengthen drug safety by increasing DUR utilization. Rep. Jeon Hye-sook, who proposed the bill, plans to speed up the review of the bill by putting legislative feasibility first in order to prevent drug crimes from becoming increasingly rampant. She plans to push for legislation mandating the use of DUR when prescribing and dispensing non-reimbursed high-risk drugs and narcotic psychotropic drugs, following legislation mandating DUR confirmation of narcotic medication history.
- Policy
- Why many choline alfoscerates accepted price cuts
- by Lee, Tak-Sun Sep 05, 2023 05:39am
- Pic Chongkundang Gliatirin Soft Cap (left) and Gliatamin(right), leading choline alfoscerate drugs Companies are reluctant to self-manufacture their brain function enhancer choline alfoscerates despite price cuts. Only 20% of the companies decided to protect their drug price by switching to self-manufacture, and the prices of the others that opted to continue manufacturing through contract manufacture organizations (CMOs) were cut. This fact was evident in the insurance price ceiling reevaluation results announced by the Ministry of Health and Welfare on the 1st. Among the 51 choline alfoscerate 0.4g capsule formulations, 40 of them received price cuts of up to 15% of the highest price (KRW 445/capsule). 11 products have maintained the original drug price, and 10 of them have maintained their drug prices by conducting self-bioequivalence tests of their self-manufactured (imported) products. The products that maintained their price are ▲Chong Kun Dang Pharmaceutical’s ' Chongkundang Gliatirin Soft Cap.’▲Daewoong Bio’s 'Gliatamin Soft Cap. ' ▲Korea United Pharm 'Glicetil Soft Cap.' ▲GuJu Pharma ' Cereforin Soft Cap.' ▲Cosmax Pharma 'Choline Max Soft Cap. 400mg' ▲Mothers Pharm 'Memoem Soft Cap.' ▲Korea Prime Pharm 'Gria Soft Cap.' ▲Hanmi Pharm’s ' Cholinate Soft Cap. 400mg' ▲Su-Heung’s 'Alfogreen Soft Cap.' ▲Kukje Pharm ' Cholencina Soft Cap. ' A majority of products gave up protecting their drug prices with self-bioequivalence tests due to high investment risk. In 2020, the government made the decision to reimburse choline alfoscerate preparations only for dementia and apply selective reimbursement and cover only 80% of the cost for its main indication, mild cognitive impairment. Also, starting in 2021, clinical re-evaluation is underway to verify the substance’s efficacy for dementia and mild cognitive impairment. If the clinical trials show bad results, their indication may be reduced or deleted. This is why the analysis is that companies have been passive in maintaining their drug price by conducting self-bioequivalence tests. Even products with annual outpatient prescriptions exceeding KRW 10 billion did not defend their drug prices. Based on UBIST, the capsule formulations that posted sales of over KRW 10 billion, such as Daewon Pharmaceutical's Alfocholine (KRW 21 billion), Arlico Pharmaceutical's Choliatin (KRW 18 billion), Yuhan Corp’s Alfoatilin (KRW 17.5 billion), Jeil Pharmaceutical's Glitin (KRW 16 billion), and HLB Pharmaceutical's Glitia (KRW 12.9 billion) and Hutex’s Silvercerin Tab. (KRW 11.7 billion), maintained consignment production, and the insurance price ceiling of the drugs fell to KRW 445. Some have also pointed out that companies decided to not defend drug prices to pursue a low-price strategy as competition is intensifying. Dongkoo Bio Pharm’s Glifos Soft Cap. already lowered its price to KRW 445 in September last year. Nevertheless, UBIST showed Glifos’s sales grew 36% from the previous year to KRW 14.8 billion in outpatient prescriptions last year. An industry official said, “Choline alfoscerate has significant product risk due to clinical reevaluations. Also, we did not take any action to defend drug price because we are pursuing a low-price strategy."
- Policy
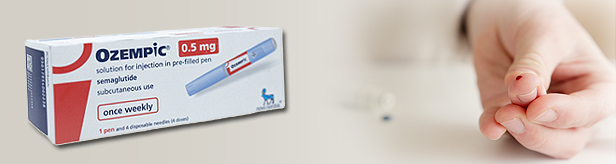
- New GLP-1 RA stops reimb discussions in KOR
- by Lee, Tak-Sun Sep 04, 2023 05:04am
- Novo Nordisk The debut of a new GLP-1 analog used as an adjuvant to dietary and exercise therapy in diabetes patients has been postponed in Korea. This comes as a disappointment as only one single-agent drug is currently reimbursed in Korea, and the high global demand for the drugs has rendered domestic supply short. According to the Health Insurance Review and Assessment Service and the industry on the 1st, discussions for the reimbursement of Novo Nordisk’s single agent GLP-1 receptor agonist ‘Ozempic Pre-Filled Pen' have come to a stop. This drug was recognized as appropriate for reimbursement by the Drug Reimbursement Evaluation Committee in February under the condition that the company accepts a price below the evaluated amount. Afterward, the pharmaceutical company accepted the evaluated amount and began drug pricing negotiations with the National Health Insurance Service. However, the company withdrew its The speculation is that the excess demand in the global market rendered it difficult for the company to supply the drug to Korea as well, for at least one year. Currently, the only single-agent GLP-1 receptor agonist available with reimbursement in Korea is Dulaglutide (Trulicity). Exenatide (Bydureon, AstraZeneca), which had received new reimbursement standards, had withdrawn its approval in September 2021. Another same-class drug, LyxumiaR had also withdrawn its approval in 2020. All of the companies gave up selling their drugs in the Korean market after losing to the long-acting once-weekly Trulicity. Since its release in May 2016, Trulicity has continued to its sole lead in the domestic market. Based on IQVIA, Trulicity recorded sales of KRW 59.4 billion last year. As another long-acting once-weekly treatment, Ozempic was expected to serve as a rival to Trulicity. In the global market, Ozempic’s sales have already surpassed that of Trulicity. However, its unstable supply due to global demand hindered its domestic launch. Ozempic is listed in the US, Japan, Italy, France, Switzerland, and the UK among the A7 countries. The Korean Diabetes Association and the Korean Endocrine Society had recommended that it would be appropriate to reimburse Ozempic as a long-acting GLP-1 receptor agonist same as Trulicity, as a comparative study between the two same-class drugs has also been conducted. GLP-1 is a hormone that regulates glucose and appetite and has various effects on the cardiovascular system. Its effect on glucose and appetite is achieved through GLP-1 receptors in the pancreas and brain, and GLP-1 analog drugs selectively bind and activate these GLP-1 receptors.
- Policy
- Kanoa, the upper limit price was reduced further by PVA
- by Lee, Tak-Sun Sep 01, 2023 05:44am
- Ankook Pharm Daelim-dong office building It was found that the upper limit of the choline alposerate formulation Kanoa Soft Cap sold by Ahnook Pharm has been reduced more with PVA (use-drug linkage agent) than the Kanoa Soft Capre-evaluation of the upper limit. There are a total of 18 items that have been re-evaluated and reduced by both the PVA this time, and among them, Kanoa was the only one with a larger PVA reduction rate. According to the industry on the 31st, the upper limit of Angook Kanoa did not meet the BA directly in the re-evaluation of the upper limit amount, so the upper limit was decided to be reduced from 471 won to 445 won. According to the Ministry of Food and Drug Safety, Kanoa soft capsule is produced by Seoheung, and Kanoa tablets are commissioned by Vivozon. In this reassessment of the upper limit, the choline alfoscerate formulation was divided into 523 won if all the requirements were met, and 445 won if only one requirement was met. It is said that there are no items that do not meet both requirements, including DMF. Accordingly, the re-evaluation rate of Kanoa's cap was -5.5%. However, the actual upper limit, which will take effect from the 5th, is lower than this. The reason for this is that the cut rate of -9.3% was applied by the usage-drug linkage negotiations. The final cap is 427 won, which is the lowest price among the choline alposerate formulations. Kanoa's outpatient prescription amount was 6.8 billion won as of last year's Ubist, up 68% from the previous year. This time, there are a total of 18 items in which the drug price will be reduced at the same time according to negotiations on the re-evaluation of the upper limit and the PVA type. Among them, the results of the PVA with a large reduction rate of Kanoa have been applied. On August 23rd, the upper limit of the re-evaluation of the upper limit, which was disclosed earlier, was marked as 445 won. However, pharmacies and wholesalers seem to need to be careful because the upper limit of Kanoa is finally adjusted to 427 won.
- Policy
- MOHW ‘is discussing expanding non-face-to-face treatment’
- by Lee, Jeong-Hwan Sep 01, 2023 05:44am
- The Ministry of Health and Welfare formalized the expansion of the scope of its non-face-to-face medical treatment pilot project, and announced plans to expand the scope of first-time examinations, ease the standards for re-examination, and increase the ‘within 30 days of initial treatment’ term allowed for reexamination of acute conditions. In the case of first visit criteria, the authorities will focus on reviewing the need to improve medical access in areas that lack medical institutions at night, on public holidays, and during holidays. For re-examinations, the authorities will discuss ways to improve convenience for patients and medical institutions by addressing the complex eligibility standards and expanding the 30-day term for re-examinations. On the 31st, MOHW announced that it would begin discussing improvements with the advisory group ahead of the end of the guidance period for the non-face-to-face medical treatment pilot project. The MOHW referred to Minister Kyoo-Hong Cho’s statement on how the ministry "will periodically evaluate the performance of the pilot project supplement and develop shortcomings, to prepare a stable institutionalization plan’ on the day of implementation of the pilot project. In other words, the MOHW plans to fully utilize the test bed and prepare supplementary plans, and collect opinions fit for the purpose of the pilot project. As such, the MOHW plans to focus on improving the pilot project model by discussing setting appropriate criteria for first and repeat visits based on time and region and ensuring both the safety of non-face-to-face treatment and medical accessibility. The Ministry of Health and Welfare announced that after the end of the guidance period, it will continue to monitor illegal cases, such as the prescription of drugs that are not permitted in non-face-to-face treatment, such as narcotics and medicines that can be misused or abused. Second Vice Minister Minsoo Park said, "As this pilot project prepares for the institutionalization of non-face-to-face medical treatment, we need to make various attempts and reflect the demand in the field. We plan to collect public opinion through advisory group discussions and holding public hearings and use the results in improving the pilot project model."
- Policy
- The 1st results of the pharmaceutical benefit re-evaluation
- by Lee, Tak-Sun Sep 01, 2023 05:43am
- The HIRA does not plan to disclose the first results of the benefit re-evaluation reviewed by the committee on this day. After notifying the pharmaceutical company of the results, we plan to receive objections and then disclose the results during the second review. According to HIRA and the pharmaceutical industry on the 31st, the results of this year's salary adequacy reevaluation review will be discussed as an agenda item at the committee meeting held on the 6th. The ingredients subject to re-evaluation this year are Hyaluronic acid eye drops; Rebamipide, Limaprost Alfadex, Loxoprofen Sodium Hydrate, Levosulpiride, and Epinastine, etc. Among these, the market size of Hyaluronic acid eye drops, which are used for artificial tears, is the largest at approximately 200 billion won, drawing high interest from the industry. As there is great interest in Hyaluronic acid eye drops, the results are also unpredictable. However, in the pharmaceutical industry, as there is strong opposition from medical staff and there are opinions that benefits should be maintained for elderly patients, it is expected that benefits will be maintained after social discussion. An industry insider declined to comment, saying, “There is sufficient evidence for the effectiveness of Hyaluronic acid eye drops, but I don’t know how to judge it.” There are many predictions that other re-evaluation targets will not be deleted altogether even if there are reimbursement restrictions for some indications. In the case of Rebamipide, which is used for gastritis and gastric ulcers, there are many papers on its efficacy against gastric ulcers, so it is expected that reimbursement will not be maintained. The analysis also shows that there is sufficient clinical evidence for one of the two indications for Limaprost Alfadex, represented by Opalmon. The HIRA plans not to disclose the results of the first deliberation of this committee, but to notify only pharmaceutical companies. We plan to receive objections, reconsider them, and then make the results public. Last year, after the appeal, the committee deliberation was made public in October.
- Policy
- Cold medicine bills increased by 90% due to the coronavirus
- by Lee, Tak-Sun Aug 31, 2023 05:25am
- Jeong Hae-min, head of the NHIS Pharmaceutical Management Department, is answering questions at the Professional Journalists Due to the increase in confirmed cases of COVID-19 last year, cold medicine bills alone increased by 90% compared to the previous year. Based on the past five years, claims for COVID-19-related drugs were the highest in 2022. The NHIS conducted negotiations on linking usage and drug price reduction for 36 items related to COVID-19 and agreed on a reduction rate by applying a correction plan. The NHIS Pharmaceutical Management Office announced this at a meeting with the Professional Journalists Council on the 29th. According to NHIS, as a result of analyzing the billing amount for 2,637 cold medicine and antibiotic items last year, cold medicine billing amounted to 664.5 billion won and antibiotic billing amounted to 1.0575 trillion won. This is an increase of 90.1% and 24.0%, respectively, compared to the previous year. When looking at trends over the past five years, the number of pharmaceutical bills related to COVID-19, which was approximately 1.5 trillion won from 2018 to 2019, decreased to 1.2 trillion won from 2020 to 2021 but recovered to 1.7 trillion won last year. The NHIS also applied this COVID-19 drug while conducting PVA ‘Type C’ negotiations this year. Jeong Hae-min, head of the Pharmaceutical Management Department, explained, "Starting in June, we have supported a stable supply of cold medicines by easing the reduction rate of cold medicines and antibiotics monitored by the Ministry of Food and Drug Safety through the COVID-19 medicine correction plan derived through consultation with the Pharmaceutical Association." As a result of the negotiations, 'multi-type' negotiations were conducted on 36 items related to COVID-19 (18 pharmaceutical companies, 22 same product groups), and an overall agreement was reached. The NHIS plans to prepare guidelines for coordination and negotiation in response to the situation in which some essential medicines have become unprofitable due to the rise in raw material prices after the coronavirus outbreak, preventing smooth supply and repeatedly being sold out. It has been pointed out that the data submitted for mediation negotiations is based on HIRA's withdrawal prevention drug application data, so the amount of data is large and complex, putting a large burden on the company. Accordingly, the NHIS explains that it has been forming a consultative body with the Pharmaceutical Association since March to discuss and collect opinions from companies, simplify submitted data, and determine the ratio of general management costs and profits to be reflected in cost analysis. The 7th council discussion was held until August, and the plan is to compile the collected opinions establish mediation negotiation guidelines around October, and hold a company briefing session.
- Policy
- NHIS may cover high-priced new drugs with refund-type RSA
- by Lee, Tak-Sun Aug 31, 2023 05:25am
- The National Health Insurance Service is planning to improve the Risk Sharing Agreement (RSA) Scheme and is also discussing measures to improve the Price-Volume Agreement (PVA) system to reflect rewards for the innovative value of new drugs. The Department of Drug Management at NHIS said so at a press conference with correspondents on the 29th. Hae Min Jung, Director of the Department of Drug Management at NHIS, said “We are reflecting the results of the research service that was conducted to improve the RSA system, and advancing the system to achieve both the rapid listing of high-priced drugs and financial soundness of NHI finances. The demand for expansion of RSA subjects has been increasing, so we are planning to expand the drugs eligible to apply for performance-based refund-type RSA from one-shot treatments to high-priced drugs. We are also considering improving related systems, such as by new types of financial sharing plans.” Jung also added that discussions are underway with relevant organizations and the industry to come up with measures to reduce the administrative burden. Also, additional projects will be carried out for the post-marketing control of drugs that waived the submission of pharmacoeconomic evaluation data, which HIRA is seeking to improve. Jung said, “One of the results of the research service suggested the need for post-marketing management and system improvement for drugs were exempted from the submission of pharmacoeconomic evaluation data. Therefore, we plan to refer to the results of the research service on ‘Preparing measures to improve the PE exemption system’ that has been carried out by HIRA this year and come up with additional improvement tasks.” Since December 2013, when RSA was first implemented, to August 1, 2023, 68 drugs and 123 items have received reimbursement in Korea through the RSA system, and the RSA contract for 18 drugs and 26 items has been terminated. The amount refunded by pharmaceutical companies under the RSA system amounted to KRW 328.1 billion in 2022, and the refund amount has been increasing in line with the rise in the number of RSA contracts signed over the past five years. Hae Min Jung, Director of the Department of Drug Management at NHIS, is answering questions at a press conference held on the 29th The NHIS is seeking to improve the RSA system to manage high-priced drugs. As of 2022, 14 high-priced drugs costing more than KRW 300 million per person a year had filed claims of KRW 179.3 billion, which is around KRW 420 million per patient. This accounts for around 0.8% of total drug expenditures (KRW 22.9 trillion) spent by the NHIS. 37 drugs cost more than KRW 100 million per person a year, and the claims amount of the drugs amounts to KRW 348 billion. Due to these high-priced drugs, drug expenditures in health insurance finances have been increasing by more than KRW 1 trillion every year and reached KRW 22.9 trillion in 2022. This is why NHIS is seeking ways to share the financial risks through the RSA system when listing high-priced drugs, and to minimize the increase in drug costs by reducing prices of drugs that showed a rapid increase in usage through PVA negotiations. The government is also discussing measures for applying preferential treatment to innovative new drugs when applying PVA. This part is being carried out as part of the 'Measures to Improve the Insurance Drug Price System for Appropriate Compensation for the Innovative Value of New Drugs' that is being carried out by the Ministry of Health and Welfare. Jung said, “Improvement plans, such as appropriate compensation for the innovative value of new drugs, may be announced through the Health Insurance Policy Deliberation Committee meeting at the end of September at the earliest. The plan also includes the PVA part, which includes the pharmaceutical companies’ request that new drugs that meet the innovativeness requirements should be exempt from negotiations when they are subject to PVA 3 times in a row. Whether the requests were accepted will be clear when the final plan is disclosed at the end of September.”
- Policy
- Opdivo reimb as first-line therapy in gastric cancer in KOR
- by Lee, Tak-Sun Aug 29, 2023 05:28am
- The immuno-oncology drug Opdivo will be available for use as a first-line treatment, and the administration subjects for Imbruvica and G-CSF injections will be expanded. The Health Insurance Review and Assessment Service started collecting opinions on the amendment of the reimbursement standards that contain the changes above from the 24th. The adjusted reimbursement standards will be implemented on September 1st. Like the NCCN guidelines, Opdivo will be reimbursed as first-line treatment for HER 2-negative, unresectable, locally advanced, or metastatic gastric cancer. Reimbursement will be approved for the combination treatment (first-line, palliative therapy) 'nivolumab(ingredient name Opdivo) + capecitabine + oxaliplatin,’ and combination treatment (first-line, palliative therapy) 'nivolumab + oxaliplatin + leucovorin + fluorouracil.’ More specifically, patients with PD-L1 expression positive (expression rate≥5) HER2-negative advanced or metastatic gastric, gastroesophageal junction, or esophageal adenocarcinoma are allowed reimbursement, and HER2-positive patients are excluded. In addition, patients whose disease recurred 6 months after completing neoadjuvant chemotherapy, adjuvant treatment, and curative anticancer chemoradiation therapy are also allowed reimbursement. Imbruvica Cap 140mg is approved as first-line monotherapy for patients with previously untreated chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL) who are aged 65 years or older with comorbidities. More specifically, patients over 65 years of age with previously untreated active CLL or SLL, whose ▲(CIRS) score >6, or a ▲Creatinine Clearance < 70 mL/min, With the reimbursement extension, Janssen’s Imbruvica will voluntarily lower its insurance ceiling from KRW 58,555 to KRW 48,015. The reimbursement for G-CSF injections will also be extended. It is used to treat pancreatic cancer, and the 'adjuvant FOLFIRINOX(oxaliplatin + irinotecan + leucovorin + 5-FU)' combination will be additionally approved. The G-CSF injections approved are pegfilgrastim (Neulasta Prefilled Syringe Inj), pegteograstim (Neulapeg Prefilled Syringe Inj), tripegfilgrastim (Dulastin Prefilled Syinrge Inj), lipegfilgrastim (Lonquex Prefilled Inj), and eflapegrastim (Rolontis Prefilled Syringe Inj). Among these, Kyowa Kirin Korea’s Neulasta Prefilled Syringe Inj and GC Biopharma’s Neulapeg Prefilled Syringe Inj will be voluntarily lowering their insurance price ceiling. As a result, Neulasta’s price will be lowered from KRW 778,334 to KRW 764,324, and Neulapeg from KRW 567,086 to KRW 565,952. Also, the 'oxaliplatin + capecitabine' combination therapy will be reimbursed for patients with colorectal who have received neoadjuvant or adjuvant chemotherapy.
- Policy
- Hyundai Pharm's Hypejil 3mg
- by Lee, Tak-Sun Aug 29, 2023 05:28am
- 현대약품 치매증상치료제 하이페질정 5mg, 10mg. 9월부터 3mg도 출시된다.A 3mg low-dose product is listed for the first time in a Donepezil tablet used to treat Alzheimer's. The main character is Hyundai Pharm's 'Hypejil Tablet 3mg'. This drug will be paid from September to 486 won for the party according to the calculation criteria. According to the industry on the 25th, 3 mg of Donepezil is the first time that Donepezil 3mg is paid in Korea. There have only been 5mg, 10mg, and 23mg of Donepezil tablets so far. Donepezil 3 mg a day is not an effective dose, so in principle, it is not used for more than 1 to 2 weeks. Normally, rine sedatives require caution as they can increase stomach acid or increase the motility of the digestive system due to choline esterase inhibitors. Therefore, gastrointestinal medicine is often prescribed along with Donepezil at the site. Donepezil 3mg is used as an initial dose for the purpose of reducing these gastrointestinal side effects. Since it is only used as an initial dose, the market size is small, so domestic pharmaceutical companies have not made 3 mg. But 3 mg of donepezil is sold overseas. Hypezil tablet 3mg is the upper limit of the closest product among its products, and the content is calculated at 486 won by applying the content formula. The content formula is calculated at x150% if the content of the applied product is twice the content of the compared product. Hyphenic tablet 3mg and the proximity content product is Hypejil tablet 5mg, and this product is 646 won for the party. It is actually calculated at x133% because 5 mg of Hypejil tablets is less than twice as 3mg of Hypejil tablets. Since the current 3mg low content is the only product, it is likely to maintain its monopoly until a latecomer appears. It is noteworthy whether the 3mg market of Donepezil, which is used for initial treatment for the purpose of reducing side effects, will be newly formed with the appearance of this product.